Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.28QAP
Related questions
Question
Using the images below, determine what is the unknown structure (Uk21).
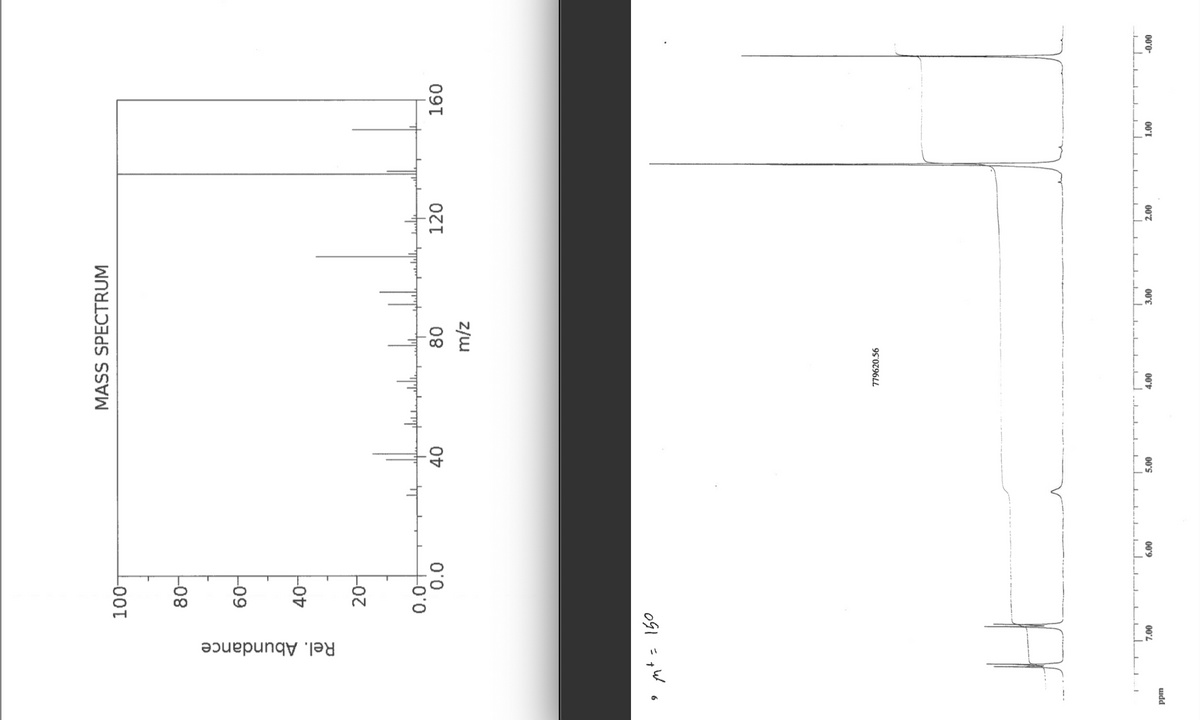
Transcribed Image Text:Rel. Abundance
00'0-
00'I
00'7
00'E
uudd
00'
00'S
00'9
00'L
779620.56
OG1 =pW o
z/w
096
120
0.0
20-
40어
F09
F08
MASS SPECTRUM
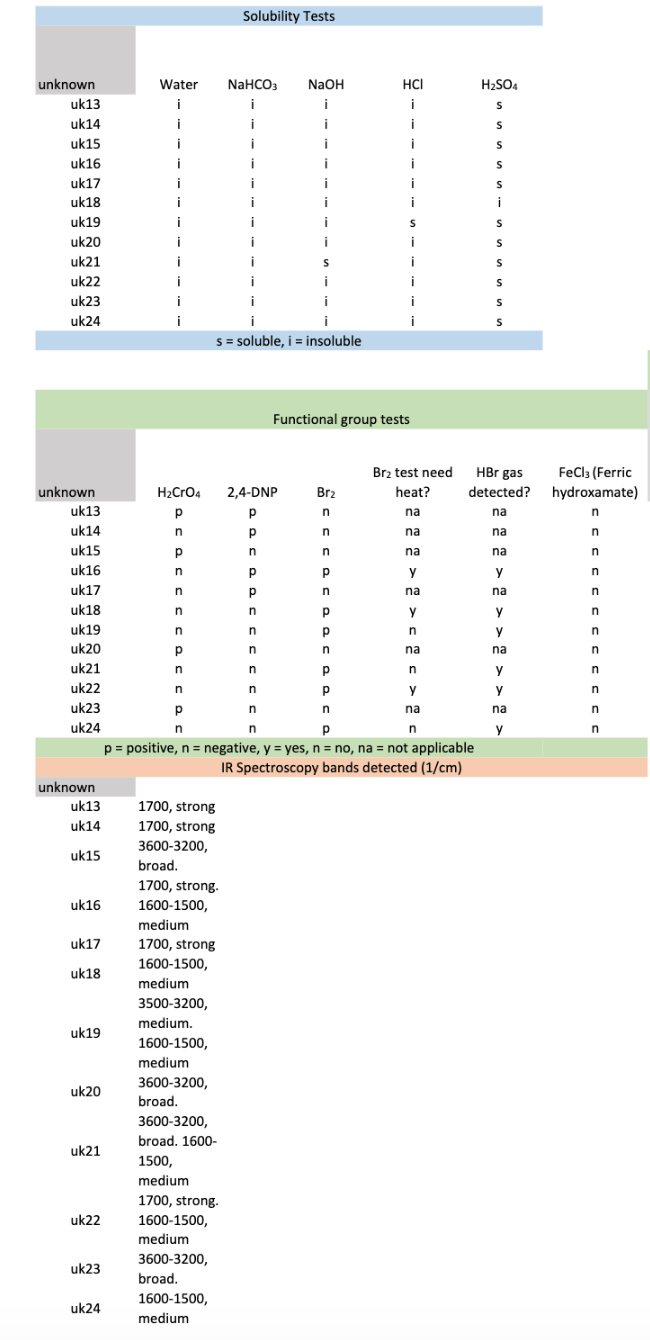
Transcribed Image Text:Solubility Tests
unknown
Water
NaHCOз
NaOH
HCI
H2SO4
uk13
i
i
i
i
uk14
i
i
i
i
uk15
i
i
i
i
uk16
i
i
i
i
uk17
i
i
i
i
uk18
i
i
i
i
uk19
i
i
uk20
i
uk21
i
uk22
i
uk23
i
i
i
i
uk24
i
i
i
S = soluble, i = insoluble
Functional group tests
FeCl: (Ferric
detected? hydroxamate)
Brz test need HBr gas
unknown
H2Cro4
2,4-DNP
Brz
heat?
uk13
na
na
uk14
na
na
uk15
na
na
n
uk16
p
y
y
uk17
n
na
na
uk18
n
y
y
uk19
y
uk20
n
na
na
uk21
n
y
uk22
y
y
uk23
n
na
na
uk24
n
n
y
p = positive, n = negative, y = yes, n = no, na = not applicable
IR Spectroscopy bands detected (1/cm)
unknown
uk13
1700, strong
uk14
1700, strong
3600-3200,
uk15
broad.
1700, strong.
uk16
1600-1500,
medium
uk17
1700, strong
1600-1500,
uk18
medium
3500-3200,
medium.
uk19
1600-1500,
medium
3600-3200,
uk20
broad.
3600-3200,
broad. 1600-
uk21
1500,
medium
1700, strong.
uk22
1600-1500,
medium
3600-3200,
uk23
broad.
1600-1500,
uk24
medium
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

