Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: When determining the equilibrium concentrations for the reaction [A][B] AB(g) = A(g)+B(g) K. = [AB] ...
A: Consider the given information is as follows; Kc=x2AB - x .......(1...
Q: Is the use of an indicator to determine a concentration using titration as good as using a pH meter?...
A: The explanation of the following is:
Q: 7.) Given: Solubility product for a series of hydroxides: BiOOH Ksp = 4.0 x 10-10 = [BiO+][OH-] Be(O...
A:
Q: Aqueous hydrochloric acid (HC1) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A: Given data : Mass of HCl = 20.0 g Molar mass of HCl = 36.46 g/mol Mass of NaOH = 12.3 g Molar mass o...
Q: A solution that has a hydrogen ion concentration equal to 3.52 x 10-3 has a pH = _____________?
A:
Q: Vhat is the pOH of a 0.20 M NH4CI solution ? Kp for NH3 is 1.8 x 10-5. O11.28 O 2.72 9.02 4.98
A:
Q: The diagram represents the equilibrium state for the reaction X,(g) + 2 Y(g) = 2XY(g) Assuming the v...
A:
Q: The following reaction is first order in N2O5N2O5: N2O5(g)→NO3(g)+NO2(g)N2O5(g)→NO3(g)+NO2(g) The ...
A:
Q: OH H30* (d) CH3-CH-CH2- ċ-CH2-CH3 A 9. OH H30* (е) CH3-C-CH2-C-CH3 ČH3 OCH3 (f) CH30-C-CH2-C-OCH3 + ...
A: In presence of H+ and heat , alcohol compound undergoes E1 elimination through carbocation formatio...
Q: Consider the reversible reaction. PCI, = PCI, + Cl, What substances are present at equilibrium? PCI,...
A: A state of rest or balance due to equal action of opposing forces. Chemical equilibrium, conditions ...
Q: 3. When 50.0 grams of magnesium react with silver nitrate solution according to the following equati...
A:
Q: How many electrons, protons, and neutrons are there in the ion of the hypothetical 445A-3+ element 1...
A:
Q: Choose the best buffer system to maintain the pH at 7.00. O Acid Z and NaZ, K, = 8.0 x 10-9 O None o...
A:
Q: When 2,400 mg zinc is added to hydrochloric acid, 450 mL of hydrogen gas forms at a temperature of 3...
A: To solve this problem we will use the given balanced chemical equation and ideal gas equation .
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction: Hg2+(aq) +...
A: From Nernst equation Ecell = Eocell - 0.0591/n (logQ) at equilibrium Ecell = 0 Q = Kc Q = react...
Q: Name the highlighted chemical group in each molecule. Lewis structure name of highlighted group H H ...
A: Functional group: Functional group is a group atoms which can change the chemical and physical prope...
Q: 4. Given the reaction: PCls(g) = PC13(g) + Cl2(g) (AH° = +87.8 kJ/mol). Which of the following chang...
A: The correct option are as follows:
Q: An unknown hydrocarbon contains carbon, hydrogen, and oxygen. Combustion of 1.5000g of the hydrocarb...
A:
Q: Consider the following equation 2H2 (g) + 2NO (g) → 2H2O (g) + N2 (g) which has the rate law Rate =...
A:
Q: The following reaction is carried out with [PCl5]i = 0.400 M at 25°C (i means initial) The reaction...
A:
Q: Complete this reaction Mn, O, Mn; + A providing A, B and C
A:
Q: This reaction generates 7.70 L of oxygen at a temperature of 18.7°C and a pressure of 0.92 atm, what...
A: Here the decomposition reaction of KClO3 solid gives KCl solid and O2 gas. The volume of O2 produced...
Q: 6. Propose a route to Compound 2 from only bromobenzene and methyl formate as carbon sources, plus a...
A: Let us talk about the grignard reagent. Here firstly the bromobenzene is used. The we have to reac...
Q: ОН CH,=CH–Ĉ–CH3 CH3-CH2-CH2-C-H A + CH,CH3
A: It is Robinson annulation reaction.
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic i...
A: first we have to determine given molecule is diatomic or polyatomic Then we have to find given mole...
Q: 2 NO + 2 H2 → N2 + 2 H2O Reaction Rate [NO] (М) [H2] (М) Experiment 1 0.420 0.122 0.136 2 0.210 0.12...
A: Initially we find order with respect to both NO and H2 Then find value of rate constant Finally we...
Q: What is the pH of a solution prepared by mixing 15.0 mL of 0.24 M KOH and 25.0 mL of 0.24 M of hypoc...
A: Given data : Concentration of KOH = 0.24 M Volume of KOH = 15.0 mL Concentration of HClO = 0.24 M Vo...
Q: pads/CHM%20111%20Empirical%20Formula%20.pdf D|A Mass of crucible and lid (g) 30 673 g Mass of crucib...
A:
Q: luz luz (E) (E) (2E,4Z,6Z,8E)-deca-2,4,6,8-tetraen
A: A pericyclic reaction is a concerted reaction in which the number of rings in the transition state i...
Q: 2. A solution may contain any of the three group I cations. A white precipitate is formed the additi...
A: The most important cations in 1st group are Ag+, Hg2+2, and Pb2+. The solution may contain any of th...
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 6....
A: As given that heat flow out of the water that means heat is lost by it and it's temperature would ...
Q: What is the calculated value of the cell potential at 298K for an electrochemical cell with the foll...
A:
Q: Find the H3O*| in a Cabernet Sauvignon with a pH of 3.66. Πν ΑΣφ ? [H3 O]+ = M Submit Request Answer
A: pH of solution is defined as negative logarithm of hydrogen ions.
Q: A sample of pure t-butyl alcohol weighing 7.512 g was found to have a freezing point of 24.7 °C. Whe...
A:
Q: Arrange the following aqueous solutions, all at 25°C, in order of decreasing acidity. Rank from most...
A: Acidity order, Most acidic. Most basic 0.0023M HCl> Ph--5.45 = p...
Q: the moles of gl
A:
Q: Acetic acid is a weak acid, meaning it does not fully dissociate in water. Instead, there is an equi...
A: The solution is as follows:
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric coeffici...
A: Given, Chemical equation : H2(g) + P4(s) ---------> PH3(g)
Q: Photosynthesis in plants converts carbon dioxide and water into glucose (C,H1206) and oxygen accordi...
A:
Q: Question 7 Consider a spin-1/2 solid where the two energy levels are split by 25 GHz. (a) At a tempe...
A: Given:-
Q: Determine the percent ionization of a 0.230 M solution of benzoic acid. Express your answer using tw...
A: Given Data : Initial concentration of acid = 0.230 M We know , ka for benzoic acid is 6.5 × 10-5 ...
Q: Industrial production of hydrogen gas uses the reaction shown below. If 1.00 metric ton of propane r...
A: Theoretical yield :- The maximum amount of the product formed when reactants are consumed as per ba...
Q: Rewrite this measurement with a simpler unit, if possible. kg m 4.7 2 2 m m Note: If you can simplif...
A: • The given measurement is- 4.7 kg•m m2• m2 • We n...
Q: Balance the following chemical equation for the reaction of hypothetical elements X, Y, and Z. On yo...
A: ____X3Y6Z2 + ___Z2 → ______Z2 + ___XY2
Q: Which of the following is a BrØnsted-Lowry acid? O C Cl 4 Ο ΝΗ O PH, Ο ΝΗ5 O Br 2
A:
Q: What is the name of the sugar shown here? OH OH O L-xylose O D-xylose O D-erythrose O L-erythrose O ...
A: To identify the name for given Sugar.
Q: m Calculate the kinetic energy of a 34. g bullet moving at a speed of 458. Round your answer to 2 si...
A: Given, mass of a bullet = 34. g = 0.034 kg Speed, v = 458. m.s-1
Q: Ammonium perchlorate (NH,CIO,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts wi...
A: given :- ammonium perchlorate → nitrogen gas + oxygen gas + chlorine gas + water mass of ammonium ...
Q: Consider the following reaction: 4 Zn (s) + 10 H*(aq) + 2 NO3 (ag) 2 4 Zn2+ (aq) + N20 + 5 H2O (1) T...
A: Equilibrium constant is the ratio of product of concentration of products raised to their stoichiom...
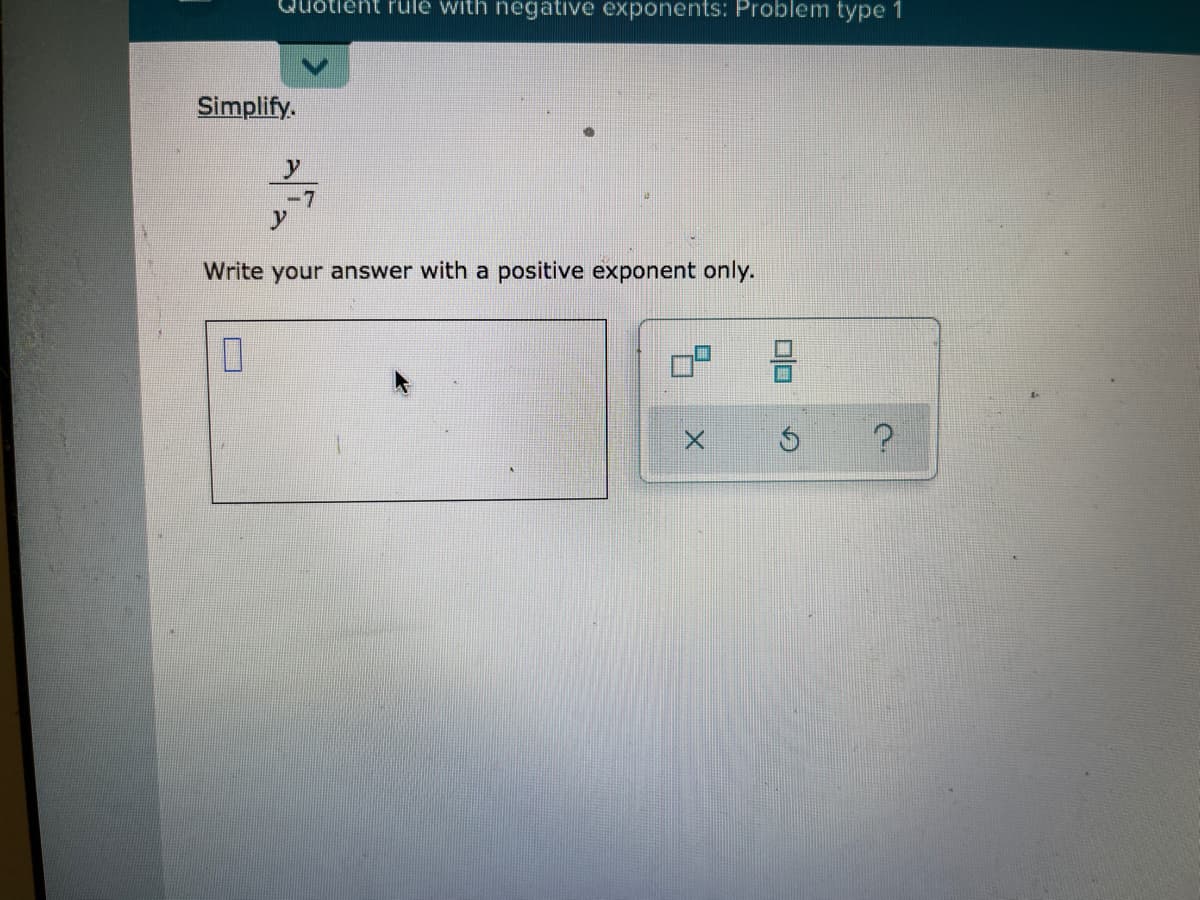
Step by step
Solved in 2 steps

- This question was rejected, but this question is part of a practice set and as you can see it is out of zero points. Other practice questions from this set were answered.Find the density for each trialPlease answer ASAP, thanks! Note: Express all answers to the nearest fourth decimalplace, with correct units.

