Q: Block # 4 Mass 41 451g Length 3 32 Find density and percent error of the solid. Show work and circle…
A: Here we are making multiplication and division to solve for the answer. According to the rule of…
Q: Can someone help me write out this conversion in the same way it is written out in the answer…
A: At STP 1MOLE OF GAS OCCUPIES 22.4 L N 2+3H 2 -----> 2NH3
Q: Keat for the Control in experiment
A: (d) Kcat = Vmax/[E] Given, [E] = 0.001 micro molar i.e. 0.001 micromol/L
Q: Argb 46H of A = -300 kJ/mal of B=-350 kt of =+100 kT ol of D = -300 kilapl %3D
A: The chemical reaction : A(g) + 2B(L) ---> 2C(L) + H2SO4(L) Change in enthalpy of formation…
Q: 100 80 - 40- 20- ot 10 20 30 40 50 60 70 80 m/z Relative Intensity
A:
Q: Please answer in 4 decimal placess. Thank you!
A: Given :- Initial moles of gas is 70 mmol = 0.07 mol Temperature is 373K Volume changes from: V1 =…
Q: Using the Q test, decide whether the value 222 should be rejected from the set of results: 193, 222,…
A: Dixon's Q test or Q test, is used for identification and rejection of outliers from the given…
Q: Why is a pipette more accurate than a burette?
A: Burette is a long glass cylinder with a stopcock at the bottom to release the liquid. Pipette has a…
Q: Read the meniscus with the uncertain error range (+/-). ml
A: According to the given image, the meniscus lies in between 5 mL - 6 mL.
Q: Is this correct or does the true value need the same sig figs as the estimated value?
A: Number of significant figures gives an idea on the accuracy of the measured value.
Q: Read the length of the metal bar with the correct uncertainty error range (+/-). 5 10 15 20 cm
A: Length is measurement of anything from end to end.
Q: There are THREE parts. Please circle the final answer!
A: The SN2 reaction is a nucleophilic substitution reaction where a bond is broken and another is…
Q: The density answers are so close that I need help trying g to figure which substance goes with what
A: Substance 8 d= m/v = 8.5/15.6 = 0.55 g/ cm3 substance 9 d= m/v = 8.0/15.6 = 0.51…
Q: indicate which pattern of arrow pushing it represents 0: N.
A:
Q: Table I: Calibration of dropper: UMCO2/drop Trial # of drops Average ml/drop (ml/drop x 10UMCO/ml)…
A: How to find the average no of drops? Average no of drops = Sum of all no of drops in each…
Q: 57.A common sampling plan that uses master tables to interpet the results a. 100% inspection C.…
A: 57 and 59 are same questions and thus have been solved only once.
Q: the VT of a column is 30 cm3 , and VS is 20 cm3 , and Vo is 7 mL, find Vi (in mL).
A:
Q: nteger value between one and infinity
A: Answer : a) the principal quantum number- 1)an integer value between one and infinite. b)the angular…
Q: not
A: We will. Calculate concentration of OH-
Q: Calculate the percent relative error
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: numbering provided on the ring. Answer Bank H Br H CI H. H LO
A:
Q: 4 Br 1. 4 signals 2. 2 signals NH2 3. 5 signals
A: Given : We have to tell number of signal in C-13 NMR spectrum.
Q: a) By how much should the student expect their sample measurements to increase by? Why? b) Will…
A: (a) In spectrophotometry, the absorbance is directly proportional to the concentration of analyte…
Q: Convert 16 nm to μm.
A: • We need to convert 16 nm to ____ μm
Q: 4 darts strike near the center of the board. Whoever threw the darts is: * Precise Neither accurate…
A: 1. Precision refers to how close your measurements are to one another. Accuracy refers to how close…
Q: Only 98 a and b and c please
A: 98. a) The balanced equation for the reaction is : 2Cl2O → 2Cl2 + O2 As we can see both O2 and…
Q: Convert the mS/cm solution as a µS/cm solution. 27.17 mS/cm 70.28 mS/cm 25.50 mS/cm 36.39…
A:
Q: Simplify. y y Write your answer with a positive exponent only.
A: The given Fraction of Polynomial: yy-7
Q: "Precision" is the ___________ of a measurement a. probability b. repeatability c. variability…
A: Precision:The closeness of two or more calculated measurements regardless of the accuracy of those…
Q: Level = 6ft of water (h20 density = 0.036 Ib/in^3) Find gauge reading in psi = Find gauge reading…
A: As it is known that- 2.31 feet of water head will be equal to 1 Pound per square inch (psi) psig:…
Q: The calculated Ccal will _________ if you insert into the test tube the thermometer and stopper…
A: Answer: No effect or No change.
Q: Simplify. <-59 X. X Write your answer with a positive exponent only.
A:
Q: Correct the one that marked wrong
A:
Q: Rxn: A + B -> C + D [A] M [B] M Rate M/s TRIAL 1 0.21 0.70 6.3 x 10^-3 TRIAL 2 0.63 0.65 1.89 x…
A: The rate of reaction equation for the given equation is:…
Q: (d) HO, (3 methods) HO, ( 2 methods) oto OH (f) (3 methods)
A:
Q: In a particular trial, a student got a value for R of 0.085. Calculate the percent deviation of this…
A: Percent deviation is given by: % deviation=Rexperimental-RacceptedRaccepted×100 ......…
Q: 1) Comment on the Pearson’s R of the generated line equation, before you compute for the unknown…
A: We are given the concentration and absorbance data for the standard reference solution. Also the…
Q: In a particular trial, a student got a value for R of 0.071. Calculate the percent deviation of this…
A: In the particular trail the student got the value of R=0.071. The experimental Value of R = 0.071…
Q: standard deviation for each treatment.
A: To determine the mean and standard deviation for each treatment. The mean is the sum of all the…
Q: please find the percent error not for each one but for the three trials together:)
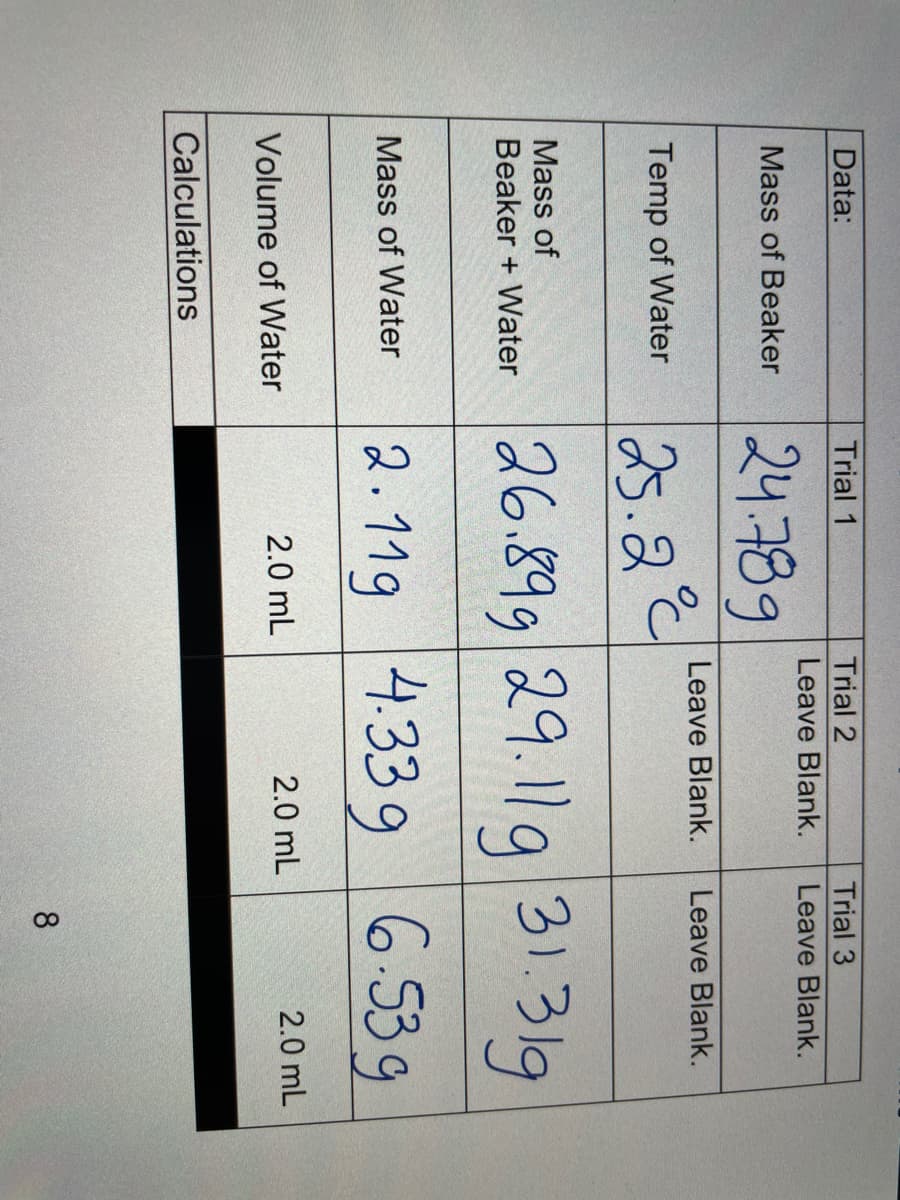
A: In the given question we have to calculate the percent error for the density of water. we know…
Q: The end point is more accurate than the equivalent point False True
A:
Q: Sample A: Sample B: Sample C: Sample D: Sample E: Sample F: 22mm 18mm 35mm 30mm 28mm 28mm 21mm 17mm…
A: Since the units are in mm, this means we are taking about some distance, length or height.
Q: How do I calculate difference Ha? First page is my data. Part A is for the question
A: Run-1 (part-A) q = m c∆T q = heat of reaction m = mass Volume of water = 100 mL and density of…
Q: MULTIPLE CHOICE NUMBERS 1-5
A: Dear student the answer for five multiple choice question is given below., The Brønsted-Lowry…
Q: C. [H+] = 12 M * Your answer d. [H+] = 5.4 x 10^-5 M* Your answer
A:
Q: need help wilth calculating percent relative error. What will be the real value when it comes to…
A: Percentage error is the difference between the estimated number and the actual number when compared…
Q: Find the density of liquid ? if : the depth of immersion is 3 cm, the load weight is 5 gram, and the…
A:
Q: "NH₂ over 3000 cm -1 Peak 2- and 3 d 2
A: According to the question, we need to choose the correct option.
Q: Density of solution: Trial 1: 1.2 g/mL Trial 2: 1.2 g/mL Trial 3: 1.2 g/mL Average density = 1.2…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- 6. A nitrogen gas (MW = 28) occupies a volume of 5ft3 at 2550 lb/ft2 abs and 755oR. What are its specific volume and specific weight? (3 DECIMAL IN FINAL ANSWER PLS!!)that answer of -243°C marked as a wrong answer. Do we calculate Sig Figs in that ? (1.23 atm)(16.5 L)/ (8.3 moles)( 0.0821 atmxL/molxK) = 29.77295643 K do we round up to 29.8K 3 Sig Figs? or 30K to 2 Sig Figs? and if 30K-273 K = -243.0°C or 29.8K-273K = -243.2°C ??Calcium Chloride Ammonium Chloride Trial 1 Trial 2 Trial 3 Trial 1 Trial 2 Trial 3 Mass of water 100g 100g 100g 100g 100g 100g Mass of salt 5.36g 10.28g 15.10g 5.29 10.43 15.31 Moles of salt Initial T 21.0 21.0 21.0 20.0 20.0 20.0 Final T 29.6 36.2 43.5 17.1 13.2 10.1 ΔT 8.6 15.2 22.5 -2.9 -6.8 -9.9 ΔH(Joules) ΔH(kj/mol)
- A pipet delivers 10.9 mLmL , 10.1 mLmL , and 10.9 mLmL in consecutive trials. Find the mean volume of the samples.Please answer fast it’s very important and urgent I say very urgent so please answer super super fast please For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.4661) Mass of flask, aluminum foil, and rubber band 68.45g 2) Temp. Of boiling, water 98.20 0C 3) Barometric pressure 755 mmHg 4) Volume of flask (volume of vapor occupies flask) 152 ml 5) Mass of flask, aluminum foil, rubber band, and condensed vapor 68.60g 6) Mass of condensed vapor is (5)-(1) Find Molecular weight of unknown ______ g / mol Calculation PV = (m/M)RT) è M = (mRT) / PV = ? Unknown liquid- Based on Molecular weight determine which unknown below has this molecular weight: Methanol, Ethanol, Isopropanol, Propanol. UNKNOW IS : __________________________
- brian wants to hit the beach and go diving. However if his body doesn't take up enough space he could sink and drown. He tries to measure himself with a "ruler" but the life guard sais that's not good enough.A. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).desired Mg mass =0.0340g "Atmospheric pressure (mmHg): 733.59Trial 1 and Trial 2 values separated by commas:Mass of Mg (g): 0.038, 0.031Temperature (°C): 20.2 (both trials)Vapor pressure of water (mmHg): 17.86 (both trials)Volume of gas (mL): 42.95, 35.80 need calculations for table.
- You are absolutely the BEST. Finally someone that dosen't calculate this exercise as 836f66dgsffjgvkik!!!! I understand everything you did, but I have one question. Some experts says that km can be calculated by saying Vmax from date divided by 2 and then they find Km by going down on x-axis on the graph. But, you used the MM equation, to solve for Km. Is both method the same? Or is one of them more precise than the other? *And if theres no need to reply back after you answering my question, then I will thank you again for your help. I have more these types of exam questions that I will probably upload later, and I hope someone like you will look at it.10. You have 24 US gallons of water in a basin and you decided to put it in the 220ml bottles. How many bottle you will need? Hint: 1 US gallon = 3.7854liters Kindly include the figure, analysis, and findingsA student was given an unknown hydrate, which could be one of threepossibilities: magnesium sulfate heptahydrate, sodium dichromate tetrahydrate orbarium chloride dihydrate. Using the following data identify the unknown.Wt. of crucible/lid empty 39.46 gWt. of crucible/lid/hydrate sample 46.84 gWt. of crucible/lid/sample after heating 45.77 g



