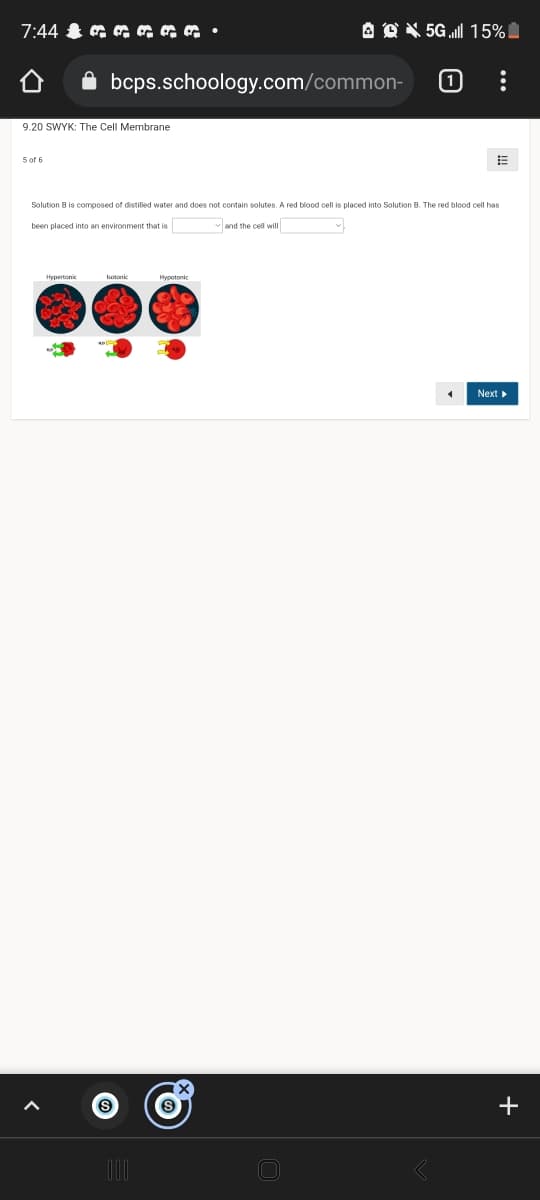
Solution B is composed of distilled water and does not contain solutes. A red blood cell is placed into Solution B. The red blood cell has been placed into an environment that is and the cell will Hypertone hotonk Hypotonie
Solution B is composed of distilled water and does not contain solutes. A red blood cell is placed into Solution B. The red blood cell has been placed into an environment that is and the cell will Hypertone hotonk Hypotonie
Chapter2: Crystallization
Section: Chapter Questions
Problem 3Q
Related questions
Question
Fill in the blank
Transcribed Image Text:7:44 & G GGa •
A OX 5G „ll 15%.
bcps.schoology.com/common-
(1
9.20 SWYK: The Cell Membrane
5 of 6
Solution B is composed of distilled water and does not contain solutes, A red blood cell is placed into Solution B. The red blood cell has
been placed into an environment that is
vand the cell will
Hypertonic
Hypotonic
000
Next
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning