Q: Which electrophile is most likely to undergo a 1,2-hydride shift during an electrophilic aromatic…
A: In electrophilic aromatic substitution of benzene in presence of AlCl3. First of all the alkyl…
Q: Complete the following reaction sequence by drawing in the expected products. LOH SOCI₂ 1.second…
A: Note - Dear student, since the given question is a multiple question, hence I solved first question…
Q: THE T S PRODUCT WHAT RESULTENC FROM THE ELIMENATCA RxN CampovNO 1 TH F? GF OTASSIUM WITH T-BUTOXTDE…
A: The main requirement for E2 elimination is that both the leaving groups should be anti periplanar to…
Q: 5. Reactivity to EAS CHO TEN BEN MEN DECREASING REACTIVITY TO EAS:
A:
Q: D.I.2 Elechophilic Ar omatic fubs ( SEN)[ Comc eptual] CH3 C3 mol) ON ON Reactin i HンS0+ NOL ZON-…
A:
Q: Label peaks associated with the C=C double bond and the anhydride functionality in IR spectrum:…
A: Peaks in the IR spectrum of any compound represent a particular vibrational stretching of a bond.…
Q: Part B What products are formed when the following compounds react with CH3MgBr, followed by the…
A: Grignard reagent (RMgBr) gives nucleophillic addition reaction with carbonyl compounds and forms…
Q: Which set of reagents would be appropriate to synthesize bromobenzene from benzene? O1. HNO3 in…
A: HNO3 in H2SO4 gives nitration to benzene ring. Here, electrophilic substitution reaction takes…
Q: : 21:03 1 : 5. Provide the following: i) the major organic product of the reaction shown, and ii)…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: (а) 80% eе (b) 50% eе (с) 60% ee
A:
Q: Question 4 Rank the following activating groups in order of decreasing strength of a H3C- (CH3)½N-…
A: The activating groups has to be ranked in order of decreasing strength. The given compounds are,…
Q: 1. For each pair of compounds, which has the polar n bond that will undergo nucleophillic additon…
A:
Q: Generally, describe the mechanism for heterogeneous catalytic hydrogenation an provide an…
A: This question belong to stereochemistry. Mechanism have mentioned below. Here two distereomers…
Q: Complete the following multistep synthesis, depicting the possible stereochemical control in the…
A: Products of following reaction are
Q: Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S—…
A: Nucleophilicity of particular species defined as ability to share the pair of electron with…
Q: Heating the oxetane below to 560°C gives two products A and B (Scheme 2) A + CI Oxetane Scheme 2 The…
A:
Q: Which reaction(s) afford pentylamine in high yield and minimal side products 1. NaN3 A Br 2. LİAIH4…
A: We have to tell among the given reaction which are pentylamine is high yield.
Q: 0.1 a) Account for the differences observed in the pka vatues of the following saturated…
A:
Q: I OH(-) |I CH3O(-) III CH3OH IV H2O. the relative order of their reactivity in an SN2 reaction with…
A:
Q: SN2 reaction
A:
Q: What protecting group would be suitable in each of the following reactions. HOAC, THF, H,0 OH OTBS…
A: Protecting groups are respectively: 1:-"Carboxybenzyl group"(Cbz,benzyloxycarbonyl)…
Q: Identify compounds A and B, two synthetic intermediates in the 1979'synthesis of the plant growth…
A:
Q: ОН CS2CO3 o'Cs* C2H5I OC2H5 DMF heat H3CO H3CO H3CO trans-4-methoxycinnamic acid ethyl…
A:
Q: CATION ELIMINATION TESTS ADDED REAGENT(S) OBSERVATIONS NaOH Dark brown ppt Excess NaOH Undissolved…
A: According to conditions given above , the possible cation should be Fe3+.
Q: During nitration of chlorobenzene, the sigma complex formed from meta attack will have additional…
A: During migration, +M groups form ortho and para products.
Q: Identify the reagents X in the following scheme ОН NaBH4 with H30+ KMN04 with H30+ O All are correct…
A:
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4-dimethylpentane,…
A: The substitution of one of the hydrogen of the alkane group with a halogen atom is called…
Q: The stabilized ether contains a stadilizer-antioxidant?
A: There are four statements about stabilization of ether contains a stadilizer antioxidant..... A)…
Q: Select the electrophile from which the major product is formed.
A: The given organic reaction is a Friedel-Crafts alkylation reaction which takes place in presence of…
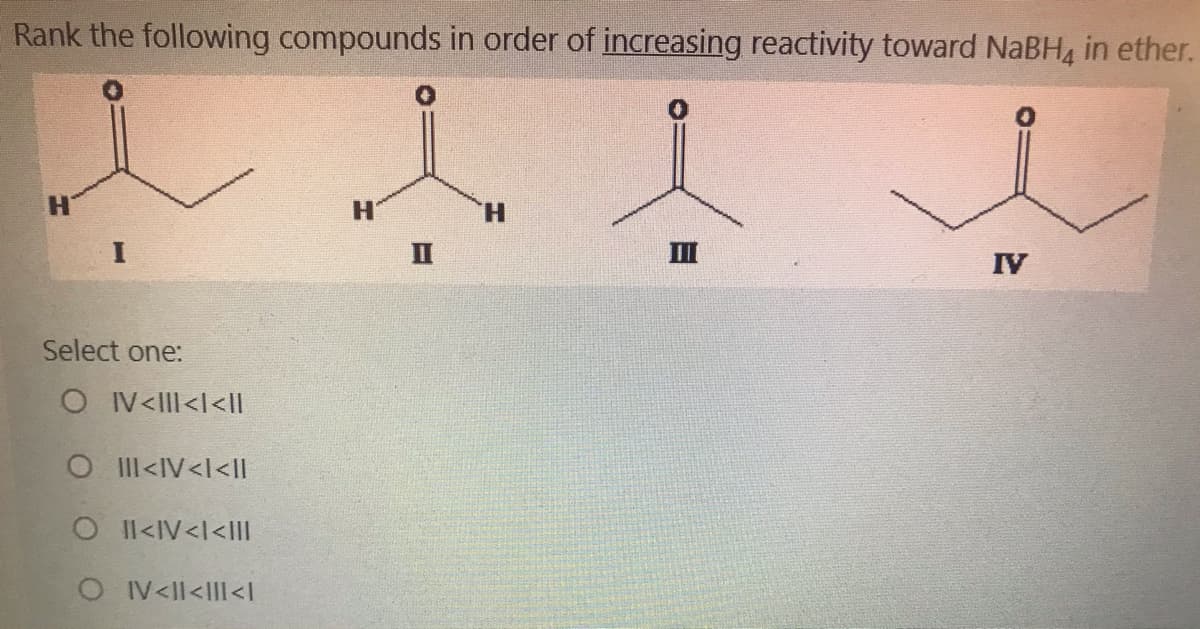
Q: Rank the following compounds in order of increasing reactivity toward NaBH, in ether. H. H. H. II II…
A:
Q: 18) Obtain all possible products starting from benzene. HNO3 Zn H₂SO4 HCl(aq) Et-Li (excess) H3O+ 2…
A: Nitration of benzene occurs in presence of HNO3 and H2SO4. Reduction of nitro group occurs in…
Q: 1. Why P(CH₂CH₃)₃ dissociiation is faster than PF3? 2. What is the product from cis-[PtCl2(NH3)2] +…
A: Explanation: 1 After dissociation we get an alkene in first compound P(CH₂CH₃)₃ and an F- in the…
Q: Slow addition of organolithium reagent A to B afforded C, anintermediate in the synthesis of the…
A: To find: The stepwise mechanism of the given process
Q: d. Aflatoxin B, hemiacetal racemizes readily in basic solution. OCH3 O: OH Aflatoxin B1 hemiacetal
A: As per the rules, only the first question can be answered. Organic reaction mechanism.
Q: Rank the following compounds in order of Increasing reactivity to aromatic electrophilic acylation.…
A: The presence of groups on the benzene ring can increase or decrease its tendency for electrophilic…
Q: In the following cases rearrange the compounds as directed : (Delhi 2010)(i) In an increasing order…
A: When a molecule has nitrogen that has an unshared electron pair, it is termed as an amine.…
Q: (e) DMP, CH2CI2 РСС, СН2CI2 Pyridinium chlorochromate in Aco OAc -OAc methylene chloride solvent OAc…
A:
Q: Ch20-21 Recitation part II. Read 21.9 (except 21.9c part); 21.10; 21.5; 20.8 1. From the pairs of…
A: Hello. Since multiple questions have been posted, the first question shall only be solved in this…
Q: raw the neutral organic product for the reaction shown. Show stereochemistry clearly. Select Draw…
A: Birch reduction is an organic reaction that is used to convert arenes to cyclohexadiene. The…
Q: Rank the following compounds in order of increasing reactivity toward NaBH4 in ether. H H. II II IV…
A: Here the NaBH4 adds the H- to the more electrophilic centre . Hence the electrophilicity order of…
Q: Explain this result: Acetic acid (CH3COOH), labeled at its OH oxygen with the uncommon 18O isotope…
A: It is given that acetic acid is reacting with a base NaOH and is undergoing acidification and the…
Q: The following catalytic cycle shows and example of what? 6 5 OC Rh Ph3P Ph P 0= MPPh 3 D H со H pph3…
A: We have to tell the given catalytic cycle to indicate what. The given options are Alkene…
Q: what is the best mpound to make X o Give mechamism? compound OH (b) BY BY Br Nat
A:
Q: Predict the stability from most to least stable and explain why. SN2 Readious (a) Br Draw the…
A: We have to predict the stability from most to least stable as follows in step 2:
Q: hat are the reaction condtions for the transformation shown? Ph (HOB. Ph Ph Pd(OAc)2 PPH3 Et3N DMF…
A:
Q: Arenediazonium salts are less stable than aliphatic diazonium salts. True False
A: Diazonium salt is a group of organic compound that share one common R-NH2+X- group of functions. R…
Q: 28) Choose which of the following statement(s) is (are) not FALSE regarding the debromination of…
A: In dehalogenation of vicinal dihalides, the vicinal dihalides moeity is converted to an alekene…
Q: HO NH2 Boc NaOEt, DMSO Boc O, DMAP THE -NH2 но 80 °C Boc POBr OH POBr NH3 Boc Boc Br NaOEt EIOH 70…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- (a) Give a plausible explanation for each one of the following :(i) There are two – NH2 groups in semicarbazide. However, only one such group is involved in the formation of semicarbazones.(ii) Cyclohexanone forms cyanohydrin in good yield but 2, 4, 6-trimethylcyclohexanone does not.(b) An organic compound with molecular formula C9H10O forms 2, 4, – DNP derivative, reduces Tollens’ reagent and undergoes Cannizzaro’s reaction. On vigorous oxidation it gives 1, 2-benzene-di- carboxylic acid. Identify the compound.from the reaction of elimination of two moles of water from one molecule of ethanediol when treated with H2SO4 (concentrated) is obtained: a) C2H4 b) C2H2 c) C2H5 d) C2H6Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IVRank A, B, and C in order of increasing SN1 reactivity.What product is made when the chemical below is heated with NBS in CCl4. NBS is N-bromosuccinimide?
- Explain how the reaction of (CH3)2CHCH(Cl)CH3 with H2O yields two substitutionproducts, (CH3)2CHCH(OH)CH3 and (CH3)2C(OH)CH2CH3Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Explain the following observation. Ethyl 3-phenylpropanoate (C6H5CH2CH2CO2CH2CH3) reacts with electrophiles to afford ortho- and para-disubstituted arenes, but ethyl 3-phenylprop-2-enoate (C6H5CH= CHCO2CH2CH3) reacts with electrophiles to afford meta-disubstituted arenes.
- Arrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IArrange the following with respect to increasing reactivity to E12-bromo-2-methylbutane undergoes hydrolysis reaction with water, H2O toform compound W. Compound X and compound Y are produced when 2-bromo-2-methylbutane undergoes elimination reaction with alcoholic ofsodium hydroxide, NaOH. (ii) What is the type of reaction involved in the formation of compound W? (iii) Identify the major product of the elimination reaction between compound Xand compound Y based on Zaitsev’s rule.



