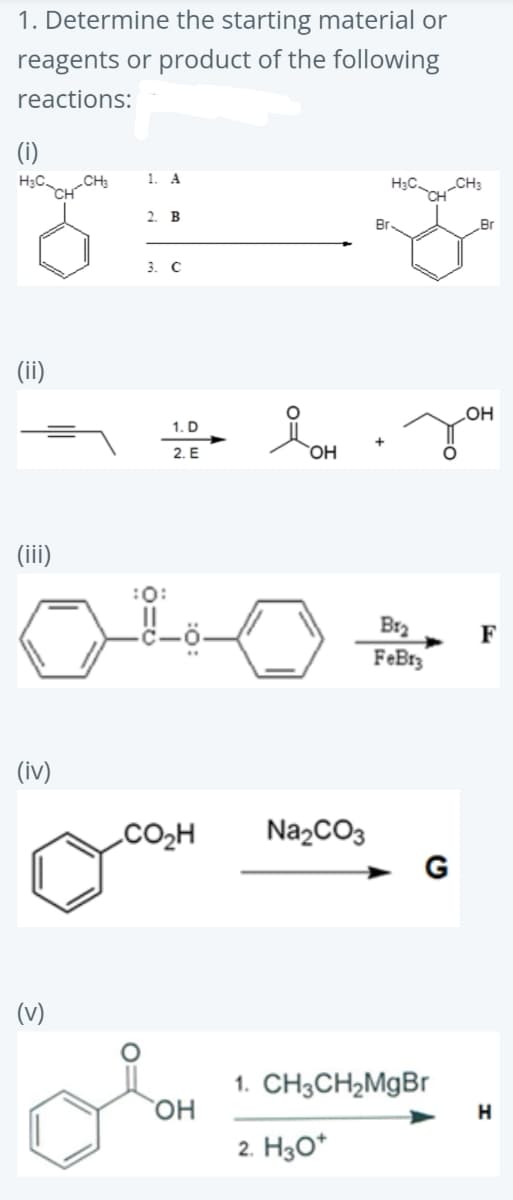
Q: 1) о ? 2) H*/H20 O a. OH Ob. OH OH Od. OH
A:
Q: 2. Pure iron reacts with sulfuric acid, H2SO4(9), to form a solutíon of iron sulfate, Fe2(SOs, in…
A: A balanced chemical equation is necessary so as to abide by the law of conservation of mass and to…
Q: Write the balanced chemical reaction 1.BaCl2(aq)+Na2SO4+NaCl 2.Cu(s)+AgNO3(s)-->Cu(NO3)2(s)+Ag 3.…
A: Solving first three subparts in accordance with guidelines. We will balance number of atoms om both…
Q: --------indicates right arrow - The ^ indicates the subscript of element/compound Match these…
A: Verification of each chemical reaction is done below-
Q: Given: K 2 CO 3 +2HCl-->2KCl+H 2 O+CO 2 a. When 0.5 moles of K 2 CO 3 are used in the reaction, how…
A: Here we have to use general maths
Q: `When you are balancing the reaction ___H2 + ___N2 → ___NH3 , what number goes in front of N2?
A: Solution Balancing chemical equations involves the addition of ratio coefficients to the reactants…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: In a balanced chemical equation the number of atoms, of all the elements involved in the reaction,…
Q: Reactions. 6. KMNO4
A: KMnO4 is strong oxidising agent At leat one benzylic hydrogen containing alkyl benzenes upon…
Q: (0) HO. 1. Br, PBr3 (0) 2. H20 name of reaction (p) 1. excess CH 2. Ag20, H20 (p) -NH name of…
A: Answer to o, p and q:
Q: Data Table 2 contains nine double displacement reactions. Write a balanced chemical equation for…
A: Balancing reaction:Balanced reaction is a chemical reaction in which number of atoms for each…
Q: 41.56 g of CaCO3(s). CaO(s) CO2(g) -> CACO3(s)
A: Applying mole concept and gas equation formula P1V1/ T1= P2V2/ T2
Q: grams of the excess reagent are left over after the reaction is complete. Pb(NO3),(s) +2 KI(s) → 2…
A: Given that, mass of KI and Pb(NO3) 2 And find limiting reagent.
Q: According to the following reaction: 3Cl, (g) + 3H,0 (1) SHC1 (aq) + HCIO3 (aq)
A: We have to convert grams of Ck2 to moles of water
Q: 19. For the following reaction, 2H2S (g) + O2 (g) = 25 (s) + 2H2O (g) Explain what happen to a. [02]…
A: The following question is based on concept of Le Chatelier's principle. Le Chatelier Principle is…
Q: HNO3 (aq) LIOH (aq) HNO3 (ag) + LIOH (aq) → H:O () + LINO (aq) HNO3 (aq) + LIOH (aq)H20 (1) +…
A: Answe. HNO3 (aq) + LiOH (aq) = H2O(l) + LiNO3 (aq)
Q: What si na limiting reactant of NaOH/0.05moles+HCl/0.05moles=
A: Given: To find limiting reactant of reaction of NaOH/0.05moles HCl/0.05moles
Q: Given the reaction 2 Cl2 (g) + 2H₂O(g) → 4HCl(g) + O2(g) What is AHO of this reaction? AHO Cl₂ = 0…
A:
Q: CH4(g)+2O2(g)→CO2(g)+2H2O(l)CH4(g)+2O2(g)→CO2(g)+2H2O(l) Use the series of reactions that follow:…
A: Using Hess's law we have to determine the enthalpy change of the reaction. The enthalpy change of a…
Q: ____ HCl + ____ MgO ---> ____ H2O + ____ MgCl2 What coefficients will balance the equation above?…
A: EXPLANATION: Balanced chemical equation is defined as in which when we add all the masses of the…
Q: Combustion reaction of hexame is; GH14 (g) + O2(g) CO2(g) + H₂O(g) H2 Balanced reaction will be: 2…
A: 11. Ans :- Given, When hexane (C6H14) is burned in oxygen, the products are carbon dioxide and…
Q: Proactinium is a highly toxic metal and one of the rarest found on Earth. One method for preparing…
A: Stoichiometry is mainly the estimation of substance's quantity which are participating in any…
Q: he combustion of propane (C3Ha) in the presence of oxygen produces CO, and HO in the folliowing…
A:
Q: Consider the reaction 3H2 + N2 NH3 Where DH is -17.2
A: according to le chateliers principle at equillibrium any change in…
Q: reactions dete CS2 => 4.5 mol S
A: This given question is based on concept of mole concept and stoichiometry.
Q: Choose the correct product(s) of the given reaction from the choices provided: O3, Zn/H,O (ехсess)…
A: The ozone (O3) oxidizes the alkene to aldehyde. First, it makes intermediate with alkene trioxolane,…
Q: ONe 채용 CA () 1) 01 DHCI/M 3) 20 )
A:
Q: 1. What is the final overall reaction for these three reactions below? to brts Reaction 1: C3 Hs (g)…
A: According to Hess’s law of constant summation Overall reaction is 3C + 4H2 à C3H8 ΔH 4= ___kJ All…
Q: Could you complete the reaction equation with the correct chemical formula(e) for the product(s)?…
A:
Q: A. COMBINATION or SYNTHESIS REACTION: A + B→AB 1. Fe +_O2 → _Fe;O3 2. Li+_d →_ Lid _C;H2 + _F…
A: Answer A. Combination reactions 1) 4Fe + 3O2 → 2Fe2O3 2) 2Li + Cl2 →2LiCl 3) C2H2 + 2F2 → C2H2F4 4)…
Q: 97. What is the substance X in this reaction? (CH2)6N4 +2H2SO4 + 6H2O→ 6HCOH + 2(NH4)2SO4 (NH4)2SO4…
A: Balance the reaction atoms
Q: CH,CH Br (a) (a) name of reaction Br NANH2, NH, (b) (b) name of reaction Br (c) (c) name of reaction…
A: (a) ->base can give elimination reaction. (b)->NaNH2 give nucleophilic addion proceeds through…
Q: se ( x Edward V x O procedur x G traductor x E Imt rgt c x Course H X Part The x Part A A X…
A: 2.4 moles of ethane undergoes combustion reaction with 3.5 moles of oxygen. Here we have to…
Q: 50.0mL of 0.180 M sodium hydroxide solution is added to 50mL of .200 M lactic acid solution. The…
A: NOTE : The formula of Lactic acid given in question is incorrect. it will be CH3CH(OH)COOH
Q: What mass of H, SO, is needed to react compictely with 0.1g NaOH? H,SO, + 2 N2OH, + 2H,0)
A: Given, H2SO4(aq) + 2NaOH(aq)→Na2SO4(aq) + 2 H2O(l) mass of NaOH react = 0.1 g mass of H2SO4 react =…
Q: si OTMS OMe TiC14 H* F Reaction 4
A:
Q: Which reaction below is an example of a decomposition reaction? O 2 KI + Pb(NO3)2→ 2 KNO3 + Pbl2 O 4…
A: Different type of reactions: a) Precipitation reaction: In precipitation reaction an insoluble salt…
Q: Determine the physical states for the products and then determine which factor does NOT drive…
A: (i): Since KOH is a soluble salt, it is completely soluble in water. Hence state of KOH is aqueous…
Q: Mass of benzaldehyde used 1.1715 g Mass of acetone used 0.2903 g 0.291 g Mass of filter paper Mass…
A: The balanced reaction taking place is given as, Given : Mass of C7H6O taken = 1.1715 g Mass of…
Q: A student carried out the reaction shown by reacting 10.0g P4 with excess Cl2 and obtained 35.0g of…
A: Given: Mass of P4 = 10.0 g Mass of PCl3 ( actual yield) = 35.0 g Molar mass of P4 = 123.89 g/mol…
Q: 3. For which reaction will AH and AE be exactly equal to each other? A) C3H60 (1) + 4 02 (g) →3 CO2…
A: The change in enthalpy of reaction is difference between enthalpy used to break bond in a chemical…
Q: 2. Given: 2H2S(g) + O2(g) 25(g) + 2H20(g) a) What happens to (H20] if O2 is added? b) [O2] if H2S is…
A: Le chatelier principle says that equilibrium will shift in such a way so as to undo the effect of…
Q: Each step in the following process has a yield of 90.0%.90.0%. CH4+4Cl2⟶CCl4+4HClCH4+4Cl2⟶CCl4+4HCl…
A: The number of moles of carbon tetrachloride formed in the first step and HCl formed in step two are…
Q: CzH6 CI pO, log) NAOHG)-> GHy(g)+ NaCI Cag)+ H,POu 9. 26 added 33.0q soled NaOtH 40.00 L Of to 0.017…
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: duct for the following reactions. 1. LDA, THF, -78 C; 2. TMSCI; 3. heat 1. BuLi, THF, -78 C; CHS 2.…
A:
Q: Solu bility nles, leadl) indicde (P.blz) is an inseluble Comppard. Camplete the folloning reaction…
A: The chemical equations should be balanced in accordance with the law of conservation of mass. This…
Q: Use: 3 Ca(NO3)2 (aq) + 2 Na3PO4 (aq) --> 6 NaNO3 (aq) + Ca3(PO4)2 (s) to determine the limiting…
A: The given reaction is : 3 Ca(NO3)2 (aq) + 2 Na3PO4 (aq) --> 6NaNO3 (aq) + Ca3(PO4)2 (s)
Q: A +B C + D AG = -1.45 kcal/mol AG° = - RT LnKg R=0.001987 kcal/mol.K
A:
Q: HBr (aq) + KOH (aq) KBr (aq) + H2O (1) PbCO3 (s)→ PbO (s) + CO2 (3) Zn + FESO4ZNSO4 + Fe
A: Interpretation: We have to classify them as neutralization , gas forming or redox reaction.
Q: uation: Ca(OH), + 2HCI → CaCl, + 2H,0 e limiting reactant? chloric acid m hydroxide
A: Moles of Ca(OH)2 = mass/MM = 7.37/74 = 0.0996 mol Moles of HCl = M × V = 0.380 × 0.035 = 0.0133…
Step by step
Solved in 2 steps with 1 images

- when H2O2 is added into acidified K2Cr2O7 which colour appears?Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaFOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Given: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…Please complete reactions in clear handwritten of all subparts
- In the beginning of this video https://www.youtube.com/watch?v=9Ng6Zv9oLzk, what reason is given on why the dienophile likes the diene?How to prepare the Organometallic Reagents ?I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!
- please quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?What is (are) the elimination product(s) obtained from the following reaction? A)only I B)I and II C) I,II, and ,III D) II, III, and IV



