Refer to the following phase diagram (not to scale!) for neon : 26.2 1.00 atm 0.425 24.4 24.5 27.1 44.4 T Kelvin At a pressure of 1 atm, the temperature 24.5 K is called the of Ne. The normal boiling point for Ne is at K The triple point pressure for Ne is atm The critical pressure for Ne is 26.2 atm. At temperatures above 44.4 K and pressures above 26.2 atm, Ne is a Ne is a • at 14.7 atm and 14.4 K. Ne is a at 1.00 atm and 24.9 K.
Refer to the following phase diagram (not to scale!) for neon : 26.2 1.00 atm 0.425 24.4 24.5 27.1 44.4 T Kelvin At a pressure of 1 atm, the temperature 24.5 K is called the of Ne. The normal boiling point for Ne is at K The triple point pressure for Ne is atm The critical pressure for Ne is 26.2 atm. At temperatures above 44.4 K and pressures above 26.2 atm, Ne is a Ne is a • at 14.7 atm and 14.4 K. Ne is a at 1.00 atm and 24.9 K.
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter11: Liquids And Solids
Section: Chapter Questions
Problem 11.41QE
Related questions
Question
100%
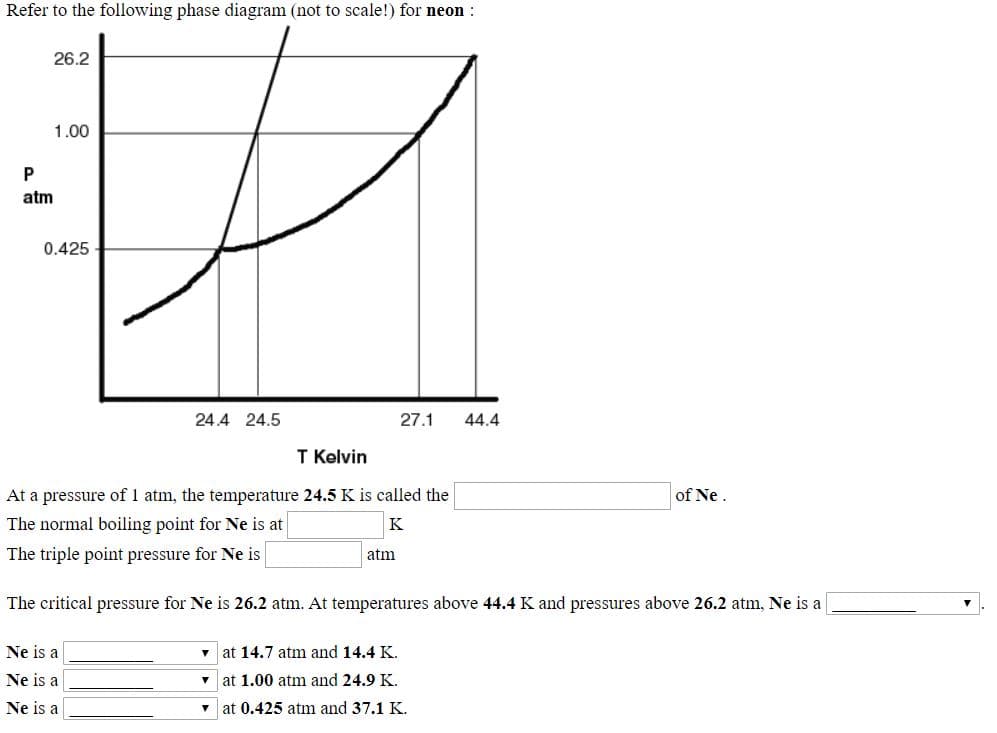
Transcribed Image Text:Refer to the following phase diagram (not to scale!) for neon :
26.2
1.00
atm
0.425
24.4 24.5
27.1
44.4
T Kelvin
At a pressure of 1 atm, the temperature 24.5 K is called the
of Ne.
The normal boiling point for Ne is at
K
The triple point pressure for Ne is
atm
The critical pressure for Ne is 26.2 atm. At temperatures above 44.4 K and pressures above 26.2 atm, Ne is a
Ne is a
• at 14.7 atm and 14.4 K.
Ne is a
at 1.00 atm and 24.9 K.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning