Q: 1. Mass of crucible lid 2. Mass of crucible + lid + sample before heating 3. Mass of crucible + lid+…
A:
Q: Write both the charge balance equation (CBE) and mass balance equation (MBE) for the following…
A: Mass balanced equation consist of the equilibrium concentration of species present in the solution…
Q: - Volume to molecules. A human red blood cell has an average volume of about 90 femtoliters.…
A: i ) Here volume of cell is given in femtoliters, so first we need to convert volume to mL (…
Q: Calculate the average translational kinetic energy, Ex, for one mole of gas at 669 K. Translational…
A: Answer: In both the questions we have to calculate the average kinetic energy of gas particles at…
Q: How many dots are shown in a dot diagram for the noble gases (except He)? A: 2 B: 8 C: 10 D: 18…
A: Noble gas have 8 valence electrons in its valence shell except He which have only two valence…
Q: Organic product of the E llone pairs and charges counteric
A: Organic reactions are those in which organic reactant react to form organic product. In the given…
Q: Calcium hydride (CaH 2) reacts with water to form hydrogen gas: CaH 2 (s) + 2H 20 (1)→ Ca(OH) 2 (aq)…
A: Given :
Q: Driving 1000 miles a month is not unusual for a short-distance commuter. If your vehicle gets 23.30…
A: Balanced chemical equation: 2C8 H18 + 25O2 -----> 16CO2 + 18H2O 1 Gallon =…
Q: Then, use a dotted line (or a second color) to draw a second wave that has the same velocity but…
A: Wave possesses three characteristics wavelength, amplitude and frequency so, Number of oscillation…
Q: A compound is 40.0% C, 6.70% H, and 53.3% O by mass. Assume that we have a 100.-g sample of this…
A:
Q: If the ?a of a monoprotic weak acid is 7.8×10^−6, what is the pH of a 0.12 M solution of this acid
A:
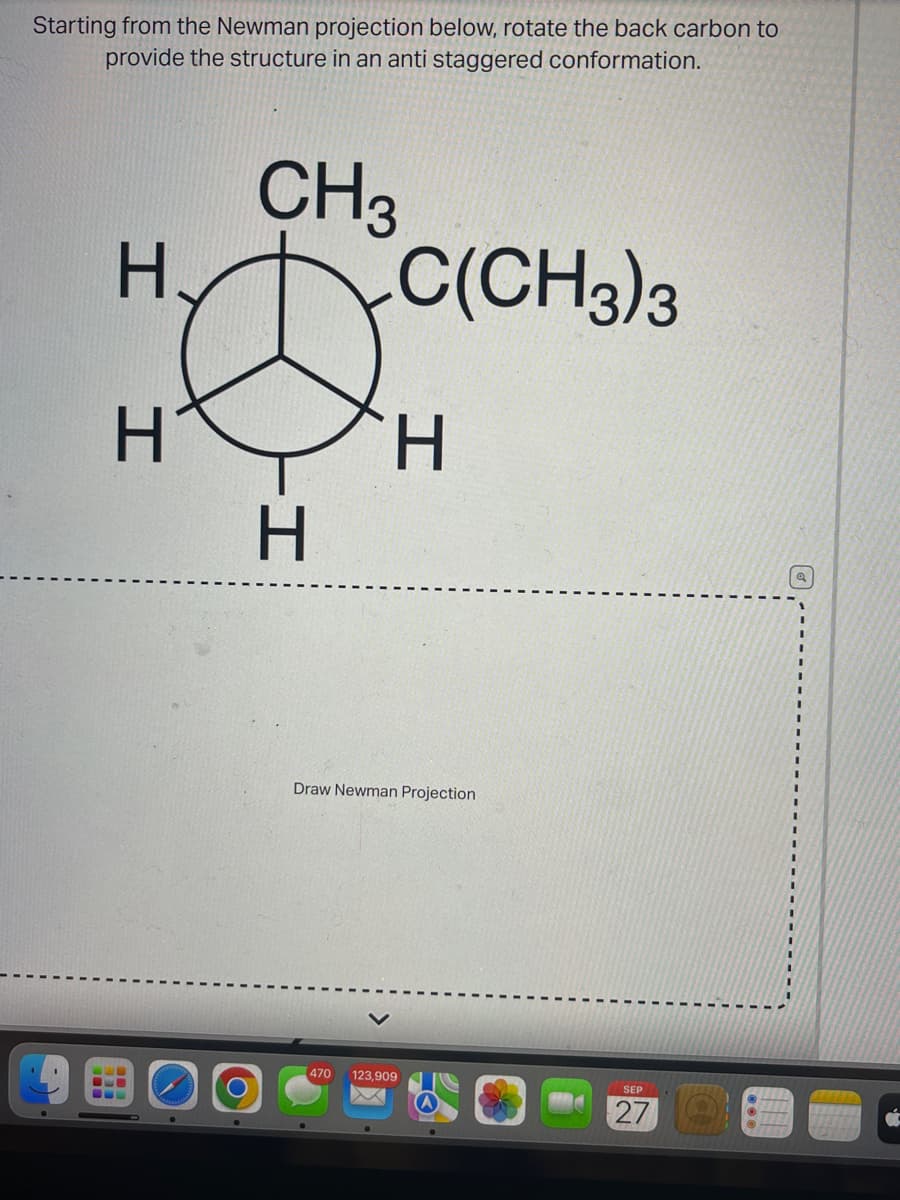
Q: Hi, could someone show me how to draw the Newman Projection for C2-C3 bond of 2,2-dimethylbutane and…
A: The Newman projection represents the dihedral angles of different conformations of a molecule. In…
Q: Propose syntheses for the following compounds. Use benzene or toluene as your starting aterial. Br…
A:
Q: Which of the statements are evidence that gases do not always behave ideally? CO₂ gas becomes dry…
A: Here we have to identify the statements which are not predict the ideal behaviour of gas.
Q: Provide the least stable (highest energy) chair conformation Relative cis/trans stereochemistry…
A: Given -> Compound-> trans-1,2-dimetylcyclohexane -> trans (1,2) => ( a,a) , (e,e)
Q: n H₂C: isobutylene CH3 CH₂ CH3 A CH₁₂ n poly(isobutylene)
A:
Q: Calculations: Calculate the concentrations of dichromate and permanganate in the unknown solution?
A: Absorbance is the amount of light absorbed by a solution at a given wavelength. According to…
Q: A sample of Br₂(g) takes 40.0 min to effuse through a membrane. How long would it take the same…
A:
Q: Cr(H2O)5Br]2+
A: Coordination chemistry is branch of chemistry in which we deal with coordination compound.…
Q: 2.The mass of 15.0 mL of a particular liquid was measured to be T1=7.47g,T2=7.45g and T3=7.53g.Find…
A: Given data, Volume (V) = 15.0mL True value = 7.5 g/mL Masses of particular liquid; T1 = 7.47 g , T2…
Q: 1) Which of the following molecules DOES NOT have a stereogenic center in the S configuration: CO₂H…
A: stereogenic center :- The carbon center of any compound in which it is connected with four…
Q: 51. Which of the following conjugate acid/conjugate base pair serves as a buffering system in higher…
A: In the body of animals, chemical buffer system consists of three individual buffers: the carbonic…
Q: 2. Draw the mechanism for the following SN2 reaction. Draw the reaction coordinate diagram labeling…
A:
Q: Find the empirical formula for myristic acid. Express your answer as a chemical formula. Express…
A:
Q: How many molecules (not moles) of NH3NH3 are produced from 7.93×10−4 gg of H2H2?
A:
Q: 5. What amounts of reagents are needed to make the following buffer solutions? amchioride. Z B)…
A: The components of the buffer solution are weak acid, HCN(aq), and conjugate base, NaCN or CN-(aq).…
Q: 3. A 0.3535 g sample of KHP requires 19.27 mL of NaOH to reach the end point. What is the…
A:
Q: Why is the ration of strengths of acids and bases significant in titration?
A: Please find your solution below : Titration is a method of quantitative chemical analysis which is…
Q: [References] a. Account for this fact: Although methanol (CH3OH) (bp, 64.7 °C) has a higher molar…
A: To answer: a) Although methanol (CH3OH) (bp, 64.7 °C) has a higher molar mass than water (bp, 100…
Q: Compare the boiling points of the various isomeric hydrocarbons shown in the table below. Notice the…
A: The main factor here is Vander Waals Force i.e. playing the main role for the deviation of the…
Q: X Exercise Q1. Name the following compounds (a-d): S N da p N=N (b) (a) (c) N (d)
A:
Q: Consider the reaction described by the equation C₂H₂Br₂(aq) +31-(aq) - - The rate law is rate =…
A:
Q: Which of the gases has the fastest-moving molecules, on average, at a given temperature? O NO₂ OCH…
A: We have asked two questions. We have to tell which gases have fastest moving molecules in one…
Q: hydrogen by mass. Assume a 100.-g sample of this compound. How many grams of each element are in…
A: Just using the stoichiometry Here
Q: Complex or Compound Name Metal Metal d Charge electrons Molecular Geometry Obeys 18- electron rule?
A: Sn(acac)3]NO3
Q: a Vapor pressure curves for CS₂2 (carbon disulfide) and CH3 NO₂ (nitromethane) are drawn below. What…
A: The graph shows relation between the vapor pressure versus temperature in °C.
Q: 2. You are hanging out with your friend Angel, and you decided to warm up some snacks in the…
A:
Q: (a) What types of intermolecular forces do morphine and heroin each possess? (b) Which compound can…
A:
Q: Butane (C4H10) is the liquid fuel in lighters. How many grams of carbon are present within a lighter…
A: Given data : Volume of butane = 7.25 mL Density of butane = 0.601 g/mL Molar mass of carbon = 12.01…
Q: 1) Rank the following titrations in order of increasing pH at the halfway point to equivalence (1 =…
A: Note: As per the guidelines, solution of first question has been made. For the expert solution of…
Q: Indicate the valence orbitals and valence electrons for the following atoms: a. Lead b. Astatine c.…
A:
Q: Use the combined gas law to solve the following problems: 8) If I initially have a gas at a pressure…
A: According to Bartlby guidelines , I solve only first question . You post other questions separately.
Q: A flexible container at an initial volume of 4.11 L contains 7.51 mol of gas. More gas is then added…
A:
Q: Check TLC (thin-layer chromatography) or CC (column chromatography) as the more appropriate answer…
A: Column chromatograpy is an technique to separate bulk compounds and needs more time for separation,…
Q: IR Spectrum Transmittance 70- --09 60- 50- 40- 30- 20- 10- 3500 2993 2948 3000 -2831 2500 Wavenumber…
A: Groups that are accompanied by change in dipole moment shows IR spectra at specific position…
Q: 2. In a titration, 50.00 mL of 0.875 M HCl solution was titrated with 0.506 M NaOH solution. What…
A: Sodium hydroxide reacts with hydrochloric acid, to form sodium chloride and water. The equation for…
Q: Part B Determine the theoretical yield of aspirin for the reaction. Express your answer in grams to…
A:
Q: the chemical formula for sodium thiosulfate pentahydrate.
A:
Q: Ammonia, NH3, may react with oxygen to form nitrogen gas and water. 4 NH3(aq) + 30₂(g) →→ 2N₂(g) +…
A: Thereotical yield - The maximum amount of product formed at given condition from given amount of…
Q: A chemical engineer must report the average volume of a certain pollutant produced by the plants…
A: Given -> Volume of South fork(V1) = 0.7 L Volume of Oglala(V2) = 1.71 L Volume of cross creek…
Step by step
Solved in 3 steps with 1 images

- Fill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.myo-Inositol, one of the isomers of 1,2,3,4,5,6-hexahydroxycyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation of myo-inositol.
- For pentane draw Newman projections for the Syn-periplanar, conformation. the Anti- periplanar conformation and a Gauche conformation. Use C2 as the front carbon and C3 as the back carbon. Label each conformation, circle the highest energy conformation andunderline the lowest energy conformation.a) Sighting down the C3-C4 bond, draw the gauche (60 degrees) and anti (180 degrees) Newman projections of 2,4-dimethylhexane. b) Circle the conformation that you drew that is lower energy.Sight along the C2-C3 bond of 2, 3-dimethylbutane, and draw a Newman projection of the most stable conformation.
- Draw a Newman projection of the anti conformation of 1,2-dichloroethane.Draw the most and the least stable conformations of 2,3,4,5-tetramethylhexane as Newman projections and name the conformations.what are the sawhorse projection and Newman projection of ANTI-CONFORMATION of n-pentane rotating between c2-c3?
- Draw the Newman projections for one staggered and one eclipsed conformation of 2,2-dimethylheptane, viewed down the bond from C3 to C4.Create a 3D model of the most stable chair conformation of (1R,3S)-3-chlorocyclohexan-1-olwhat is the sawhorse projection of the LEAST STABLE ECLIPSED CONFORMATION OF N-PENTANE ROTATING BETWEEN C2-C3?



