Read the following statements. Select all that are true. O For a spontaneous process, Asys S > 0 O For a spontaneous process, Asys G° > 0 For a spontaneous process, Anurr S > 0 O For a spontaneous process, Asys S < 0 For a spontaneous process, Asys G" < 0 O For a spontaneous process, Aegs G< 0 O For a spontaneous process, AunivS >0 O For a spontaneous process, Asya G > 0 Evaluate
Read the following statements. Select all that are true. O For a spontaneous process, Asys S > 0 O For a spontaneous process, Asys G° > 0 For a spontaneous process, Anurr S > 0 O For a spontaneous process, Asys S < 0 For a spontaneous process, Asys G" < 0 O For a spontaneous process, Aegs G< 0 O For a spontaneous process, AunivS >0 O For a spontaneous process, Asya G > 0 Evaluate
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter16: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 2RQ: What is the second law of thermodynamics? For any process, there are four possible sign combinations...
Related questions
Question
100%
Why is this incorrect and what is the right response?
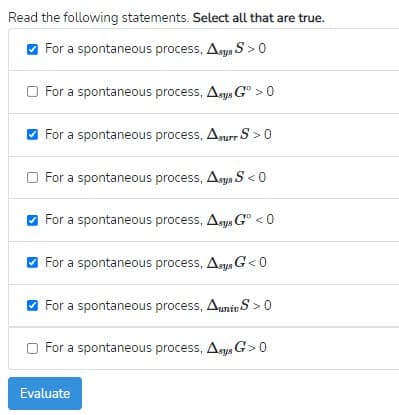
Transcribed Image Text:Read the following statements. Select all that are true.
O For a spontaneous process, Asys S > 0
O For a spontaneous process, Asys G° > 0
For a spontaneous process, Anurr S > 0
O For a spontaneous process, Asys S < 0
For a spontaneous process, Asys G" < 0
O For a spontaneous process, Aegs G< 0
O For a spontaneous process, Auniv S >0
O For a spontaneous process, Asys G>0
Evaluate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning