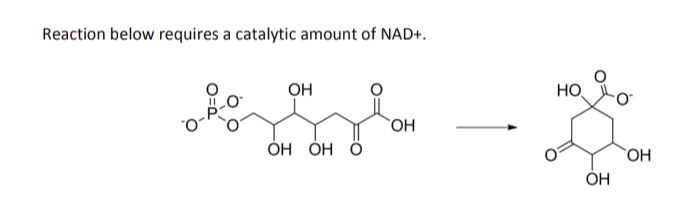
Reaction below requires a catalytic amount of NAD+. OH HO HO. ОН ОН О `OH
Q: АТР b. e. a. d. ADP + PO4 Progress of the Reaction Free Energy
A: Adenosine triphosphate (ATP) is the energy currency of the cell. The ATP is broken down when the…
Q: 4. reduced coenzyme: [Select ] [ Select ] NADH 5. pathway: I Select] FAD FADH2 NAD+ 6. final…
A: Conversion of pyruvate into acetyl CoA is called Pyruvate decarboxylation or pyruvate oxidation. It…
Q: A reduced compound isa. NAD+ b. FAD c. NADH d. ADP
A: A cell is the fundamental unit of life. All living organisms are made up of one or many cells. Each…
Q: In our dehydrogenases activity experiment, methylene blue went from blue to colorless because it O…
A: Methylene blue which is also known as methylthioninium chloride serves as a redox indicator. In a…
Q: 1.Oxygen is the final electron acceptor in the electron transprt system A. True B.False
A: Electron Transport System is located in the mitochondrial inner membrane.
Q: Drag items from the left column to the correct order in the right column. In carbon reduction, ATP…
A: This is a carbon fixation reaction, it is a part of a photosynthesis reaction in which the carbon…
Q: The synthesis of ATP from ADP and a P O is a catabolic reaction which stores energy for future cell…
A: ATP is synthesized from ADP and phosphate. ADP + Pi + free energy => ATP + H2O
Q: What metabolic nucleotides necessary for oxidation? Check all that apply. O NADH O coenzyme Q O NAD¹…
A: When Fatty acid molecule is broke down to generate acetyl - CoA then it is called as beta oxidation.…
Q: Biological oxidation-reduction reactions always involve: a. direct participation of oxygen. b.…
A: Oxidation reactions involve a loss of electrons and reduction reactions involve gain of electrons.…
Q: The phosphoanhydride bonds of ATP are termed highenergy bonds" What does high energy bond" mean in…
A: Adenosine triphosphate (ATP) is a nucleotide that consists of an adenine base coupled to a ribose…
Q: The reactions occurring as electrons move through the electron transport chain are also Redox…
A: The electron transport chain takes place in the inner mitochondrial membrane, it transports the…
Q: 1 ATP hydrolysis is _ exergonic as NADH oxidation. O Much more (more than 2x more) O Sightly more…
A: Metabolism is a term that is used to describe all chemical reactions involved in maintaining the…
Q: Click all that apply: ] affinity for protons would be increased ) pKaR would be increased here would…
A: 6 of the polar amino acids are uncharged: asparagine, glutamine, cysteine, serine, threonine, and…
Q: When optimal conditions fall out of range for enzymes, their effectiveness is lessened due to O…
A: Correct answer is denaturation Option D is correct, Denaturation When optimal conditions falls out…
Q: why does the rate of reaction level off at 6.0%
A: Answer: Introduction: Enzymes are bio-catalyst that increases the rate of a chemical reaction…
Q: What must pyruate lose in order to become ethanol? O CH3 O CO2 O NADH ATP H20
A: The cellular metabolic activities are controlled by different enzymes, coenzymes, and reaction…
Q: Catabolism breaking down anabolism building back up with products
A: The role of catabolism and anabolism is explained in step 2.
Q: R :0 + 2e + H* H,N. H' NADH NAD+
A: The term NAD denotes Nicotinamide Adenine dinucleotide. It comprises two nucleotides that include…
Q: Which of the below electron carriers is actually carrying electrons? O NADH O FAD O ATP O NAD+
A: The electron transport chain (ETC) is a very important process in aerobic cellular respiration. In…
Q: Suppose the reaction system CH4 (g) + 202(g) = CO2(g) + 2H2O(1) has already reached equilibrium. The…
A: Oxidation of methane takes place in the bacterial group called methanotrophs. Methanotrophs oxidize…
Q: Which of the following best indicates a reason why Hisneeds to be a good proton donor (not acceptor)…
A: Catalytic triad is a set of three coordinated amino acids that can be found in the active site of…
Q: Which of the following electron carriers is carrying 2 protons and 2 electrons at the same time in…
A:
Q: Let's do the Tricarboxylic Acid Cycle thing. Structural Enzyme, Formula activator and formula of the…
A:
Q: true or false NAHD generates more ATP per molecule than FADH2 beucase NADH carries more electrons…
A: The electron transport chain is the series of metabolic reactions that occurs in the inner…
Q: Oxygen is the final electron-accepting molecule in the electron transport system. The resulting…
A: Introduction An electron transport chain is a collection of protein complexes and other molecules…
Q: What type of reaction is: NADH --> NAD+ + H+ + 2 e-
A: NADH :- It is formed during the glycolysis & further reactions of cellular respiration. It is…
Q: The reactions of the glycolysis pathway are given below. The reactions are numbered 1-10; however,…
A: Metabolic pathways are made of both biosynthetic pathways and degradative pathways. They are…
Q: Categorize the electron carrier by the number of hydrogens that it can transfer in a single…
A: In the process of electron transport, one or more molecules accept electrons from another and donate…
Q: tolysis of water Select one: O Is called Hill Reaction. O All the choices are correct O Requires…
A: Plants are autotrophic, multicellular, eukaryotic organism that make their food with the help of a…
Q: Why do we have to breathe oxygen? O None of these is correct O As the oxygen component of CO2 O To…
A: WHY IS OXYGEN IMPORTANT 1. Oxygen is needed to convert food into energy Oxygen plays several roles…
Q: Model 2 – Electron Acceptor Molecules NAD+ + H+ NADH + FAD 2H+ FADH, + + ½O, 2H+ H,O
A: An electron acceptor is a chemical entity that accepts electrons transferred to it from another…
Q: In the electron transport chain, where do the electrons come from? Group of answer choices a)NADH &…
A: All of the electrons that enter the transport chain come from NADH and FADH2
Q: Catabolism is independent of anabolism. However, anabolism requires the ATP produced via catabolism.…
A: Catabolism : It is a metabolic pathway that breaks the complex molecules into smaller units like…
Q: %20₂ + H+ ⇒ NAD+ + H₂O This is the overall reaction for t Oxidative decarboxylation c Glycolysis…
A: The NADH in presence of electron acceptor oxygen converts into NAD+ and ultimately produces water…
Q: Exergonic reaction absorbs free energy from its surroundings. a. False O b. True
A: The activation energy of a reaction is the amount of energy in calories required to bring all the…
Q: Please answer fast
A: A redox reaction is a sort of chemical reaction in which electrons are transferred between two…
Q: Which of the following electron carriers are in the reduced form? Note - there may be more than one.…
A: In biological systems, a small number of chemicals act as electron shuttles, binding and…
Q: Anabolic reactions may be characterized as O 1) producing NADPH. O 2) exergonic. 3) breaking large…
A: Biomolecules are the biological molecules that are present inside the living organisms. These…
Q: other is without enzymes. For these reactions, the “X' indicates: a) b) free energy of glucose free…
A: Enzymes are biocatalyst that increases the speed of reaction by lowering the activation energy. It…
Q: In electron transport c Select one: О a. 1 АТР O b. 2ATP Ос. ЗАТР O d. None of the abe
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic…
Q: most
A: B. In plants the major source of NADH is from ferredoxin formed from the high energy electrons…
Q: Enzymes work by lowering the energy of activation. O True O False QUESTION 10 loy moro than it…
A: Enzyme acts as a catalyst in the reaction so that reaction can proceed at a faster rate but they…
Q: Given the active site diagram below, which of the indicated components is/are necessary for proton…
A:
Q: Consider the reaction: malate + NAD+ → oxaloacetate + NADH + H+. Calculate ΔE°’ for the…
A: The potential/capacity of a molecule to gain an electron is known as its reduction…
Q: Energy carried by NADH and NADPH resides in energetic electrons high energy hydrogen and/or…
A: NADH carries high-energy electrons to the electron transport chain in cellular respiration. NADPH…
Q: blocking the oxidation/reduction reaction can be led to: Select one: O a. produce water Ob. death…
A: Metabolism is a series of interconnected chemical reactions occurring within a cell; the chemical…
Q: NAD+ ____ when it gains a/an ____.
A: Nicotinamide adenine dinucleotide exists in two forms, those are oxidized from (NAD+) and reduced…
Q: While FAD is always involved in reactions that require the transfer of 2 electrons. NAD+ can…
A: Most of free energy released during the oxidation of glucose to carbon dioxide is retained in NADH…
Q: Which of the following statements are true? Explain. I. ATP synthesis will cease to occur when the…
A: Cells perform various functions. There are numerous metabolic reactions taking place inside a cell.…
Shaw step-by-step mechanism
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- A particular reaction has a ΔG‡ of 30.0 kJ mol-1 at 25.0 °C. In the presence of an enzyme, the same reaction has a ΔG‡ of 1.50 kJ mol-1 at the same temperature. Calculate the rate enhancement of this enzyme. (R = 8.3145 J mol-1 K-1)While FAD is always involved in reactions that require the transfer of 2 electrons. NAD+ can participate in reactions that transfer either 1 or 2 reactions. TRUE OR FALSE. If false, then NAD+ participates in how many electron rxns. Thanksss!Can someone help with this question? L-Malate + NAD⁺ ← → Oxaloacetate + NADH Above is what i selected for the equation, but I don't know how to find the equilibrium constant.
- Which of the following statements are true? Explain. I. ATP synthesis will cease to occur when the electron flow is blocked by cyanide. II. 2,4-DNP allows electron flow to continue in the ETC without the synthesis of ATP. III. -ΔG is the quantity that signifies that the reaction is SPONTANEOUS.All are characteristics of catabolic reactions except a. have energy poor products b. capture energy in the form of ATP c. require coenzyme like NAD+ d. yield a greater number of products compared to anabolic reactionsConsider the reaction: Ketoglomerate + NAD --> ketocitrate + NADH In the reaction to the left, NADH is the ___________ and is in a/an ______ state compared to NAD. Select one: a. substrate, oxidized b. substrate, reduced c. product, reduced d. product, oxidized
- Which of the following statements is true for the shown reaction? The reaction can occur in both cytosol and mitochondria Under starved conditions, the reaction becomes reversible to allow the synthesis of ketonebodies The reaction requires FMN as a cofactor Increase in NADH stimulates the reaction None of the aboveWhich of the following statements correctly describe(s) the reaction: ADP + Pi + H+ → ATP + H2O ΔG°'= +7.29 kcalmolkcalmol A. ATP is less stable than ADP. B. Hydrolysis of ATP is spontaneous. C. Phosphorylation of ADP is nonspontaneous. D. Conversion of ATP to ADP will release heat.CHOOSE THE CORRECT LETTER Hydrolysis of ATP yieldsA.-30.5 kJB. -30.5 kcalC.30.5 kJD. 30.5 kcal



