Q: Rank the following ions in order of increasing basicity. a. CH,ČH2, CH,0", CH,ÑH c. CH;CO0",…
A: Introduction: According to Bronsted Lowry's concept, an acid is a substance that can donate protons…
Q: Select the LEAST acidic compound from the following OH H3C OH ČN CH,OH CN CN
A: option - c is least acidic since conjugate anion or base is not well stablized and other options…
Q: он HO, он CI D. E
A: Acidic compound can lose its proton. Order of acidity is explained based on, that compound can…
Q: Which of the following correctly places the three compounds in order of increasing acidity? H3BO3…
A: We know that the basicity of the acids is the number of hydronium ions that the compound can produce…
Q: Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used…
A: Basicity of Nitrogen: Basicity of nitrogen depends on availability of lone pair. For example if…
Q: Rank the compounds in each group in order of increasing basicity. NH2 NH2 NH2 a. NH3 -NH2 -NH2 ON CH…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: SCH3 OCH3 'Br `N(CH3)2
A: Given four compounds and asked to arrange them to their reactivity order.
Q: Rank the following substances in order of increasing acidity: Which, if any, of the four compounds…
A:
Q: hich of the following compounds has an acidic group? A NH₂ года о ная тень D NH₂ B E НО ОН "H ОН о я…
A:
Q: . Explain how you would rank the amines given below according to their basicity, together with the…
A: Amines are basic in nature due to the presence of lone pair of electrons which is present on the…
Q: Rank the compounds in each group in order of increasing basicity.
A: a) Since the basicity of molecules depends on the tendency to donate lone pair electrons. And since…
Q: (d) Arrange the following compounds in increasing order of basicity. NH2 H CH "NH2 H H3C C D E
A:
Q: Rank the following organic compounds in order of increasing basicity. A)I< III < || CH2 B) I| < III…
A: The explanation is given below-
Q: Rank the hydrogens shown in bold in order of increasing acidity. H .H A)I< || < |II H. B)I < III <…
A: We have to predict the order of acidity of hydrogens.
Q: which of the following compound shows the strongest acidity A-ch3oh B-ch3ch2oh C-h2o D-c6h5oh 1-A…
A: D. C6H5OH shows stronger acidity.
Q: Which of the given is the strongest carboxylic acid? Çi Br CH3ĊHCOOH CH3ĊHCOOH CH;CHCOOH A B A В…
A:
Q: Rank the compounds in each group in order of increasing basicity.
A:
Q: Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used…
A: Since the basicity of N depends on 2 factors. 1) whether its lone pair electrons are involved in…
Q: Which Sn1 reaction of each pair would you expect to take place more rapidly? to + H,0 CI (1) + HCI…
A:
Q: СООН СООН СООН СООН Br NO2 CH3 CI I II III IV
A:
Q: Rank the compounds in each group in order of increasing basicity.
A:
Q: 2.51 Rank the compounds in each group in order of increasing acidity. a. b. C. OH CI CI NH2 OH OH SH…
A: Acidity of a compound means how easily a compound release hydrogen ions. Organic compounds are the…
Q: 4. Predict the product of the nitration reactions CH3 HNO3 H2SO4 CI of HO, CHO N. LOCH2CH2CH3 Br2…
A: Acidity of the compound depends on the number of electron withdrawing groups attached to it. If the…
Q: what priority functional group of ff organic compound? a. carboxyl b. hydroxide c. hydroxyl d.…
A:
Q: Criven below Some Structure Dank the aming from strongest to weakest loose? Explain (9) H₂N (b) (c)…
A:
Q: Rank the following compounds in order of increasing acidity, putting the least acidic first. CH3COOH…
A:
Q: Rank the following compounds in order of increasing basicity. CH3 H3C CH3 H3C 00 KO 11 CH3 CH3 IV A)…
A:
Q: Arrange the following compounds in decreasing order of acidity * |- CICH2CH2CH2COOH Il –…
A: Which one of the following is correct
Q: Rank the compounds in each group in order of increasing acidity.
A: a. When we see the first set of compound given we compare the electronegativity of the halogens ,…
Q: Rank the following compounds in order of increasing acidity, putting the least acidic first. CH:COOH…
A:
Q: Rank the following compounds in order of increasing acidity: ОН ОН HO- HO- ČH3 NO2 I II III IV
A: We have to rank the following given compounds in the increasing order of acidity as follows in step…
Q: Rank the compounds in each group in order of increasing basicity.
A: The basicity of a molecule is a measure of its ability to accept a proton (H+). The less…
Q: Rank the following substances in order of increasing acidity: Which, if any, of the four compounds…
A:
Q: Which Nitrogen containing compound is the most basic? B None of these OC (A) A H-CEN (B) NH4 (C) N.…
A: Lone pairs which are easily available for donation of electrons are considered as basic in nature.…
Q: 1. The structure of compound A is shown below. он NH2
A:
Q: Carboxylic Acid Derivatives Resolve the reactions.
A: Acetic a hydride with ammonia gives amide. Lactum with lialh4 gives amine and alcohol
Q: Organize the carboxylic acid derivatives below in order of decreasing reactivity. (most reactive > >…
A: Carboxylic acid which have electron withdrawing group is more reactive because in that case carbonyl…
Q: CH3CH₂CH₂COOH CHCI₂COOH CICH₂CH₂CH₂COOH 1 2 3 CH₂CHCH₂COOH CH₂CH₂COOH CH,CH,CHCOOH CI 5 Cl 6 Which…
A:
Q: Rank the following compounds from the least acidic to most acidic, provide a brief explanation. он…
A: The answer is given as follows
Q: Arrange the four compounds in increasing order according to the acidity of conjugate acid. (CH3)2NH…
A: Given Acidic order
Q: Rank the compounds in each attached group in order of increasing basicity.
A: The basicity of a molecule is a measure of its ability to accept a proton. NH3 is less basic than…
Q: Which functional group is the most acidic? a. amide b. carboxylic acid c. alcohol d. phenol e.…
A: Organic compounds are characterized by different functional groups such as amide, carboxylic acid,…
Q: Rank the following substances in order of increasing acidity || CH3CCH3 || CH3CCH2CCH3 || CH3COH -OH…
A: pKa is given by the negative logarithm of Ka which may be expressed as pKa = -log Ka where Ka is the…
Q: Which is a stronger acid? Why? CH3 CO OH or CH3 OSO OH
A:
Q: Rank the following compounds in order of increasing basicity. CH3 H3C° CH3 II 4® // H3C- CH3 CH3 N.…
A: Anion are very strong bases . Anion are present in two option III AND IV but in anion III N2O group…
Q: Rank the compounds in each group in order of increasing acidity. CI Br .COOH .COOH .COOH а. C. СООН…
A: 'Since you have posted a question with multiple sub-parts , we will solve first three sub-parts for…
Q: Rank the following organic compounds below in terms of increasing acidity, with 1 being the least…
A: In this question, we will arrange all four Compounds in Increasing order of Acidity from least…
Q: N H2NNH2 C N NH3 CEN CI C ΝΗ NH₂OH CH3 Q N CH3
A: Given, Rank each of the following sets of nitrogen bases in terms of basicity and explain your…
Q: 5. Rank the following compounds in order of increasing acidit (2 OH OH COOH NO₂ II Br III COOH IV
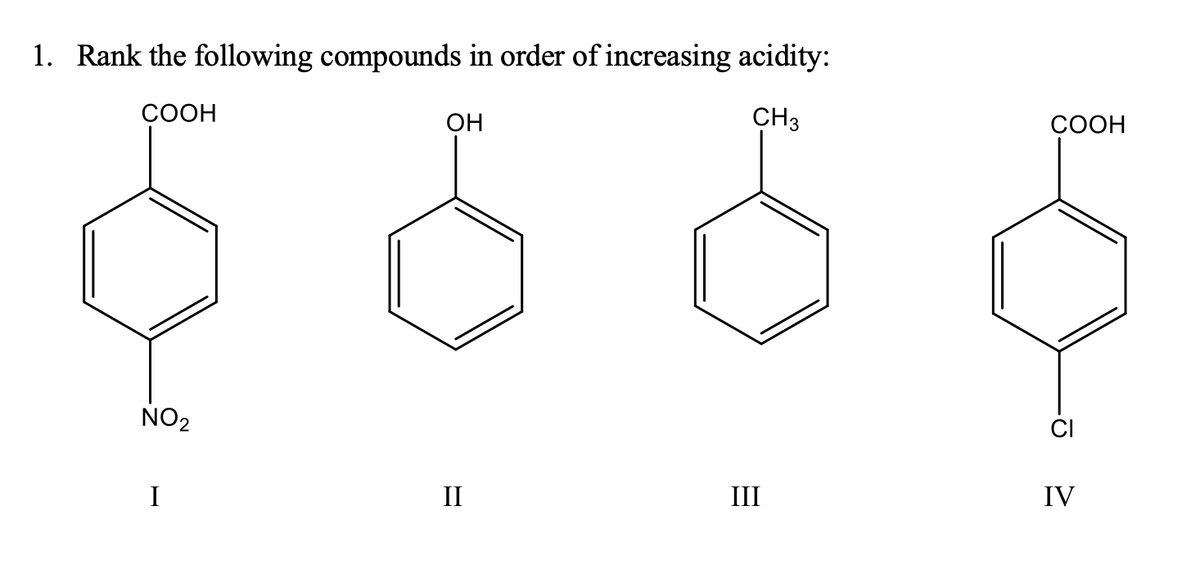
A:
please write clearly!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Rank the nitrogens in order of decreasing basicity 1, 2, and 3Aniline (conjugate acid pKa 4.63) is a considerably stronger base than diphenylamine (pKa 0.79). Account for these marked differences.Which is true regarding the direction of the following reaction? CH3COOH (aq) + H2PO-4 <<>>> CH3COO- + H3PO4 a) the reaction favors the reactant side b) the reaction favors the product side c) the reaction favors both reactants and products equally d) the table of acidity does not proviede enough information to answer this question

