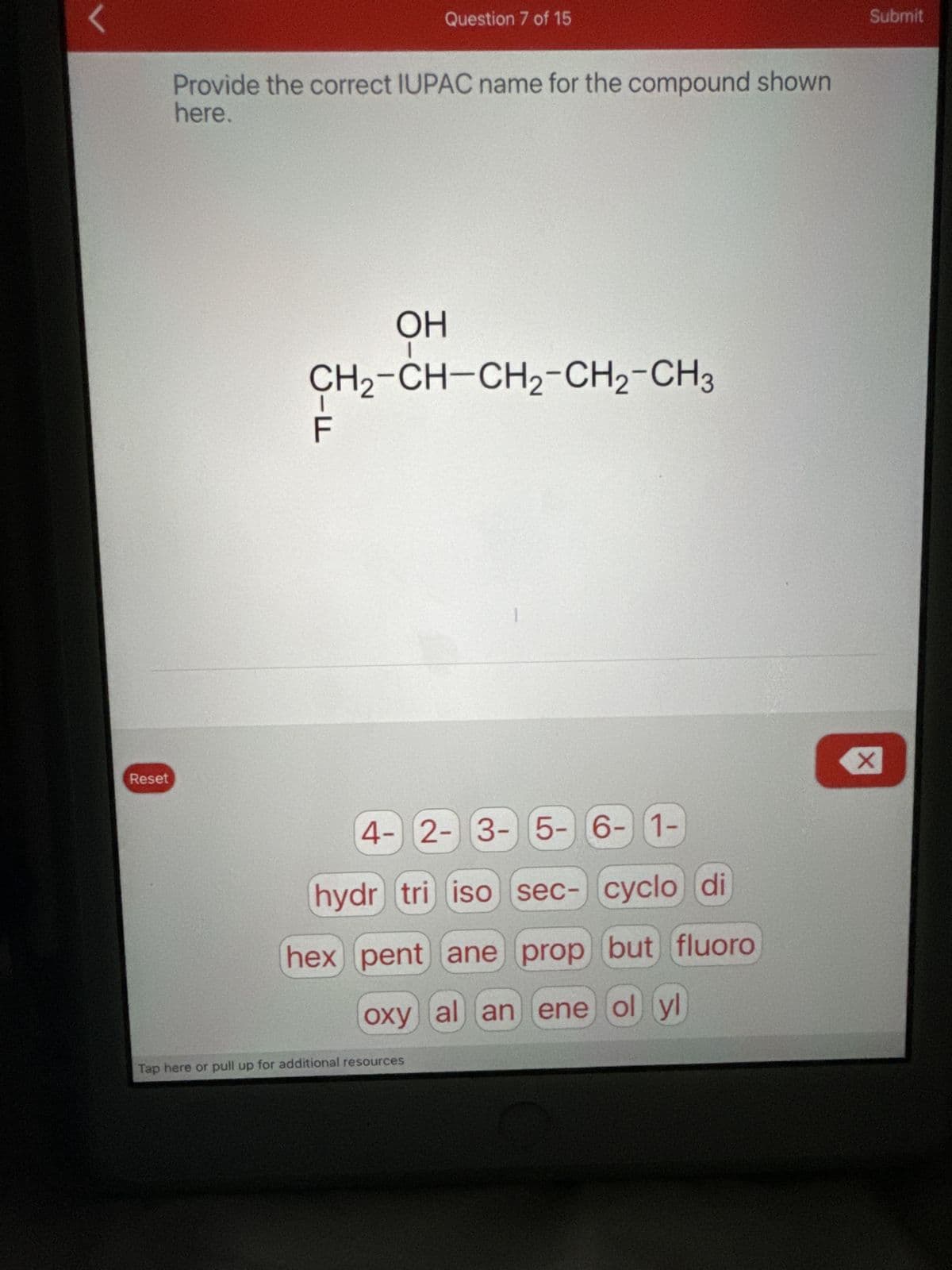
Reset Question 7 of 15 Provide the correct IUPAC name for the compound shown here. OH CH2-CH-CH2-CH2-CH3 ד 4-2-3-5-6-1- hydr tri iso sec- cyclo di hex pent ane prop but fluoro oxy al an ene ol yl Tap here or pull up for additional resources ☑ Submit
Q: Provide the major product for the following reaction? 0 H₁ OH
A:
Q: (a) Classify the transformation below as oxidation, reduction or neither. (b) Please identify the…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: (B) Identify the IUPAC name for the following compound. Br. OH A.…
A: Alkene show two stereochemistry :-Z indicates that the higher priority substituents are on the same…
Q: A scientist prepared a 0.034 M NH4Cl salt solution by dissolving the solid salt in water. Calculate…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Calculate the concentration of the hydroxide ion in a solution that contains 5.5 × 10- 5 M of…
A: [H3O+] = 5.510-5 MKw = 1.010-14
Q: The energy gap of GaAs is 1.4 eV, a lot higher than that of Ge, the element between them in the…
A: The objective of the question is to identify the reasons why is having a higher band gap than .
Q: Select all of the reagents from the list that can be used to accomplish the transformation shown…
A: The process during which the addition of H2 molecules occurs across an unsaturated double or triple…
Q: plz explain
A: The objective of the question is toWrite an equilibrium expression for the given reaction.Calculate…
Q: Predict the products and stereochemistry for the following reduction reaction NaBH4 (a) EtOH H2…
A: Sodium borohydride is a reducing agent that reduces carbonyl compounds to corresponding…
Q: What element is represented by X in the atomic symbol notation 195 is over 78X ? Select one: A.…
A: The objective of the question is to identify the element represented by X in the atomic symbol…
Q: Complete the reactions below, briefly explaining in each case type of reaction taking place. NH2 V.…
A: The missing product and the reagents has drawn with proper explanation. Please find the attachment…
Q: When you work on a modern SEM, how do you determine the charging voltage on the specimen surface?
A: A contemporary Scanning Electron Microscope (SEM) is a powerful piece of equipment that can image…
Q: Provide the major product for the following reaction? CH₂PP O
A: We have to find the major product.
Q: Draw the products of the following reactions. Remember Markovnikov's rule (if applicable). (a)…
A: The structures for the given reactions are:
Q: Which of these six compounds are expected to absorb radiation between 200 and 400 nm? (a) (b) (c)…
A: Given:The compounds are:Find the compounds which absorb radiation between 200 and 400 nm.
Q: Briefly explain the principle and application of potentiometry or pH meter or lon selective…
A: The method of measuring the voltage in a solution between two electrodes is called potentiometry.…
Q: Write structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: Information about the question
Q: (a) 11. (b) What is the major product of the elimiation below? |||-- EtONa EtOH (C) (d)
A: Given that, the reaction is:
Q: A solution contains 1.30×102 M lead acetate and 7.48×103 M calcium nitrate. Solid potassium sulfate…
A: Given:Calculate concentrations of sulfate ion when precipitation first begins. Precipitation…
Q: Acetic acid reacts with NaOH. At equilibrium the [CH3COOH] = 0.407 and the [CH3COO-] = 0.249 Ka =…
A: The objective of this question is to calculate the pH of a solution of acetic acid (CH3COOH) and…
Q: Predict the major product(s) of the following reactions. OH CHECI, AICI 3 2) -NO₂ CL₂, AICI 3 3) Br…
A: Given reaction are examples of aromatic substitution reactions.The first reaction is friedel craft…
Q: HO. Na2Cr2O7 H2SO4, H₂O SOCI₂ OH H3O+ 1. LIAI(OMe)3H 2. H3O+ excess NH3
A: The reaction scheme is shown below.We have to provide the missing products and reagents.
Q: Determine if the solution formed by each salt is pH acidic, basic, or neutral. KCIO4 Na2CO3 1.…
A: Answer:-This question is answered by using the simple concept of determination of nature of salt…
Q: What volume of 0.200 M HCI must be added to 500.0 mL of 0.300 M sodium mandelate to produce a buffer…
A: An acidic buffer solution is identified as a mixture of a weak acid (HA) with its conjugate base…
Q: chem 534 cha Hư OAc please show stereochemistry H₂COCH ? H + H H- CO₂CH3 2. 0Ac ? hv= ? 40°C 3. H₂C…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Draw the structure of the product for the following reaction and identify the type of reaction: 0 ||…
A: The reactions in which an atom or group is replaced by another atom or group are known as…
Q: A wastewater treatment plant (WWTP) releases effluent into a stream with mean depth 2 m and mean…
A: Please see the attached image for the answer.Explanation:C.In the context of river pollution…
Q: Provide all reasonable resonance structures for each molecular structure below. Use curved arrows to…
A: These are the collection of Lewis structures which represents the delocalisation of electrons in a…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The given reaction is E2 elimination.In E2- elimination mechanism;E2 = Elimination bimolecularThe…
Q: 5. Which of the following alkenes will react most quickly with HBr? Bubble in your answer…
A: To explain the reactivity of alkene with HBr. Answering only the first question as per guidelines.
Q: 5. Which of the following alkenes will react most quickly with HBr? Bubble in your answer…
A: In this reaction, carbocation gets formed as the intermediate, therefore, rate of the reaction will…
Q: please give a mechanism NH2
A: The objective of the question is to explain the reaction.
Q: raw the major products of the elimination reaction below. elimination would not occur at a…
A: The objective of the question is to predict the products formed in the following reaction given.
Q: What term best describes the Tation below? COOH H3C-CH COOH соон HC-CH3 OH HO
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: What quantity in moles of sodium atoms do you have if you have 2.10 × 10²³ atoms of sodium. (The…
A: The objective of the question is to find out the quantity in moles of sodium atoms given the number…
Q: Consider the titration of a 30.0 mL sample of 0.126 M CO32- so HI. Determine each quantity. Kb = 2.1…
A: The objective of this question is to determine the concentration of a specific reactant in a…
Q: Consider the titration of 110.0 mL analyte solution containing 0.15 mol L-1 NH3 with 0.10 mol L-1…
A: pKb = - log Kb = - log (1.80 * 10-5 ) = 4.745pKa = 14-pKb = 14- 4.745…
Q: Which of the following compounds forms an acidic solution when dissolved in water? Select all that…
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: 27 MÁ 27 Mr Dy + 8H² + 5 e¯ + Fe² = Mo² + 40+ Fe²" te
A: is given unbalanced reaction.Separating the reaction as: Oxidation half reaction: (loss of…
Q: Question 1. Indicate at which position(s) an electrophilic substitution will take place and justify…
A: To use the concept of electrophilic substitution reaction in the derivatives of benzene.Electron…
Q: The percent dissociation of chlorous acid HCIO2 in water is 8.0% when its concentration is 1.58 M.…
A: 1.Degree of dissociation of a weak acid (α) = % dissociation/1002. Ka = C × α23.[H+] = Ka × C4.pH =…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Organic reactants and products are given.We have to draw the arrow mechanism for the given…
Q: Question 2 Explain what is wrong (if anything) with each of the following calibration graphs? a) b)…
A: The calibration curve should be a straight line which should pass through the origin and the best…
Q: Balance the following redox reaction in basic solution using the half reactions method. Zn (s) +…
A: The objective of this question is to balance the given redox reaction in basic solution using the…
Q: 3. Transesterification: addition of CH3O to carbonyl 3, reform carbonyl and loss of CH3CH₂O. Ph Ph 2…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Question 4. Predict the most likely site of electrophilic aromatic substitution in each compound…
A: The rate of electrophilic aromatic substitution reaction depends on the electron density at the…
Q: Question 4: Show the major products, with stereochemistry where applicable, for the reactions of…
A: The objective of the question is to predict the major products of the reactions of 2-pentyne with…
Q: For each of the following compounds, determine the multiplicity of each signal in the 1H-NMR…
A:
Q: Question 6. Draw the mechanism of an electrophilic aromatic substitution between the following…
A: The presence of the three pi bonds in the benzene ring makes it highly electron-rich and hence it…
Q: 7)) Provide the reagent(s) necessary to carry out the conversion shown below. 0 ? он OH
A: Given reactant:So, the reactant is a cyclic ester.
Step by step
Solved in 1 steps

- Give IUPAC name of the following molecule and indicate Z or E. Answer all four of themComplete the reaction map by providing the answer from A-E. Write the IUPAC name of the products. conc HNO B conc H,SO, B, D FeBrs conc HNO, E AIC conc H2SO. conc H2SO4Look at Figure 4-12 on page 105, and estimate the percentages of axial and equatorial conformations present at equilibrium in bromocyclohexane.
- Answer the following Compounds CORRECTLY by giving the IUPAC NAME OF IT. •Label with proper notations (like hyphen, commas, periods, and grouping symbols) thank u❤️Give good explanation Asap Thanks calculate the empirical and molecular formula and degrees of saturation. Carbon= 65% Hydrogen= 6.1% Molar mass: 166g/mol what is the empirical formula? molecular formula: c9h__o3 fill in H, what is the molecular formula? degrees of saturation? round to the nearest whole number. is not 6.Using Cahn-Ingold-Prelog rules, rank these substituents from highest priority to lowest priority. A) III > I > II B) II > I > III C) III > II > I D) I > II > III E) II > III > I
- Is it option (b)1-bromo-6-chlorocyclohexene?8. Consider 1-bromo-2-methylpropane. Draw a Newman projection of the following (when viewed directlyalong the C1-C2 bond) a. staggered conformation of lowest energyb. eclipsed conformation of highest energyAssign relative priorities to each set of substituents: a. -CH2CH2CH3 -CH(CH3)2 -CH = CH2 -CH3 b. - CH2NH2 -NH2 - OH -CH2OH c. -C(=O)CH3 - CH =CH2 -Cl -C=N
- Write IUPAC names for the compounds below. If you use them, abbreviate ortho-, meta- & para- as o-, m- & p- (no italics).for the following formulas drew 2 constitutional isomers and provide names in IUPAC for ypur drawingsOChem HELP... Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form. a) Br2, CCl4 b) Br2, H2O c) OsO4, then aqueous NaHSO3 d) ICl e) mCPBA, then H3O+, H20 f) O3, then Me2S (conformational structure not required) g) BH3:THF, then H2O2, HO- h) D2, Pt i) Hg(OAc)2 in THF-H2O, then NaBH4, HO- j) BD3 :THF, then CH3CO2T



