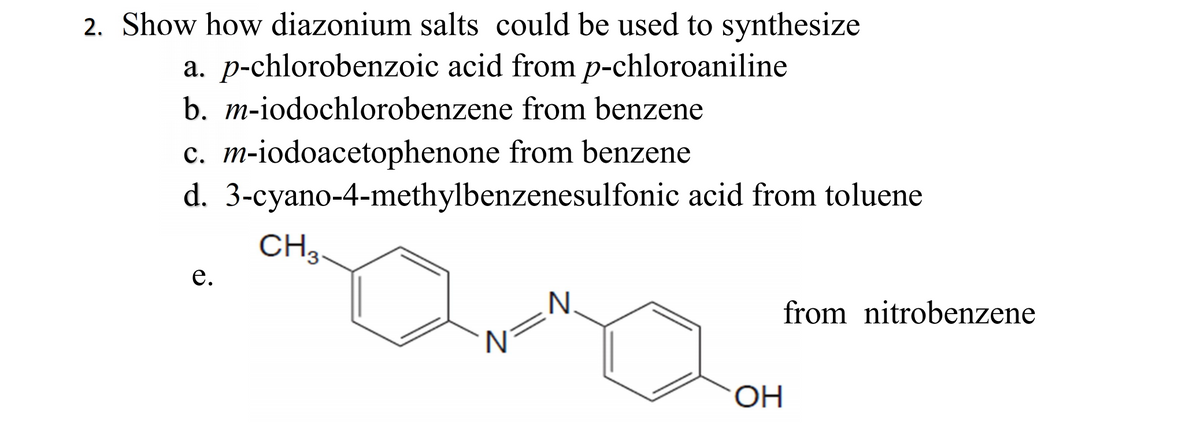
Show how diazonium salts could be used to synthesiz a. p-chlorobenzoic acid from p-chloroaniline b. m-iodochlorobenzene from benzene c. m-iodoacetophenone from benzene
Q: Give examples using arenediazonium ion salts in diazo coupling reactions and inthe synthesis of…
A: Diazonium salts are very important synthetic intermediate which can easily undergo coupling…
Q: Predict the products of the reaction of the below reagents with either acetophenone or…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: (a) Show how you prepare the following esters from the corresponding acids and alcohols using…
A:
Q: Which of the following reagents will not undergo a substitution reaction with the benzenediazonium…
A: this a substitution reaction of benzenediazonium chloride salt.
Q: Show how the synthetic scheme developed in synthesize this triiodobenzoic acid X-ray contrast agent.…
A:
Q: Which of the following compound diazonium salt will form a coloured dye on reaction with B-Naphthol…
A: Diazonium salt is formed only the reactant is primary alkyl or arylamine. If the primary amine is…
Q: An unknown compound is treated with peroxyacetic acid in dilute sulfuric acid. The product of…
A:
Q: Which of the following reagents will not undergo a substitution reaction with the benzenediazonium…
A: Those reagents which show substitution reaction with benzenediazonium chloride salt are given below…
Q: from acetone and acetoacetic ester and any other reagents NME2 from HO (b) (a) HO. from benzene and…
A: Substitution reaction is the reaction where one group is substituted with another group.…
Q: Propanol plus phosphorous tribromide will yield __________. 1-bromopropane 1,1,1-tribromopropane…
A:
Q: Give a mechanism for producing a phenol from the diazonium salt that was prepared from sulfanilic…
A: Diazonium salt It is combination of NaNO2 +Dil.HCl It is used for conversion of Amine to Diazonium…
Q: In the synthesis of 9,10-dihydroanthracene-9,10-a,ß-succinic anhydride, why was the reaction kept in…
A:
Q: reaction of 4-bromoacetanilide to 4-bromo-2-chloroacetanilide, why is the amido (rather than the…
A: Given-> 4-bromoacetanilide --> 4-bromo-2-chloroacetanilide
Q: 1-CI a) CN OH 1-CI CH3 b)
A: Applying concept of effect of functional groups on the benzene ring.
Q: Chapter 20: Functional Derivatives of Carboxylic Acids 20.54 Many types of carbonyl condensation…
A:
Q: Propose a synthesis using a reaction of a diazonium salt in at least one step of each conversion. a.…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
Q: N' 'N' CH3 CH3 II
A: I is more favorable enamine
Q: Propose a synthesis using a reaction of a diazonium salt in at least one step of each conversion а.…
A:
Q: Show the steps necessary to make 4-phenyl-3-heptene using a Wittig reaction. Start with…
A:
Q: (a) Describe the following giving chemical equations :(i) De-carboxylation reaction…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: What is the major product of nitration of p-fluoroanisole reactions?
A: Nitration of p-fluoroanisole gives the 4-fluoro-1-methoxy-2-nitrobenzene as a major product. Here…
Q: Primary amines can also be prepared by the reaction of an alkyl halide with azide ion, followed by…
A:
Q: Sodium borohydride (NABH4) is a very selective reagent. Which functional groups can sodium…
A: Sodium borohydride is powerful reducing agent than lithium aluminium hydride.
Q: Arrange the compounds in order of INCREASING reactivity towards bromination. I. Toluene,…
A: Benzene ring under go electrophilic substitution reactions Activating groups increase reactivity…
Q: The reaction for the magnesium cation with 8-hydroxyquinoline is carried out in the presence of: A)…
A:
Q: a) Br b) OH c) d) Br. Br e) NC
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: How do you prepare 4-methoxybenzoic acid from benzene
A: Solution: We want to prepare 4-methoxybenzoic acid from benzene, so this is the step up reaction. We…
Q: Propose a synthesis using a reaction of a diazonium salt in at least one step of each conversion а.…
A: Answer given as follows
Q: Propose two syntheses of 4-phenyl-2-pentanone, each involving conjugate addition of a lithium…
A: The product given is 4-phenyl-2-pentanone. Gilman reagent is an organo copper lithium reagent which…
Q: Which of the following reagents will not undergo a substitution reaction with the benzenediazonium…
A: The reactant given is benzene diazonium chloride salt.
Q: Explain the Reaction of Organometallic Reagents with Epoxides ?
A: Organometallic reagents are those reagents that have a bond between the carbon atom and the metal.…
Q: Show the steps necessary to convert benzene into p-chlorobenzonitrilie, using a diazonium salt at…
A: It is an example of electrophilic aromatic substitution
Q: Consider the Wittig reaction of p-anisaldehyde with excess benzyltriphenylphosphonium chloride in…
A: Given data, anisaldehyde Molar mass of anisaldehyde = 136.2 g/mol density of anisaldehyde = 1.12…
Q: An unknown compound is treated with peroxyacetic acid in dilute sulfuric acid. The product of…
A: The final products of reaction 4 are as follows:
Q: a. Write the structure for the β-hydroxycarbonyl compound and their dehydration product formed by…
A: The detail solution is given below
Q: 1. What is diazotization? Why are aromatic diazonium salts more stable than aliphatic diazonium…
A: Please find your solution below : 1. The reaction in which primary aromatic amine is converted into…
Q: 4- Adding HCI in the preparation of aniline from nitrobenzene. 5- When acetanilide reacts with…
A: A question based on amines that is to be accomplished.
Q: 11. The reaction for the magnesium cation with 8-hydroxyquinoline is carried out in the presence of:…
A: The reaction for the magnesium cation with 8-hydroxyquinoline is result in formation of…
Q: What product would you expect to obtain from Grignard reaction of an excess of phenylmagnesium…
A: The Grignard reagent gives a nucleophilic addition reaction with the dimethyl carbonate.
Q: Give the expected products of lithium aluminum hydride reduction of the followingcompounds (followed…
A: ANSWER: Step 1: (a). Butyronitrile on reducing with lithium aluminum hydride followed by hydrolysis…
Q: 1. How would you prepare 2-chloro-4-nitroaniline from aniline?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Use the four compounds shown below to answer the following questions: a. Why are the…
A:
Q: -NH, ) from toluene( -CH, 6. Outline a synthesis of aniline ( ) using any necessary inorganic…
A: 1. Toluene is oxidized to benzoic acid using KMnO4. Benzoic acid is converted into benzoyl chloride…
for letters A,B,C,D & E
Step by step
Solved in 4 steps with 3 images

- Hi,I need help with this chemistry question, please and thank you From the reaction between pyrrolidine and 2-methylcyclohexanone in an acid medium, enamines I and II can be formed. Describe which of the two is formed preferably and explain why?One possible side-reaction of this experiment is biphenyl, explain how it can be formed. Here is the oringial reaction bromobenzene ----->magnesium. product is benze ring with MgBr attached to ring2) Another method for the above reaction starts with 4-aminophenol hydrochloride (the conjugate acid of 4-aminophenol). The treatment of this the hydrochloride salt with sodium ethanoate (acetate) buffer produces 4-aminophenol which is then able to react with ethanoic anhydride as per our method. i. Why is 4-aminophenol hydrochloride not suitable for direct reaction with ethanoic anhydride? ii. Draw a mechanism (i.e. curly arrows) showing the deprotonation of 4-aminophenol hydrochloride by sodium ethanoate to form 4-aminophenol (the free base).
- Find the products for these two reactions. Make sure to include relevant hydrogens, deuteriums and stereohemistry.Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…please quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.
- Why cis-Ru(II)Cl2(DMSO)4 reacts with pyridine, et cetera, to give substitution of the DMSO but not the chloride ligands, but trans-Ru(II)Cl2(py)4 react with suitable Na+ and K+ salts in aqueous pyridine to afford chloride-substituted derivatives. write the reactions equations.Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Write down the mechanisms and resulting products of the reactions below.



