Q: What is the main difference between space group and point group symmetries? Which one do we prefer…
A: Symmetry group: In group theory, a Symmetry group for an object is a group of all the…
Q: Determine the point groups of the following molecule: XeF4
A: A point group express all the symmetry operations that can be execute on a molecule that result in a…
Q: XeF4 has a molecular geometry of square planar. i. How would you determine its point group? ii.…
A: XeF4 has planar geometry in which Xe present at the cut point of two diagonals of the square and 4…
Q: Please provide the POINT GROUPS of the following objects and explain why.
A:
Q: d) What is the point group of CH4? e) What is it if: i) One H is removed? ii) All four Hs are…
A: d. CH4 is an ideal tetrahedral molecule. Hence, it will have tetrahedral point group. It will…
Q: Determine the point groups for A snowflake .
A: Point groups are determined by getting symmetries by keeping one point fixed. The point group is a…
Q: Determine the point groups for Chloroethylene.
A: Chloroethylene is the ethylene molecule attack with chlorine atom that is also known as…
Q: The BF3 molecule belongs to the point group D3h. List the symmetry elements of the group and locate…
A: The group of symmetry operations of which at least one point is kept fixed is called a point group.…
Q: To what point group does CCIa belong? Using the appropriate character table, construct a reducible…
A:
Q: In a C2h point group, the character table lists the dxz and dyz orbitals in the Bg character. Do…
A: The resemblance of one part of the molecule exactly to another part of the molecule is known as a…
Q: (iii) List and depict all the symmetry elements and deduce the point group. ÇI Co.CO CO
A:
Q: Indicate the point group of the following ions. Provide molecular structures and indicate the…
A: Point group: It is used to study the symmetry properties of the molecule. There are five kind of…
Q: Which of the molecules belongs to the On point group? H. F-Xe-F H C Select one: O A O B O C O D
A: For the Oh point group of the Molecule , the Molecule must contain 6 identical element in the…
Q: List the symmetry elements and determine the point groups for each of the following molecules:
A: Point group : A Point Group reveals all the symmetry operations that can be performed on a molecule…
Q: 5. (a) Show that for a Co, point group from the following : (i) EL,? = h = 4 iit All representations…
A: The character table for C2V is given
Q: 1. Draw and write all the symmetry operations (C.. a. S.. E and i) for each molecule or object. 2.…
A:
Q: Chemistry for this in phase fully bonded phasing combination, determine: point group? # of bonding…
A: 1.Point group of IrH6 is octahedral (Oh)
Q: List all the symmetry elements of the following molecules and determine their point groups:
A: In this question, symmetry elements and point group of 1,2,3 - trichlorobenzene is to be determined…
Q: 1) For each of species, write down all symmetry operations, and the highest symmetry point group A A…
A: A symmetry operation is an activity that, once completed, leaves a molecule or item with the same…
Q: Determine the point group of the molecule shown. Your answer should have the correct formatting for…
A: The given molecule p-xylene contains following symmetry elements. Three, two fold rotational axis…
Q: b) 1,3, 5-trichloro benzene (Planar) CI CI
A: Symetry elements which are mirror image to each other. A plane which have plane of symetry that is…
Q: What is the difference between the symmetry operators and point groups of NF3 and NF2CI?
A:
Q: Provide possible molecular structures for NbF5 with C4v and D3h point groups
A: Symmetry operations C4v are E, 2 C4, C2, 2 σv and 2 σd Therefore, possible molecular structures for…
Q: What point groups result from the combination of two mirror planes oriented at 90° with respect to…
A: Point group tells about the symmetry within a molecule. The following symmetries can be present…
Q: Determine the Point group of the following molecule. Cr
A: A point group is a group of geometric symmetries that keep at least one point fixed. Point groups…
Q: Nz 0. & NO,
A:
Q: Specify the point group of each of the following species. k*** Br Br F Flil. BrCI CI 'F CI F Br CI…
A: The set of all possible symmetry operations on a molecule is called as point group. Hence, the point…
Q: (ii) List and depict all the symmetry elements and deduce the point group. NH2 N' NH2 B
A: There are several rules applied to determine the point group of a compound depending on the symmetry…
Q: The square planar molecule XeF4, has Dan symmetry. Consider the dzy and d,2_y2 0orbitals of the…
A:
Q: Determine the point group of the following molecule: Fe
A: The given compound is Ferrocene. It is a staggered compound.
Q: Show symmetry elements of trans-CHCI=CHCI and determine its point group.
A:
Q: Reduce the following representations for C2h. C2v and Cav point groups into corresponding…
A:
Q: All mirror plane of HOCI, PTCI2B12 and the other symmetry elements Or Determination of the point…
A: HOCl doesn't contain mirror plane. PtCl2Br2 contain mirror plane CCl4 point group- Td. BCl3 point…
Q: What is the point group of the molecule SOF4 (S is the central atom)?
A:
Q: 1. Work out the symmetry point group of the following (any two). (i) S,0, (ii) allene (iii) IFs
A: This Question is based upon group theory. According to question any two point group has to be given.
Q: Q3 Determine the point groups for the following molecules: Fe H. CO X- CO CO
A:
Q: I. Determine the point groups of the following: (found in NH, 1,) A. Tetrafluorocubane B D.…
A: Point group symmetry of elements. Tetra fluoro cubane is having C2 axis of symmetry. Staggered…
Q: Show symmetry elements of cis-CHCl=CHCl and determine its point group
A: We have to predict symmetry elements and point group of given compound.
Q: 2. Molecular symmetry a. Determine the symmetry operations of the molecule Ni(NH3),* and assign the…
A:
Q: The CH3Cl molecule belongs to the point group C3v. List the symmetry elements of the group and…
A: In chloromethane (CH3Cl), the central carbon is bonded to three hydrogen atoms and a chlorine atom.…
Q: O.
A: Pont group comes from the mathematical treatment to a molecule . By the point group we can explain…
Q: H. F- Xe-F H. B C D Which of the molecules belongs to the Ta point group? Select one: O A O B D.
A: Interpretation- To tell about which of the molecule belongs to the Td point group. Introduction…
Q: 2- SO 3. 2- 1)
A:
Q: H C=C H .B `H エ、 エ エーm エ エ エ
A:
Q: 2. Consider 1,3-cyclobutadiene, and answer the following: a) To what point group does it belong?
A: "Since there are multiple sub-parts in this question and it is not mentioned that which one has to…
Q: List the symmetry operations and the corresponding symmetry elements that occur in point groups.
A: Any action that ends in giving the same resemblance of the molecule as before is called as symmetry…
Q: What is the point group of CIOF4 (Cl is the central atom)?
A:
Q: Question attached
A: Point group can be described as all the symmetry operations that can be performed on the molecule…
Q: Can molecules belonging to the point groups Th or Td be chiral? Explain your answer.
A: A molecule will be symmetric the molecule is indistinguishable form the initial structure after a…
Q: What is the point group of -Hg-
A: HgI2 is linear in nature.
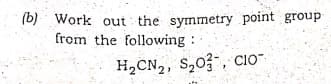
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Construct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.a In the Td point group, an S41 improper rotation is equivalent to what other improper rotation? b In the D6h point group, the symmetry operation labeled C21 is equivalent to what other symmetry operation?Determine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.
- Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.Determine the point groups of the following molecules. a Hydrogen selenide, H2Se b Partially deuterated hydrogen sulfide, or HDS c The chair conformer of cyclohexane, C6H12 d The boat conformer of cyclohexane, C6H12Why is it unnecessary to consider whether an irreducible representation from C4h is orthogonal to an irreducible representation of D6h?
- Identify the symmetry elements present in the following objects. a A ream of blank paper, no holes. b A ream of blank three-holed paper. c A round pencil, unsharpened, with cylindrical eraser. d A round pencil, sharpened, with cylindrical eraser.Linearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of methane has A1g symmetry and x- polarized light has the label T2, what are the symmetry labels of allowed excited electronic states? Use the Td character table in Appendix 3.Determine the point group of the following molecules. a cis1,2 Dichloroethylene b trans1,2 Dichloroethylene c Toluene, C6H5CH3 d 1,3-Cyclohexadiene.
- Show that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e TdWhat are the number of classes and the order of the following point groups? a C2v b D2h c D6h d S4 e Cs.In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.

