Rock candy is formed when excess sugar is dissolved in hot water followed by crystallization. A student wants to make two batches of rock candy. He finds an unopened box of "cane sugar" in the pantry. He starts preparing batch A by dissolving sugar in 500 mL of hot water It is likely that more rock candy will be formed in batch A. It is likely that less rock candy will be formed in batch A. (70 °C). He keeps adding sugar until no more sugar It is likely that no rock candy will be formed in either batch. dissolves in the hot water. He cools the solution to room temperature. He prepares batch B by dissolving sugar in 500 mL of water at room temperature until no more I need more information to predict which batch is more likely to form rock candy. sugar is dissolved. He lets the solution sit at room temperature.
Rock candy is formed when excess sugar is dissolved in hot water followed by crystallization. A student wants to make two batches of rock candy. He finds an unopened box of "cane sugar" in the pantry. He starts preparing batch A by dissolving sugar in 500 mL of hot water It is likely that more rock candy will be formed in batch A. It is likely that less rock candy will be formed in batch A. (70 °C). He keeps adding sugar until no more sugar It is likely that no rock candy will be formed in either batch. dissolves in the hot water. He cools the solution to room temperature. He prepares batch B by dissolving sugar in 500 mL of water at room temperature until no more I need more information to predict which batch is more likely to form rock candy. sugar is dissolved. He lets the solution sit at room temperature.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter2: Chemical Formulas, Equations, And Reaction Yields
Section: Chapter Questions
Problem 46AP
Related questions
Question
I need both questions answered please.
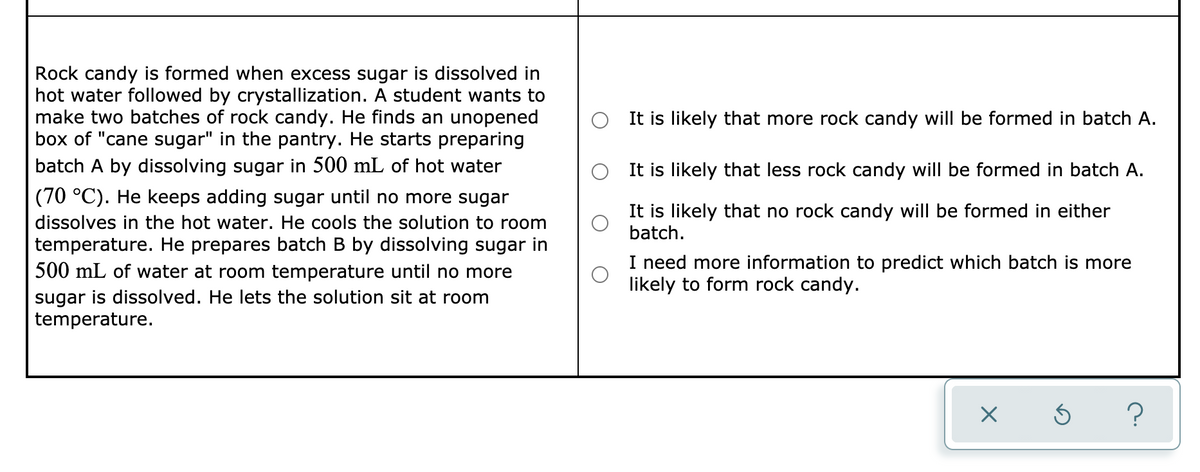
Transcribed Image Text:Rock candy is formed when excess sugar is dissolved in
hot water followed by crystallization. A student wants to
make two batches of rock candy. He finds an unopened
box of "cane sugar" in the pantry. He starts preparing
batch A by dissolving sugar in 500 mL of hot water
It is likely that more rock candy will be formed in batch A.
It is likely that less rock candy will be formed in batch A.
(70 °C). He keeps adding sugar until no more sugar
It is likely that no rock candy will be formed in either
batch.
dissolves in the hot water. He cools the solution to room
temperature. He prepares batch B by dissolving sugar in
500 mL of water at room temperature until no more
sugar is dissolved. He lets the solution sit at room
temperature.
I need more information to predict which batch is more
likely to form rock candy.

Transcribed Image Text:Predict what will be observed in each experiment below.
predicted observation
(choose one)
experiment
Can A will make a louder and stronger fizz than can B.
A student has two unopened 33 cL cans containing
carbonated water. Can A has been stored in the garage
Can B will make a louder and stronger fizz than can A.
(32 °C) and can B has been stored in the fridge (8 °C).
The student opens one can at the time, both cans make a
fizz.
The fizz will be the same for both cans.
There is not enough information to predict which can will
make the louder fizz.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning