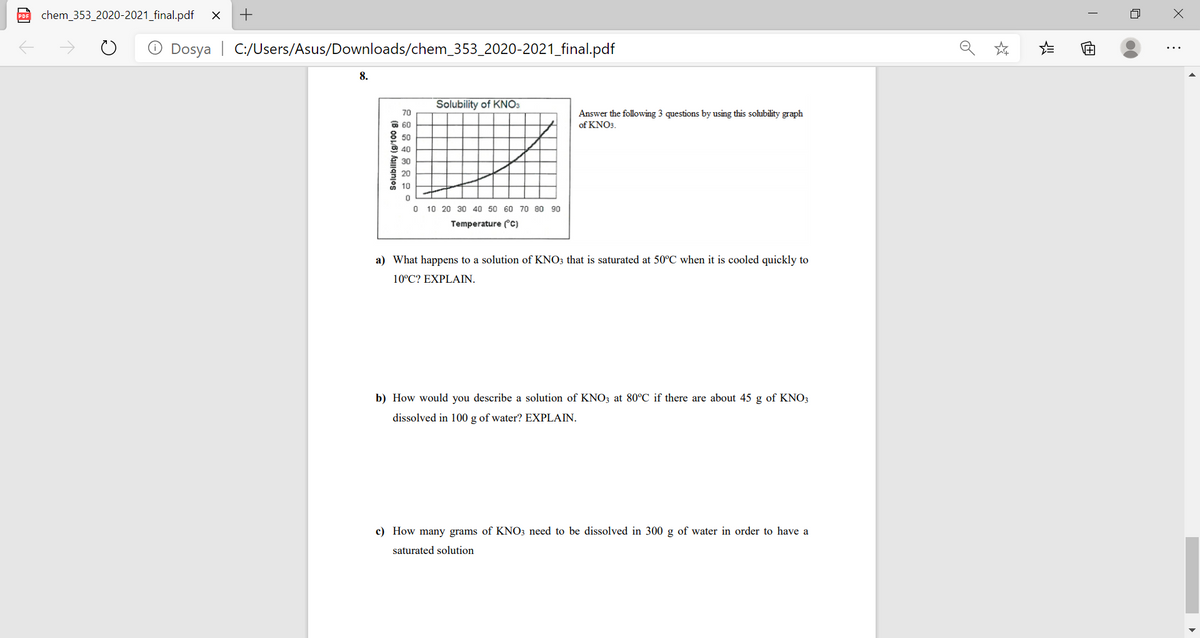
Solubility of KNOs 70 Answer the following 3 questions by using this solubility graph of KNOs. 60 50 40 30 20 10 O 10 20 30 40 s0 60 70 80 90 Temperature (C) a) What happens to a solution of KNO; that is saturated at 50°C when it is cooled quickly to 10°C? EXPLAIN. b) How would you describe a solution of KNO, at 80°C if there are about 45 g of KNO, dissolved in 100 g of water? EXPLAIN. (8 0OL6) Aamanjos
Solubility of KNOs 70 Answer the following 3 questions by using this solubility graph of KNOs. 60 50 40 30 20 10 O 10 20 30 40 s0 60 70 80 90 Temperature (C) a) What happens to a solution of KNO; that is saturated at 50°C when it is cooled quickly to 10°C? EXPLAIN. b) How would you describe a solution of KNO, at 80°C if there are about 45 g of KNO, dissolved in 100 g of water? EXPLAIN. (8 0OL6) Aamanjos
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.26QAP
Related questions
Question
Transcribed Image Text:PDF
chem_353_2020-2021_final.pdf
Dosya | C:/Users/Asus/Downloads/chem_353_2020-2021_final.pdf
Solubility of KNO3
70
Answer the following 3 questions by using this sohubility graph
of KNO3.
O 60
50
O 40
30
20
10
10 20 30 40 50 60 70 80 90
Temperature (c)
a) What happens to a solution of KNO3 that is saturated at 50°C when it is cooled quickly to
10°C? EXPLAIN.
b) How would you describe a solution of KNO3 at 80°C if there are about 45 g of KNO3
dissolved in 100 g of water? EXPLAIN.
c) How many grams of KNO3 need to be dissolved in 300 g of water in order to have a
saturated solution
Solubility (g/100 g)
of
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

