Q: Draw the resonance structures for the following compound and circle the one that contributes most to…
A:
Q: Draw the resonance structure for the following molecule: Just started with resonance, where would…
A: Resonating structures are the structures that are different in position of electrons but not in…
Q: Name the following molecule. Use all lower case and all one word with no spaces. CI
A:
Q: For each set of molecules below circle the sets that represent valid resonance forms Be sure to show…
A: Welcome to bartleby ! We have to tell the correct resonating structures among all given molecules
Q: Among the following structures Draw resonance structures with arrows showing electron movement for…
A: The movement of electron from one place to another is called resonance structure. And higher the no.…
Q: Examine the curved arrow below, and answer the question in the box. If the curved arrow creates a…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: CH3 .N. H3C- H
A: Rules for drawing resonance structure Only the electrons move, the nuclei of atoms never move and…
Q: C. Consider compound GON whose structure is shown on the right. Experimental analysis revealed that…
A: Aromatic compounds must be cyclic, planar, conjugated, follows (4n+2)π huckel rule which means it…
Q: Lable the Hydrogens, loan pairs and formula
A: The given compound contains three N atoms, seven C atoms and thirteen H atoms. So, the formula of…
Q: || || H-C- Н—С—СН- С -CH- -C- -H-
A: In the carbanion, carbon atom forms three bonds with a negative charge and a lone pair of electrons.…
Q: Draw a curved arrow starting from the t bond of Draw all atoms of the resulting resonance structure…
A: The set of Lewis structures which explains the delocalization of electrons in a molecule is known as…
Q: Benzene is the simplest member of a whole class of compounds called aromatic hydrocarbons. benzene…
A: Given aromatic compound is benzene. Its structure is shown below:
Q: CH3 N. H. H3C-C :O:
A: Write the resonance structure that would result from pushing the electrons as indicated by the…
Q: Examine the curved arrow below, and answer the question in the box. If the curved arrow creates a…
A: The lone pair on the nitrogen atom are perpendicular to the pi-bond frame work.
Q: First, add curved arrow(s) to show the resonance using the following pattern: a pi bond between two…
A: To draw resonance structures, atleast either one pi bond and a lone pair of electrons or two pi…
Q: Three resonance structures of the given anion are possible. One is given, but it is incomplete.…
A: Resonance structure: There are many organic molecule which exhibit different structures because…
Q: (Review Toples) [References) Add the unshared (lone) electron pairs to the following structure. •…
A:
Q: Draw as many resonance structures as you can for each of the following structure, and use…
A: Resonance structures arise when there exists a conjugated double bond/vacant orbital relative to an…
Q: сompound name CH — сн, — сн, CH, — сн, — с — сH, — сн, — сн, CH, — сн, — сн — сн, CH, сH, — сH, — сн…
A: According to the question, we need to determine the IUPAC name of the given hydrocarbon. First…
Q: Draw the resonance structures of the next molecule starting with the one indicated by the curved…
A:
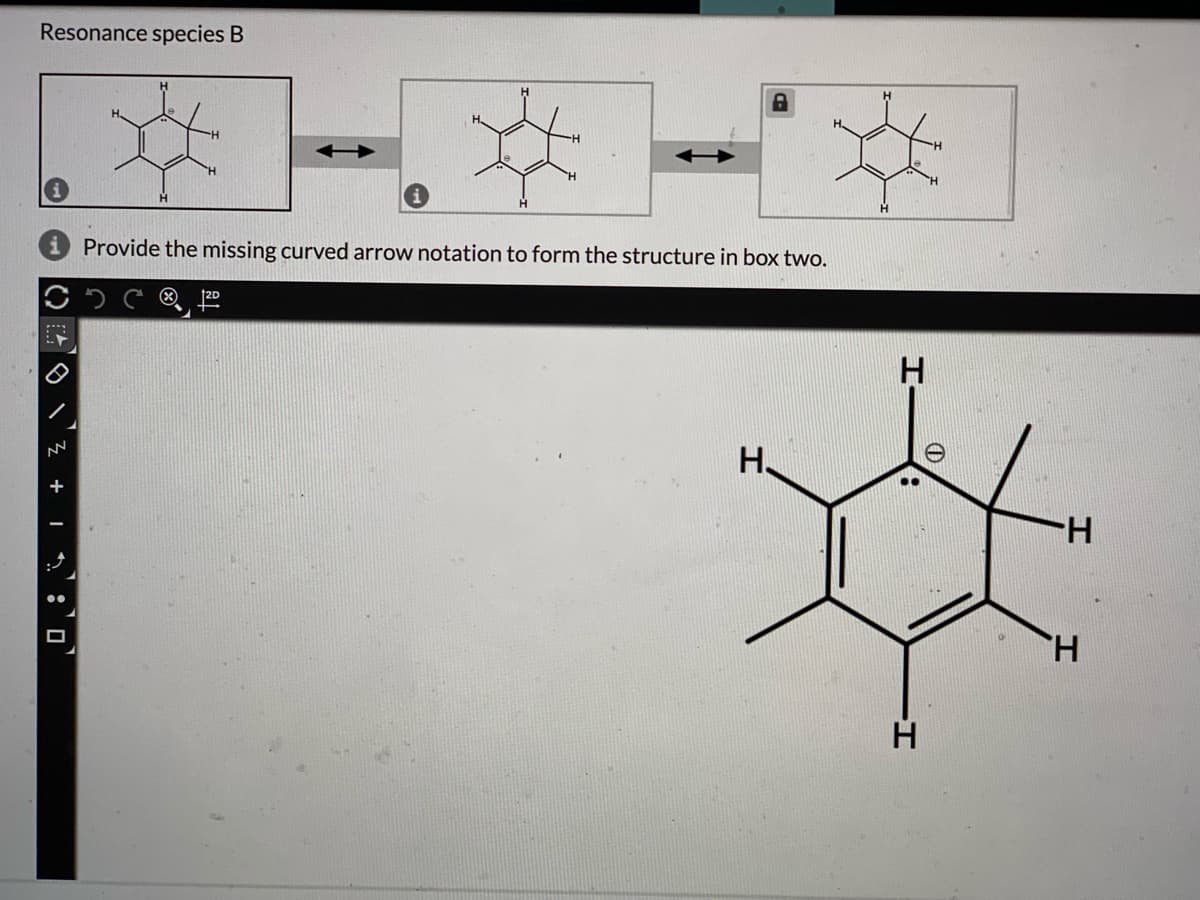
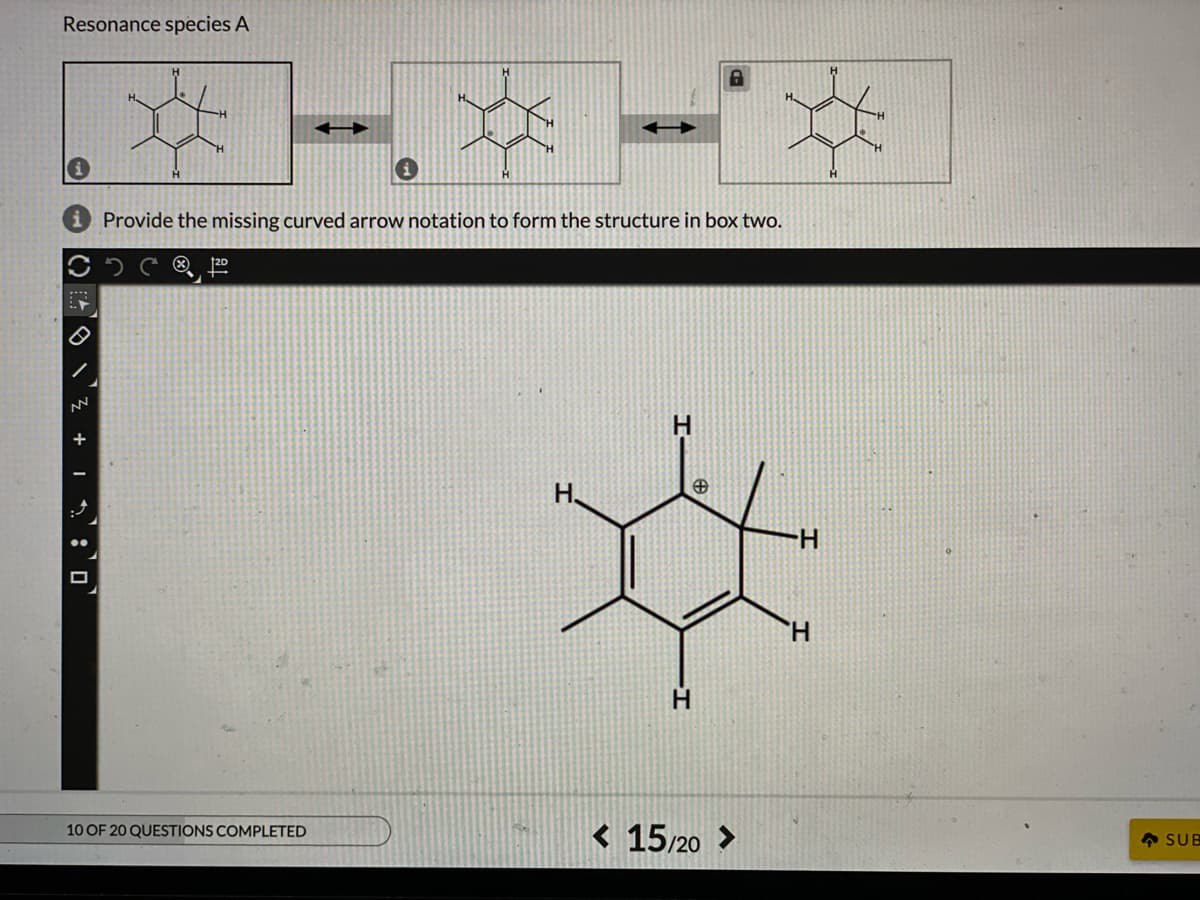
Q: Below are two sets of resonance structures. Where applicable provide the missing curved arrow…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: What happens to the bond angle (column 4) as the # of lone pairs (column 2) increases? Based on…
A: We know that order of repulsions is l.p-l.p>l.p-b.p>b.p-b.p
Q: B. Draw the correct curved arrow(s) needed to generate the resonance structures shown below and…
A: Resonance structure forms as a result of delocalization of charge over a same molecular entity.
Q: Draw the resonance structures for the following compound and circle the one that contributes most to…
A: Most contribute structure is one in which octet of all atoms is complete and negative charge is on…
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A:
Q: O Draw the curved arrows on the left resonance structure to account for formation of the right…
A: In the given structure the resonance structure is given. The curved arrow in the left structure has…
Q: Draw the curved arrow(s) for converting the first resonance structure into the second resonance…
A: Resonating structures are the structures that can exist in different forms means it's bonds,…
Q: Identify all functional groups that are present in strychnine, a highly toxic alkaloid used as a…
A: Given compound,
Q: Draw all resonance structures for the given substances. Show any relevant lone pairs of electrons,…
A:
Q: Auven", Dike dissolue like " given belowe aption Choose the Smit Similarity word
A: Polar molecules have net dipole moment and non polar molecules have zero. dipole moment
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonanre…
A:
Q: Next, add curved arrow(s) to show the resonance using the following pattern: an allylic carbocation.…
A: Here, an allylic carbocation is given. We need to modify the second structure to draw the new…
Q: Add a lone pair to NH,. Add a formal charge, Which atoms have a complete octet? if applicable. Rings…
A:
Q: Examine the curved arrow below, and answer the question in the box. If the curved arrow creates a…
A: This question is related to resonance which is a way of representing the delocalized electrons with…
Q: Describe with words how you would draw the curved arrow(s) to get to the more stable resonance form…
A:
Q: What reactant would transform the molecule from the top left to the top right. Given the same…
A: In this question, we will findout the Reagent for above transformation and for below Reaction…
Q: Add the unshared (lone) electron pairs to the following structure. • Include all valence lone pairs…
A: Given :- CH3NO2 To draw :- Its lewis structure including all unshared electrons/lone pair of…
Q: Examine the curved arrow below, and answer the question in the box. If the curved arrow creates a…
A: The curved arrows mechanism is shown as,
Q: How many σ and π bonds are present in a molecule of cumulene? (Figure 1) Enter the number of o bonds…
A: The covalent bond is formed by the overlap of the atomic orbitals between the atoms of similar…
Q: Problem: How does [BF] ion form? Problem: How does HNO, molecule ion form?
A: The details solution for this is provided below in step II.
Q: Use the "+" and "" tools to add any missing formal charges in the compound below. Please wait for…
A: Lewis structures depict the structure of particular molecule in the simplest way. It exhibits the…
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A: we have to draw the resonance structure for the given molecule
Q: Circle the most basic atom or group of atoms in each of the following molecules:
A: Hello. Since the question contains multiple sub-parts, the first three sub-parts shall only be…
Q: Draw all missing reactants andlor products in the appropriate boxes by placing atoms on the canvas…
A:
Q: Add the curved arrow(s) to each resonance structure that are necessary to convert it to the next…
A: Delocalization of pie electron in the molecule is known as resonance , and the structure obtained by…
Q: pecies is a valid resonance structure of A? Use curved arrows to show how A is converted to any .…
A:
Q: Draw all reasonable resonance structures for attached compound.
A: The possible resonance structures for given compound has to be drawn.
Q: The molecule shown here has quite a large dipole, as indicated in its electrostatic potential map.…
A: Why the given molecule has quite a large dipole , as indicated in its electrostatic potential map…
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A: The solution is given below -
Q: The ion shown has five resonance structures. ner, provide two of the resonance struuctures. •…
A: Resonance is the movement of electron without changing the position of atom .
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- For each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show how each successive r.s. was generated. b. Cross out any resonance structures that are NOT important, and explain your reasoning.Could you put them each in order from "Most electrophilic to the Least electrophillic? Thanks!pls correct me if im wronginstruction: Match rows in columns based on the chemical reactions of benzene
- Why was the formal charge of (+) placed on this carbon? I understand everything else about the question but can someone help me with explaining this. Thanks!I need alot of help on this section too!!The last part of the question asks to draw product, including non bonding electrons and formal charges. I don't understand what I've been doing wrong so any help would be great! Thank you!
- provide well-explain asnwers for the following; -)When the H dissociate from hydroxyl, the rest with different structures who can maintain the negative charge of O most ? Primary, Secondary, Tertiary. -)If the carbon connect with one or more chlorine, how about the new structure's ability to maintain negative charge ? Stronger, weaker -)Comparing the carbon connect to chlorine or fluorine, which halogen can enhance this ability ? Chlorine, Fluorine, they play same( give clear handwritten dont copy previous ans )Rewrite the entire reaction in Full Structure Format with the Ending Products [The second image is an example of Full Structure Format ] THANKSSSSSSSSSSSS

