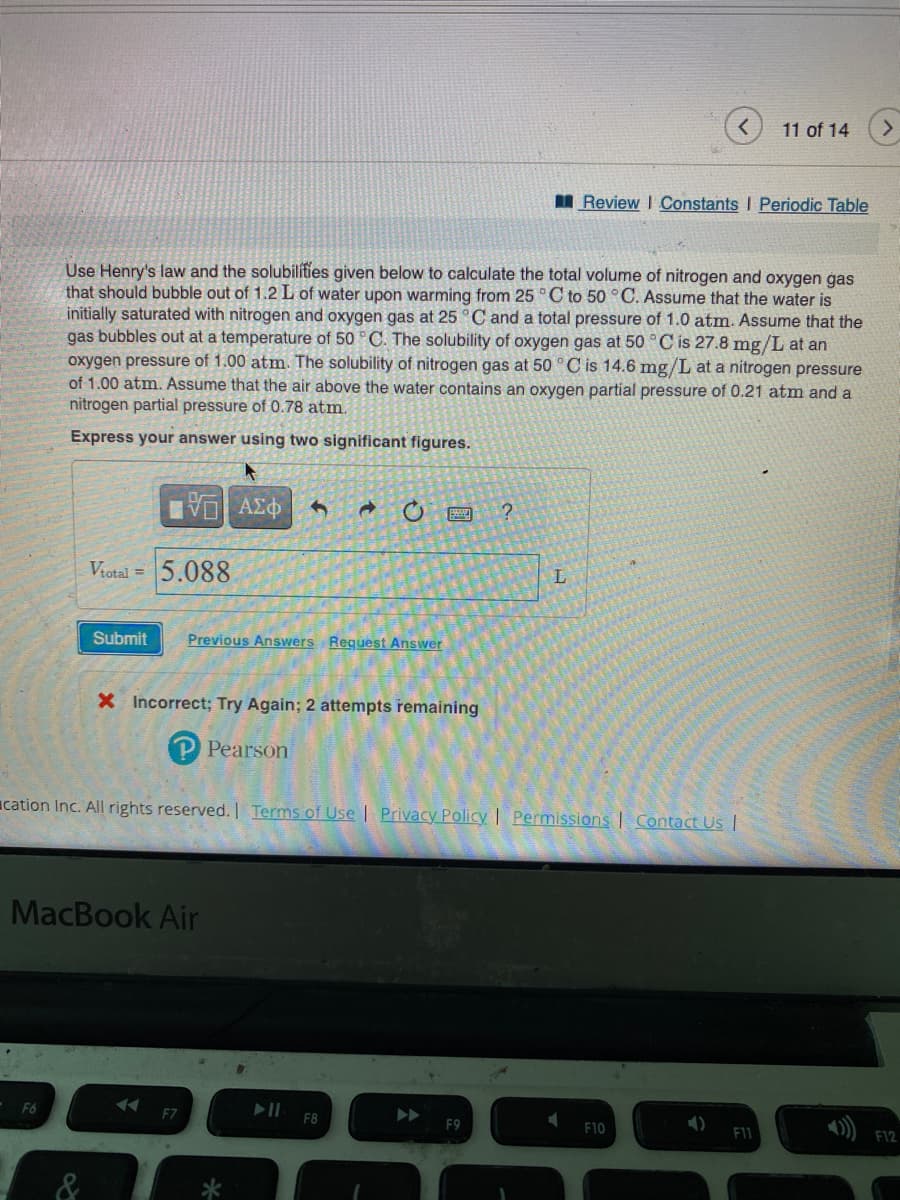
se Henry's law and the solubilities given below to calculate the total volume of nitrog- at should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume tially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 at as bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27 ygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at 1.00 atm. Assume that the air above the water contains an oxygen partial pressure trogen partial pressure of 0.78 atm. Express your answer using two significant figures. 15. ΑΣΦ Vtotal= 5.088 C Submit Previous Answers Request Answer ? L
se Henry's law and the solubilities given below to calculate the total volume of nitrog- at should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume tially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 at as bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27 ygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at 1.00 atm. Assume that the air above the water contains an oxygen partial pressure trogen partial pressure of 0.78 atm. Express your answer using two significant figures. 15. ΑΣΦ Vtotal= 5.088 C Submit Previous Answers Request Answer ? L
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter13: Solutions And Their Behavior
Section: Chapter Questions
Problem 106SCQ
Related questions
Question
Could you help me the last one
Transcribed Image Text:F6
Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas
that should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume that the water is
initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the
gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an
oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure
of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a
nitrogen partial pressure of 0.78 atm.
Express your answer using two significant figures.
Vtotal =
Submit
&
15. ΑΣΦ 1
5.088
MacBook Air
◄◄◄
Previous Answers Request Answer
X Incorrect; Try Again; 2 attempts remaining
C
Pearson
cation Inc. All rights reserved. | Terms of Use | Privacy Policy | Permissions | Contact Us |
F8
?
F9
L
Review Constants I Periodic Table
F10
11 of 14
4)
F11
>
F12
Expert Solution
Step 1
Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas that should bubble out of 1.2 L of water upon warming from 25°C to 50 °C. Assume that the water is initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a nitrogen partial pressure of 0.78 atm.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
