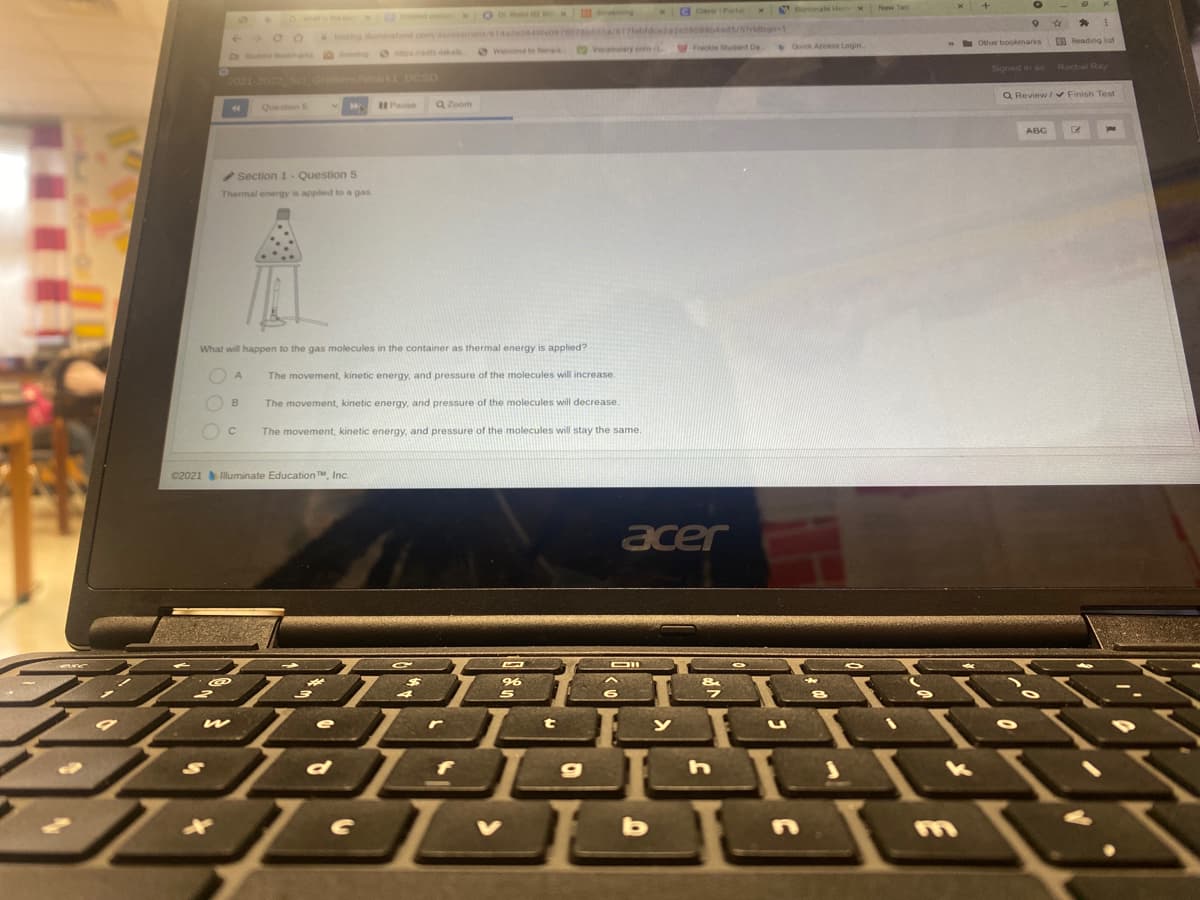
Section 1-Question 5 Thermal energy ia applied to a gas What will happen to the gas molecules in the container as thermal energy is applied? The movement, kinetic energy, and pressure of the molecules will increase. The movement, kinetic energy, and pressure of the molecules will decrease The movement, kinetic energy, and pressure of the molecules will stay the same.
Section 1-Question 5 Thermal energy ia applied to a gas What will happen to the gas molecules in the container as thermal energy is applied? The movement, kinetic energy, and pressure of the molecules will increase. The movement, kinetic energy, and pressure of the molecules will decrease The movement, kinetic energy, and pressure of the molecules will stay the same.
Chapter6: Random Errors In Chemical Analysis
Section: Chapter Questions
Problem 6.16QAP
Related questions
Question
What will happen to molecules in a container if thermal energy is apply?
Transcribed Image Text:O we wx ing
G CieverI Portal x inate Hom
tstingumnateed coma ment1dae4ote097807825a/617febfdoaaeasoss4ads/5idbgn1
Owme to na Vecabutary com
Frecke tudent De
sQuick Access Login
Other bookmarks
D om mdg . d
Signed in as Rachel Ray
2021-2022 S Granenchimark DCSD
Q Review / v Finish Test
Queston S
Pause
a Zoom
ABC
Section 1 Question 5
Thermal energy is applied to a gas
What will happen to the gas molecules in the container as thermal energy is applied?
The movement, kinetic energy, and pressure of the molecules will increase.
B
The movement, kinetic energy, and pressure of the molecules will decrease.
The movement, kinetic energy, and pressure of the molecules will stay the same
C2021 Illuminate Education TM, Inc.
acer
V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,