Select all substances with an acceptor atom that can hydrogen bond to a hydrogen atom that is part of a polar covalent bond. (CH3)2CHOH isopropanol HOCH,CH2OH ethylene glycol CH3CHCHCH3 trans-2-butene BrF5 bromine pentafluoride (CH3CH2)2NH diethylamine (CH3)2NH dimethylamine None Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
Select all substances with an acceptor atom that can hydrogen bond to a hydrogen atom that is part of a polar covalent bond. (CH3)2CHOH isopropanol HOCH,CH2OH ethylene glycol CH3CHCHCH3 trans-2-butene BrF5 bromine pentafluoride (CH3CH2)2NH diethylamine (CH3)2NH dimethylamine None Question Attempts: 0 of 1 used SUBMIT ANSWER SAVE FOR LATER
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.SE: Something Extra
Problem 51AP
Related questions
Question
How can i determine if a molecule has a molecular dipole, please give a detailed expression, my exam is at 3 so before then, preferably before 2. I provided an example below
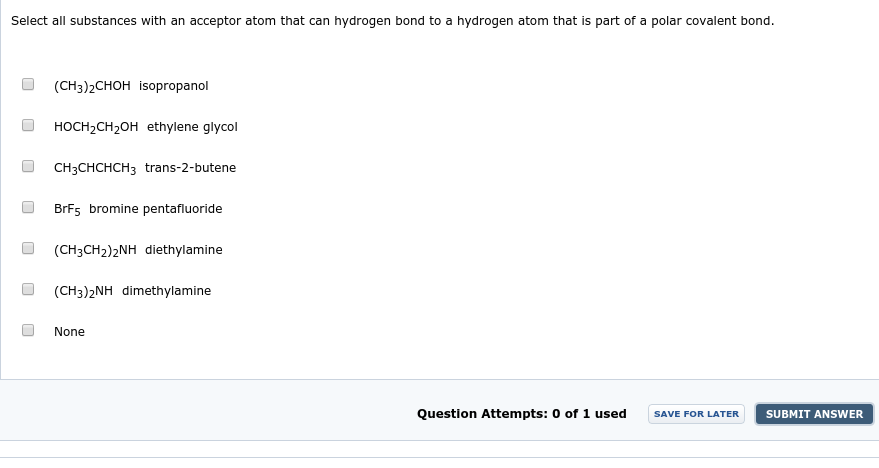
Transcribed Image Text:Select all substances with an acceptor atom that can hydrogen bond to a hydrogen atom that is part of a polar covalent bond.
(CH3)2CHOH isopropanol
HOCH,CH2OH ethylene glycol
CH3CHCHCH3 trans-2-butene
BrF5 bromine pentafluoride
(CH3CH2)2NH diethylamine
(CH3)2NH dimethylamine
None
Question Attempts: 0 of 1 used
SUBMIT ANSWER
SAVE FOR LATER
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

