S. No. A B 1 Years C D Investment amount Price per share No. of mutual fund shares 2 2018 3 2019 4 2020 5 2021 3250 32 =B2/C2 3250 40 =B3/C3 3250 37 =B4/C4 3250 39 =B5/C5 6 7 a) Total amount invested =SUM(B2:B5) 8 b) Total number of mutual fund shares =SUM(D2:D5) 9 c) Average cost of each mutual fund share=(C2+C3+C4+C5)/4
S. No. A B 1 Years C D Investment amount Price per share No. of mutual fund shares 2 2018 3 2019 4 2020 5 2021 3250 32 =B2/C2 3250 40 =B3/C3 3250 37 =B4/C4 3250 39 =B5/C5 6 7 a) Total amount invested =SUM(B2:B5) 8 b) Total number of mutual fund shares =SUM(D2:D5) 9 c) Average cost of each mutual fund share=(C2+C3+C4+C5)/4
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter5: Chemical Reactions
Section: Chapter Questions
Problem 5.3E
Related questions
Question
None
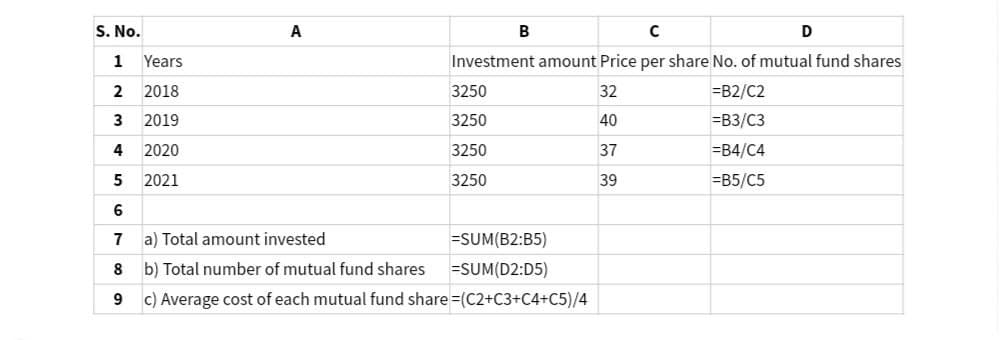
Transcribed Image Text:S. No.
A
B
1
Years
C
D
Investment amount Price per share No. of mutual fund shares
2
2018
3
2019
4 2020
5 2021
3250
32
=B2/C2
3250
40
=B3/C3
3250
37
=B4/C4
3250
39
=B5/C5
6
7
a) Total amount invested
=SUM(B2:B5)
8
b) Total number of mutual fund shares
=SUM(D2:D5)
9
c) Average cost of each mutual fund share=(C2+C3+C4+C5)/4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning