Q: Rank the following compounds in order of decreasing boiling point: sodium bromide (NaBr), methane…
A: Boiling point of a compound depends on the intermolecular force. Greater is the intermolecular…
Q: CH;CH,CH,OH CH3-C-CH3 CH3CH2CH,CH3 CH3-C-OH CH;CH,OCH, ABC DEF GHI JKL MNO
A: O…
Q: Arrange the compounds in order of increasing boiling point. NH,
A: All matter can be classified as solid, liquid and gases mainly. In these physical states of matter,…
Q: Rank the following compounds in order of increasing boiling point.
A: The boiling point of the hydrocarbons is dependent upon the Van-der waale forces and strength of…
Q: Arrange the following in order of their boiling points from lowest to highest CH3CH2CH3, CH3CHO,…
A: Boiling point is a direct link of molecular attractive forces of any specified compound. It is the…
Q: Which of the following will have the lowest boiling point? I. CH3(CH2)3 CH3 II.…
A: The compound that will have the lowest boiling point is to be…
Q: А) ОН B) LOH ОН C)
A: Melting point of a substance is defined as the temperature at which the substance changes its phase…
Q: Explain why the boiling point of CH3CONH2 (221 °C) is signicantly higher than the boiling point of…
A: Given:Boiling point of CH3CONH2 (acetamide) is higher than the boiling point of CH3CO2H (acetic…
Q: Predict which compound in each pair has the higher melting point.
A: Different intermolecular forces are found between molecules that depends on the type of molecule and…
Q: Olt Cl Question 3. The boiling point of a molecule is determined by its formula weight and the types…
A: a) Since we dont have any functional group in the molecules, hence the boiling point will be…
Q: The following structure will have a boiling point. CH3 H3C-CH-CH2-CH2 ОА. High O B. Low
A: The given structure is, The given molecule is an alkane, and each…
Q: Rank the following molecules in order of increasing strength of intermolecular forces.…
A: Strength of intermolecular forces depends on the interaction of groups or bonds in the system.
Q: Rank the given compounds in increasing order based on its boiling point. (1 = lowest)
A:
Q: rank the following compounds in order of increasing vapor pressure at a constant temperature.…
A: Given rank the following compounds in order of increasing vapor pressure at a constant temperature.…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: In general molecules with high molecular mass tend to have more boiling point than molecules with…
Q: Arrange the following compounds in order of increasing their boiling points (weakest to strongest).…
A: Given here some compounds and we are asked to arrange the givencompounds in order of their boiling…
Q: Arrange the following compounds in order of increasing boiling point i.e. start with the least. Use…
A: As we know, boiling point of compound is also depends on hydrogen bonding. As hydrogen bonding…
Q: Predict which compound in each pair has the higher melting point.
A: a. When we make the comparison we see that both the compound has tha Van der Walls interaction due…
Q: >. Based on your knowledge on TMFs, explain the difference between the boiling shown below: Η Η ΤΙ…
A:
Q: Which compound in the following pairs will have the higher boiling point? Explain your reasoning.…
A: Boiling point of a compound is the temperature at which a compound boils and this happens when the…
Q: Arrange the compounds in the order of increasing boiling point (lowest first)
A: Boiling point depends upon types of interaction between molecules Types Intermolecular force of…
Q: 1. Question : Arrange the following compounds in increasing order of their boiling points, CH3CHO,…
A: Since, you have asked multiple questions we will solve the first question for you. If you want any…
Q: A B он D
A: The boiling point of aldehydes and ketones are higher than boiling point of non polar alkanes due to…
Q: Rank the following compounds in order of increasing melting point.
A: The given molecules are represented as follows:
Q: WICH or the following compounds would have the highest boiling point? CH;CH,CH,CH,CH3 O…
A:
Q: The above compound has a boiling point than pertan-1-of because it lacks
A: Hydrogen bonding: it can also be defined as the electrostatic force of attraction between hydrogen…
Q: Which of the following has the highest boiling point? ethane, CH3CH3 formaldehyde, HCHO dimethyl…
A:
Q: Which of the following will have the highest boiling point? A. CH3CH2CH2CH3 B. CH3CH2CH3 C.…
A: Boiling point of any molecule depends on 2 things ( in the same priority order as they are…
Q: List the following compounds from highest boiling to lowest boiling: Rank the compounds from highest…
A: The factors that affect the boiling point of a compound are, Molecular mass Hydrogen bonding Polar…
Q: Rank the following compounds in decreasing order of boiling points (highest to lowest): HO II II
A: The boiling point increases by increasing the carbon atoms present in the chain. But in the given…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A:
Q: Arrange the non-polar compounds P and Q below in ascending order of boiling points. Explain your…
A: 3) We have to tell among the given non-polar compound P (2,2,3,3-tetramethyl butane) and Q…
Q: Rank the following organic compounds in order of decreasing their boiling points: CH3CH2COOH…
A: Amides are form more number of hydrogen bonds than carboxylic acids, so amides has more boiling…
Q: . Rank the following with increasing acidity: CH3OH, HCl, NH3, and CH4 2. Select the correct…
A: Answer Rank the correct with increasing acidity Acidity directly…
Q: Directions: In each item, rank the given compounds according to the given description. Write only…
A:
Q: Explain why the boiling point of CH3CONH2 (221 °C) is significantly higher than the boiling point of…
A: The boiling point of any molecule depends on the intermolecular interactions between the molecules.…
Q: 1. Rank the following compounds in order of increasing boiling point: CH;CH,CH(CH3)2, CH;CH2CO2H,…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Rank the following compounds in order of increasing boiling point.
A: Extent of the ability to form intermolecular H-bonding determines the boiling point.
Q: Arrange these compounds in order of increasing boiling point (values in °C are -42, -24, 78, and…
A:
Q: Rank the given compounds in increasing order based on the melting point
A: The temperature at which a pure substance's solid and liquid phases can coexist in equilibrium is…
Q: Which of the following liquids does have the highest solubility in water at room temperature? O…
A: The compounds given are,
Q: List the following compounds from highest boiling to lowest boiling:
A: The boiling point of each compound depends on the strength of the attractive forces between the…
Q: List the following compounds in order of increasing boiling point. H2, CH3OH, H₂S, CH4 A. CH3OH <H₂S…
A:
Q: Rank the following compounds in order of increasing (lowest to highest) boiling point HO, II III…
A: First question : ( II ) compound is nonpolar and nonpolar compound have weak intermolecular forces…
Q: Place the following substances lowest to highest boiling point. Explain your answer. CH3CH2OH…
A: Boiling point order of CH3CH2OH, CH3OCH3 & H2O
Q: For each pair of compounds, circle the compound you expect to have the higher boiling point. Explain…
A: Boiling point is directly related to the strength of intermolecular forces. These forces are…
Q: List the following compounds in order of decreasing boiling point: Rank from largest to smallest. To…
A: Boiling point of a compound depends on the intermolecular force. Higher is the intermolecular force…
Q: HIUII (4d-402) Place the following molecules in order from lowest boiling point to highest boiling…
A:
Q: Rank the following compounds in order of increasing strength of intermolecular forces:…
A: Intermolecular forces: These are the forces which helps to bind molecules and it use to hold them in…
Q: Arrange the following in increasing order of boiling point: (i) CH3CH2CH2CH2Br(ii) (CH3)3Br(iii)…
A: Note: (CH3)3Br considered as (CH3)2CHBr. The boiling point is the temperature at which a liquid…
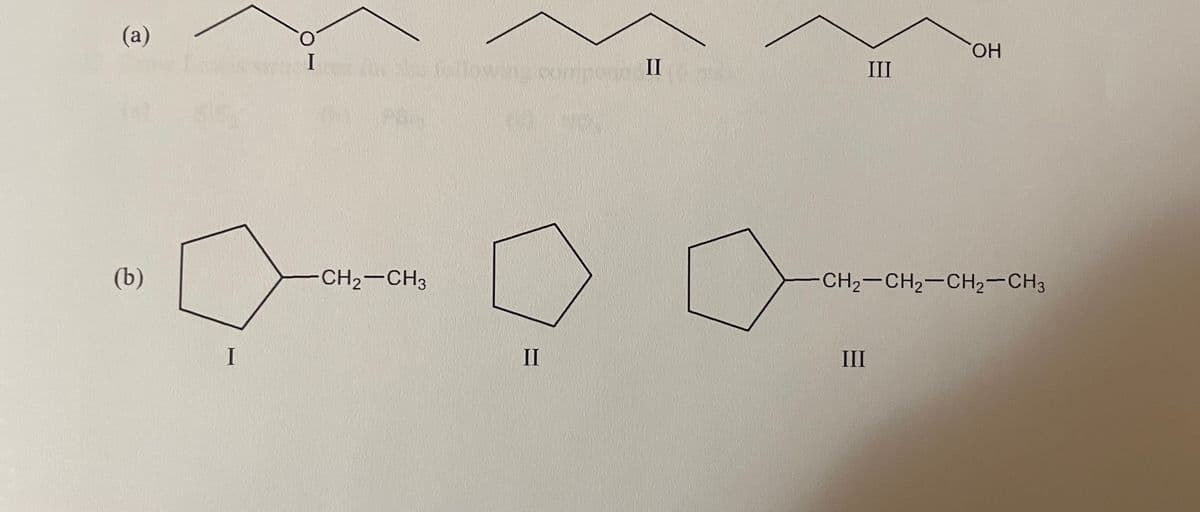
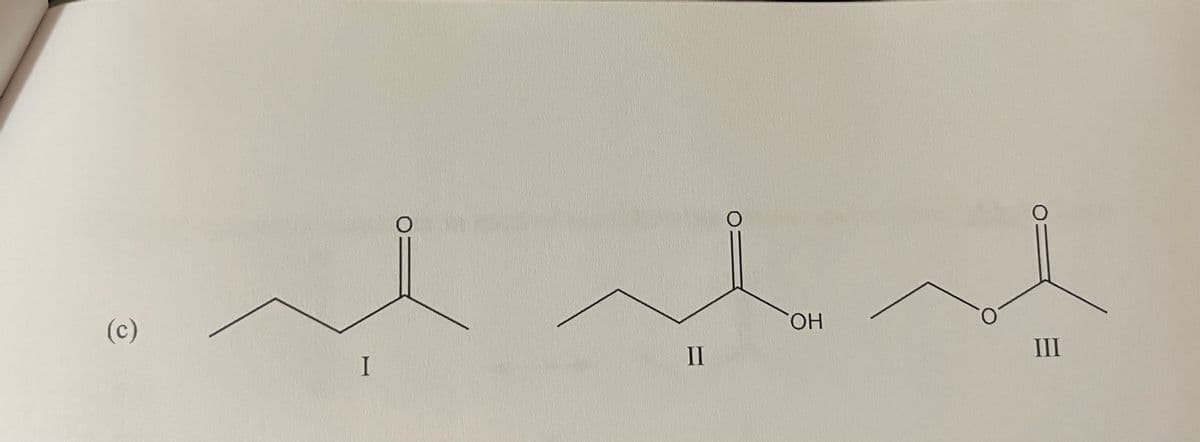
Rank the following compounds in order of increasing (lowest to highest) boiling point
Step by step
Solved in 2 steps with 1 images

- The chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever. Q.How can one enantiomer of naproxen be formed in such high yield?Trehalose, C12H22O11, is a nonreducing sugar that is only 45% as sweet as sugar. When hydrolyzed by aqueous acid or the enzyme maltase, it formsonly d-glucose. When it is treated with excess methyl iodide in the presence of Ag2O and then hydrolyzed with water under acidic conditions, only2,3,4,6-tetra-O-methyl-d-glucose is formed. Draw the structure of trehalose.Rank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −H
- Suppose we aempt the conversion of fumaric acid to deuterated malic acid with BD3·THF,followed by oxidation with D2O2in NaOD(aq). Show all the possible stereoisomers (as Fisherprojections) that may be formed, and draw the mechanistic pathways (showing stereochemistry)that lead to these possible productsBy considering the stereochemical requirements for E2-elimination, and using an appropriate illustration need help indicating whether the more stable conformer (conformer B) of trans-1 can undergo E2 elimination. Thank you :)Given the solution to Example 8.5, predict the structure of the product(s) formed when 3-hexene is treated with NBS.



