se the model to answer the following question. lement A Element C KEY Proton Element B Neutron Which of the following explanations are TRUE regarding the mass number and identity of Element C? O A. Element C has only a different identity than elements A and B O B. Element C has both a different mass number and identity than elements A and B O C. Element C has the same mass number and identity as elements A and B O D. Element C has only a different mass number than elements A and B
se the model to answer the following question. lement A Element C KEY Proton Element B Neutron Which of the following explanations are TRUE regarding the mass number and identity of Element C? O A. Element C has only a different identity than elements A and B O B. Element C has both a different mass number and identity than elements A and B O C. Element C has the same mass number and identity as elements A and B O D. Element C has only a different mass number than elements A and B
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter1: The Nature Of Chemistry
Section1.13: The Periodic Table
Problem 1.9E
Related questions
Question
100%
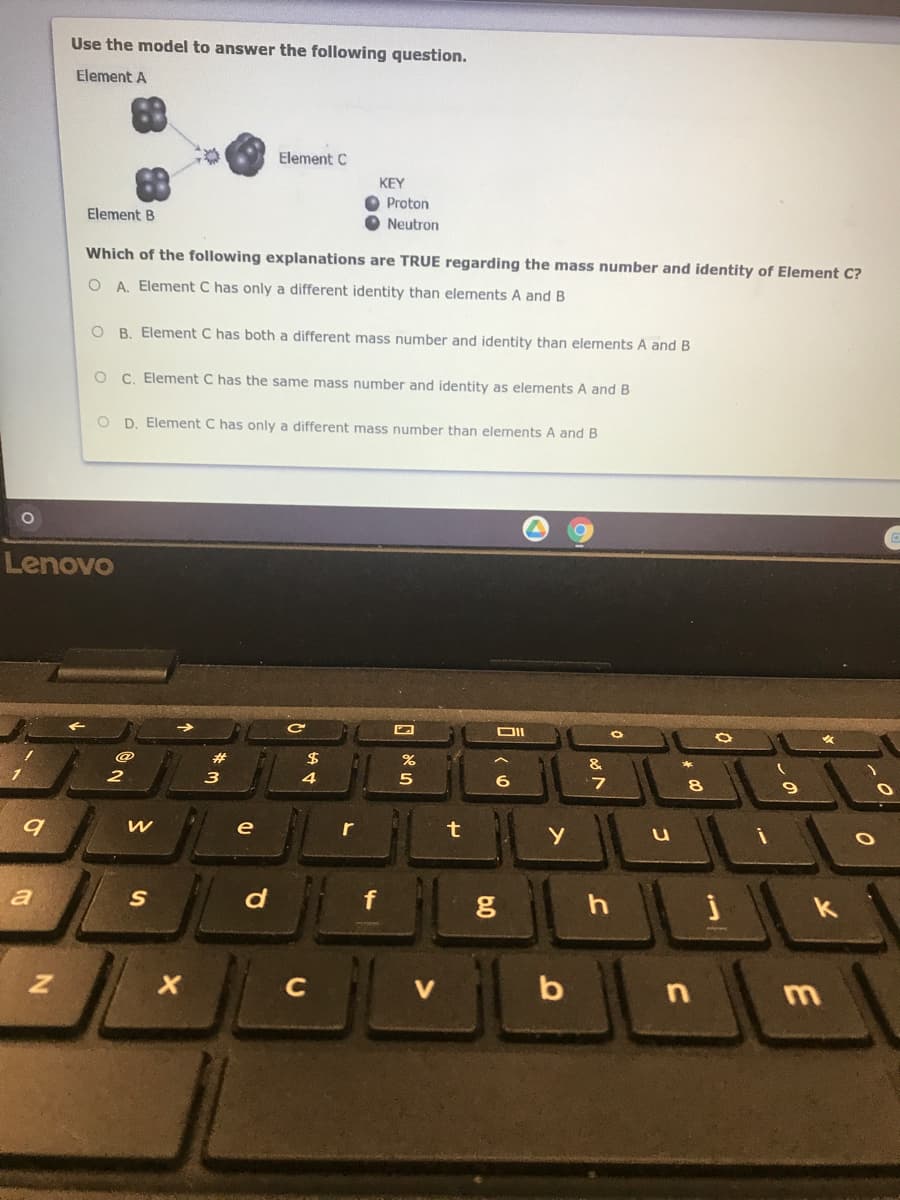
Transcribed Image Text:Use the model to answer the following question.
Element A
Element C
KEY
Proton
Element B
Neutron
Which of the following explanations are TRUE regarding the mass number and identity of Element C?
OA Element C has only a different identity than elements A and B
B. Element C has both a different mass number and identity than elements A and B
C. Element C has the same mass number and identity as elements A and B
O D. Element C has only a different mass number than elements A and B
Lenovo
DII
@
3
5
e
r
d
f
h
k
C
V
b
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning



Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning