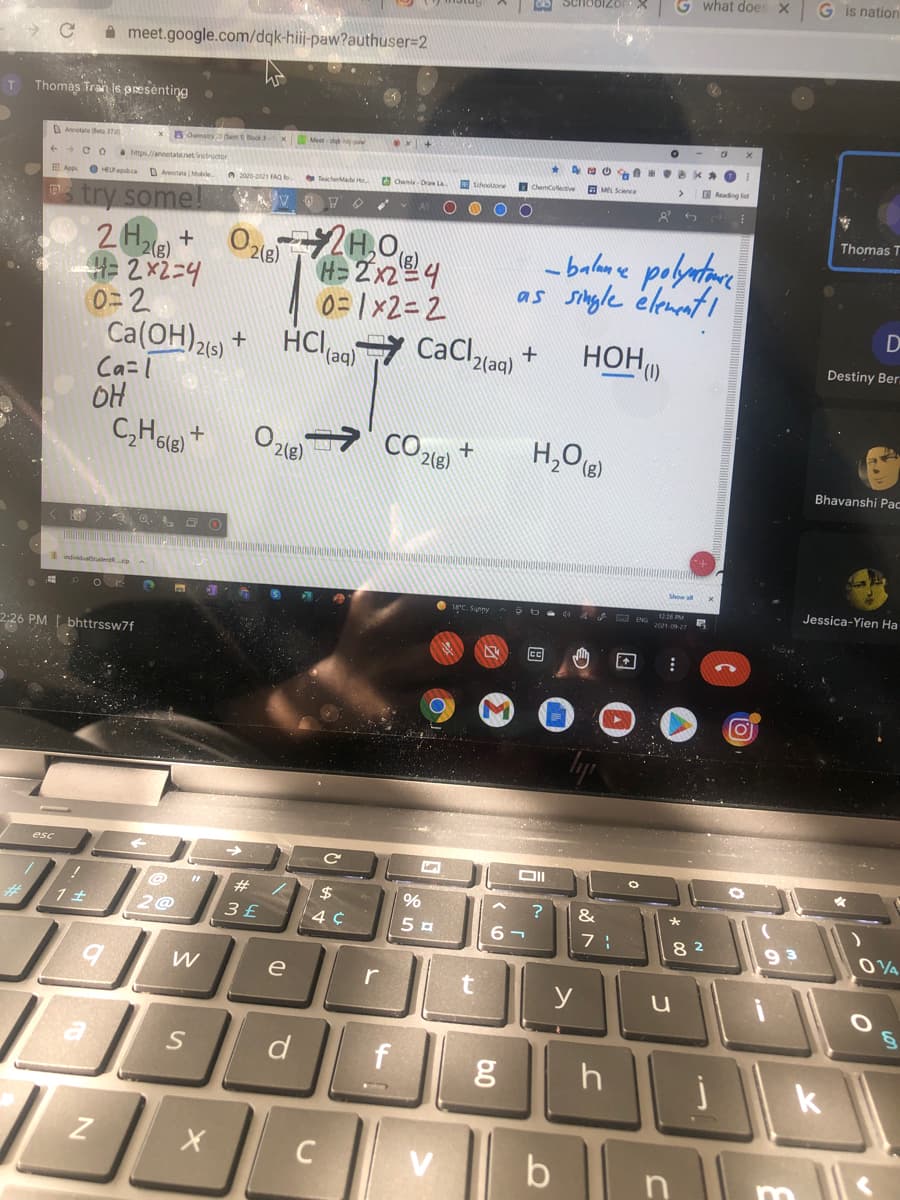
Smas Iran ls presénting OAnotate e 7 stry emt He Meet ++CO tpannetatnetruor Tcheade a Chem Or Schoole I ChenColletive a MEL Sne ading lit Ap O HE Ame Mbile s try some H= 2x24 0= 1x2= 2 - balane polatare as single élemnti HOH 0=2 Ca(OH)216) + Ca=l OH C,Hole) + HCI (aq) Cacl2(aq) 7 CaCl, CO 218) 2(8) ndividiade Show all 1224 PM ANG hhttrpeu
Smas Iran ls presénting OAnotate e 7 stry emt He Meet ++CO tpannetatnetruor Tcheade a Chem Or Schoole I ChenColletive a MEL Sne ading lit Ap O HE Ame Mbile s try some H= 2x24 0= 1x2= 2 - balane polatare as single élemnti HOH 0=2 Ca(OH)216) + Ca=l OH C,Hole) + HCI (aq) Cacl2(aq) 7 CaCl, CO 218) 2(8) ndividiade Show all 1224 PM ANG hhttrpeu
Introductory Chemistry: A Foundation
8th Edition
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter1: Chemistry: An Introduction
Section: Chapter Questions
Problem 11ALQ
Related questions
Question
Transcribed Image Text:G what does X
G is nation
A meet.google.com/dqk-hii-paw?authuser=2
Thomas Tran is psesenting
T
OAnotate eta 370
C O
a https//annotatanet instructor
a Chemis Dran La
O scholae
ChemCollective
O Reading list
I App
O HELPapb.ca Annetate(Mobile
s try some!
O O
Thomas T
2H,0
- balan e polatare
as single élement
+
H2x2-4
0=2
Ca(OH)216) +
Ca=T
OH
0=1x2=2
D
HClag)7 CaCllao) +
HOH)
2(aq)
Destiny Ber.
CO.
2(g)
H,O
+
Bhavanshi Pac
a o. Q O
Jessica-Yien Ha
2:26 PM | bhttrssw7f
cc
esc
#3
$4
2@
3 £
6 -
8 2
9 3
e
y
d
h
b |
C
V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning