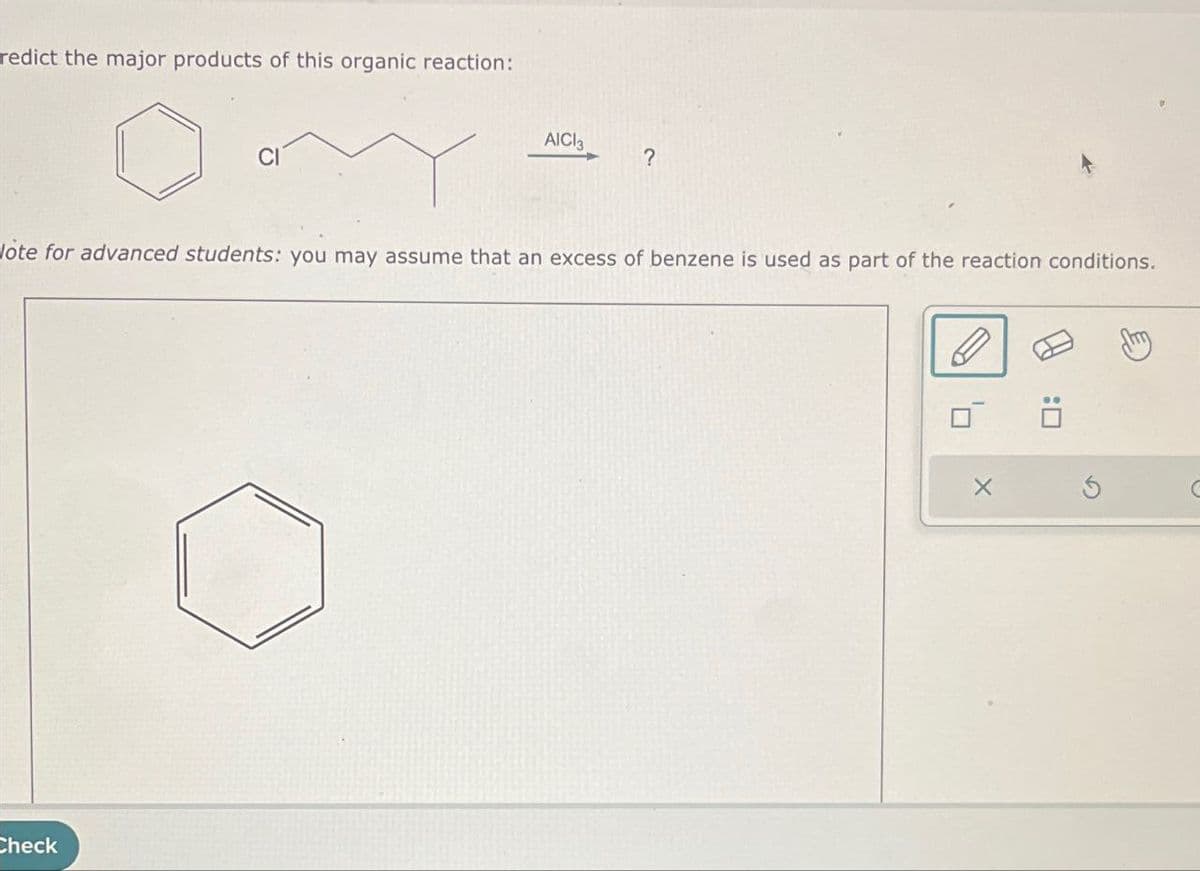
redict the major products of this organic reaction: AICI3 ? ote for advanced students: you may assume that an excess of benzene is used as part of the reaction conditions. Check X
Q: ← 68 Problem 15 of 27 Submit Draw the major product of this reaction. Ignore inorganic byproducts.…
A: Step 1:Step 2:
Q: Which statement is true about acids and bases? Brønsted-Lowry acids are proton acceptors, and…
A: The objective of the question is to identify the correct statement about acids and bases according…
Q: 5. What is the major product of the following reaction? A) (excess) OH H OH "00" B) C) D) 20 OH OH…
A: The reaction basically is protection of the ketone by cyclohexanol in presence of acid. The acid…
Q: How many grams of solid sodium hypochlorite should be added to 1.50 L of a 0.232 M hypochlorous acid…
A: The objective of this question is to determine the amount of solid sodium hypochlorite (NaClO) that…
Q: Name the following molecule using the IUPAC system но. OH
A: The objective of the question is to name the given molecule using the International Union of Pure…
Q: None
A:
Q: Why is it important to decrease the level of greenhouse gases in the Earth’s atmosphere?
A: The question is asking about the importance of reducing the level of greenhouse gases in the Earth's…
Q: Please fast expert answer solutions and dwor computer figure
A: In order to determine an aromatic compound, it must be: cyclicplanarComplete delocalization of π…
Q: Label each reactant and product in this reaction as a Brønsted acid or base. HCN+NH, →CN+NH, acid…
A: The objective of the question is to identify each reactant and product in the given chemical…
Q: What is Ksp for calcium hydroxide if the concentration of OH in a saturated calcium hydroxide…
A: The objective of this question is to calculate the solubility product constant (Ksp) for calcium…
Q: All are metabolic pathways for glucose catabolism (metabolism) EXCEPT: Question 70 options:…
A: The question is asking us to identify which of the given options is not a metabolic pathway for…
Q: Chemistry
A: Step 1: Step 2: Step 3: Step 4:
Q: Select the correct description of an indicator. a compound that changes color in the presence of a…
A: The objective of the question is to identify the correct description of an indicator in the context…
Q: P7B.1 Imagine a particle confined to move on the circumference of a circle ('a particle on a ring'),…
A: The objective of the question is to find the normalizing factor for the wavefunctions e^i*theta and…
Q: When the Ag* concentration is 1.21 M, the observed cell potential at 298K for an electrochemical…
A: The objective of the question is to find the concentration of Cr3+ in an electrochemical cell…
Q: Use oxidation numbers to identify the species oxidized and reduced in an oxidation- reduction…
A:
Q: Provide the products for the reactions shown below. Suggest a reasonable mechanism (show electron…
A: Step 1: Step 2: Step 3: Step 4:
Q: For butane, the ∆H° of vaporization is 22.40 kJ/mol and the ∆S° of vaporization is 82.3 J/mol・K. At…
A:
Q: 1:39 PM Sun Apr 14 Question 8 of 8 In this reaction: Mg (s) + 12 (s) → Mgl2 (s) If 1.84 moles of Mg…
A: In this case, for the given chemical reaction, we first identify the limiting reactant by noticing…
Q: dont provide handwriing solution ...
A: The question is asking for the reaction of an alcohol (LOH) with nitric acid (HNO3) in the presence…
Q: 1. Below are two potential methods for preparing the same ether, but only one of them is successful.…
A:
Q: Draw a correct structure of the expected product for the following reaction. -CH3 CH3 NaOH, heat…
A: Please comment down for any doubt. I hope my answer helps you.
Q: Identify and provide an explanation of the differencesbetween homogeneous and heterogeneous sampling…
A: Approach to solving the question: Detailed explanation:The concepts of homogeneous and…
Q: What are non-protein nitrogen compounds derived from? Please select all that apply. Question…
A: The objective of the question is to identify the sources from which non-protein nitrogen compounds…
Q: Draw the product of the following reaction. 1. 2. но Li
A:
Q: Question 21 Predict the FINAL (?) product for each of the following reaction or synthetic chain: H…
A: Step 1: Step 2: Step 3: Step 4:
Q: O O H3O+ OH H2O
A: The given equation is a chemical reaction involving oxygen (O), hydronium ion (H3O+), and hydroxide…
Q: None
A: To create a graph showing the relationship between Concentration and Density for a sugar solution,…
Q: Question 17 Choose the correct chemical structure for every single acronym used below: i-Prl A B C D…
A: Explanationi-prI full form is isopropyl iodide. Where i represents iso, pr represents propyl and I…
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.20 M…
A: The objective of this question is to identify which of the given aqueous solutions are good buffer…
Q: A 5.0 g sample containing a pesticide with molecular formula [C14H9Cl5], was decomposed with…
A: The objective of the question is to determine the percentage of the pesticide in the sample based on…
Q: H OH HO H Br A NaH [vahva emäs, deprotonoi OH- ryhmän] B
A: Step 1:Step 2: Step 3: Step 4:
Q: Use the observations about each chemical reaction in the table below to decide the sign (positive or…
A: The objective of the question is to determine the signs of the reaction enthalpy (ΔH) and reaction…
Q: Part A Consider the reaction A+2B C whose rate at 25 °C was measured using three different sets of…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 10 Please predict the products for each of the following reactions: 1. Na 2. PrBr เวรเ 10…
A: Step 1:At first presence of Na metal ,it released one electron , which abstract alcoholic proton and…
Q: Describe the movement of gas particles.
A: The movement of gas particles is a fundamental concept in the study of physical chemistry. It is…
Q: A buffer solution contains 0.342 M KHSO3 and 0.364 M K2SO3.If 0.0246 moles of sodium hydroxide are…
A: The objective of this question is to calculate the pH of a buffer solution after the addition of a…
Q: 6.66 x 1024 CH4 molecules mol
A: We are given with molecules of CH4 whose number is 6.66 × 1024 molecules.Number of moles can be…
Q: Calculate the K, and AG° for the following reaction at 25 °C: 2 AgNO, (aq) + Zn(s) Zn (NO,), (aq) +…
A: Step 1 The given reaction is: 2AgNO3 aq + Zns ⇌ Zn(NO3)2 aq+2Ags Equilibrium constant (Kc) of a…
Q: Question 33 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A B…
A:
Q: × Incorrect. Identify the major product(s) for the following reaction. Select all that apply. If the…
A: In bromination of alkyl in presence of hv, bromine atom will atach to secondary carbon as at…
Q: Question 29 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A…
A: Thank you.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Problem 2 of 20 46°F Sunrise C…
A:
Q: In a major human artery with an internal diameter of 5 mm, the flow of blood, averaged over the…
A:
Q: 1. Complete the following reactions: CN RMgBr CO2 02/04 MeMgBr H3O Me₂CuLi ов MeMgBr H3O Me₂CuLi
A: Step 1:• In the given reaction, RMgBr is the grignald reagent will be act as nucleophile and attack…
Q: For the reaction below which of the following answers (a-i) best describes the major product(s) you…
A:
Q: Amixture of 34.0 g of ammonia and 50.0 g of oxygen reacts according to the equation 4 NH 3 (g) + 3O2…
A: Reaction involved is 4NH3(g) + 3O2(g) -> 2N2(g) + 6H2O(g)Given mass of ammonia = 34.0 gMolar mass…
Q: A solution contains 0.0220 M Pb2+(aq) and 0.0220 M Sr²+(aq). If you add SO2(aq), what will be the…
A: Step 1: SrSO4 begins to precipitate - [Sr2+] = 0.0220 M We know at 250 C Ksp of SrSO4 is…
Q: Problem 34 of 50 Submit Draw the product that could be formed when 1,3-butadiene reacts with…
A: Step 1:Mechanism Diels-Alder Reaction:It's a pericyclic reaction, meaning all bond breaking and…
Q: In the laboratory, a general chemistry student measured the pH of a 0.580 M aqueous solution of…
A: Method 1:Dissociation equation of aqueous aniline solution will beC6H5NH2 + H2O ⇌ C6H5NH3+ + OH-Now…
Step by step
Solved in 2 steps

- In the reaction from compound 10 to compound 11, why the C=C bond is retained and is not hydrogenated? f) LiAlH4, Et2O, 08C; g) Ac2O, py, DMAP, CH2Cl2, RT, 77% over 2 steps;Organic Chemistry: 1) 2-Butanol heated with KMnO4 will produce 2-butanone 2) 2-Propanol heated with concentrated H2SO4 will produce 2-propanone? Can I have the equation :( I need to check my workHow do I calculate the percent yield based on the limiting reactant in this bromination of trans-cinnamic acid reaction? (See attachments for further information)0.149 g of trans-cinnamic acid was initial mass of the alkene 1.0 mL of glacial acetic acid (AcOH) was used 1.0 mL of Br2 was used 0.128 g of (2R, 3S)/(2S, 3R)-2,3-dibromo-3-phenylpropanoic acid was the final mas of the product
- How can you analyze or characterize a final product? What analytical techniques you can use to check the formation of the final product?2 Organic Synthesis! Palladium catalyzed reaction. Complete the reaction! Please only answer if you are 100% sure! Thank you in advance!Organic Chemistry Please help with finding the product of this reaction. Thank you
- Complete and balance the following methesis reaction in aqueous solution.Answer Entery Instructions• Parentheses are only required around polyatomic ions and only when more than 1 of those ions is present.K3PO4 (no parentheses required).Mg3(PO4)2 (parentheses required).• In each case one product is soluble (aq) and one insoluble (s). Use solubility rules to determine which product appears in each box.***Answer*** Na2S (aq) + ***Answer*** FeCl3 (aq) ➝ ***Answer*** ***Answer*** (s) + ***Answer *** ***Answer*** (aq)The addition of an alcohol to an acid chloride is an example of alcoholysis (alcohol addition with bond breakage). Consider the alcoholysis reaction below and answer the questions that follow. 1. Show the tetrahedral intermediate that is formed after the nucleophilic addition of the alcohol to the acid chloride. Be sure to include all lone pair electrons and formal charges on your intermediate structure. 2. Show the final product of this alcoholysis reaction that forms after the intermediate you made in Part 1. Do not include inorganic or charged products in your answer. Be sure to include all lone pair electrons and formal charges.Phineas and Ferbs, two brothers who enjoy vacations, doing fun things every summer. This summer the brothers and their friends carry out an organic synthesis with an unknown compound (L1) that contains 52% Carbon, 6% Hydrogen and 42% bromine, this compound (L1) is treated with magnesium in ether to obtain L2 , which reacts violently with D2O for 1-methyl cyclohexene with a deuterium atom in the methyl group (L3). The L2 reaction is treated with acetone followed by hydrolysis to give L4. Heating L4 with concentrated sulfuric acid gives L5, which decolors the bromine, obtaining L6. L5 undergoes hydrogenation with excess hydrogen and platinum as a catalyst giving rise to isobutyl cyclohexane. Determine the structures of compounds L1 through L6.
- This image is the final product that is produced. If a synthesis started with benzene and other organic compounds (four carbons or less) what would the synthesis look like to achieve this final product.Please show the steps to the organic synthesis reactions below:Solve for the starting material or product and then fill in the missing componenet to give overall reaction. Use one of this reactions I) CH3OH/H2SO4 2) 1. BH3. 2. H2O2, OH,H20 3) 1. OSO4. 2. NaHSO3 4) H3O+ 5)1. Hg(OAc)2, CH3OH. 2. NaBH4 6) Br2 in CCL4 7) Br2 in CH3OH 8)Br2 in CH3OH 9) Cl2 in H2O Show reaction mechanism



