Some indoor air-purification systems work by converting a little of the oxygen in the air to ozone, which oxidizes mold and mildew spores and other biological air pollutants. The chemical equation for the ozone generation reaction is: It is claimed that one such system generates 4.00 g O3 per hour from dry air passing through the purifier at a flow of 5.00 L/min. If one liter of indoor air contains 0.280 g of O2, what mole fraction of O2 is converted to O3 by the air purifier? What is the percent yield of the ozone generation reaction?
Some indoor air-purification systems work by converting a little of the oxygen in the air to ozone, which oxidizes mold and mildew spores and other biological air pollutants. The chemical equation for the ozone generation reaction is: It is claimed that one such system generates 4.00 g O3 per hour from dry air passing through the purifier at a flow of 5.00 L/min. If one liter of indoor air contains 0.280 g of O2, what mole fraction of O2 is converted to O3 by the air purifier? What is the percent yield of the ozone generation reaction?
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 6QRT
Related questions
Question
Some indoor air-purification systems work by converting a little of the oxygen in the air to ozone, which oxidizes mold and mildew spores and other biological air pollutants. The chemical equation for the ozone generation reaction is:
It is claimed that one such system generates 4.00 g O3 per hour from dry air passing through the purifier at a flow of 5.00 L/min.
If one liter of indoor air contains 0.280 g of O2, what mole fraction of O2 is converted to O3 by the air purifier?
What is the percent yield of the ozone generation reaction?
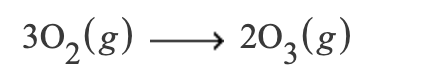
Transcribed Image Text:30,(g) –→ 20,(g)
Expert Solution
Step 1
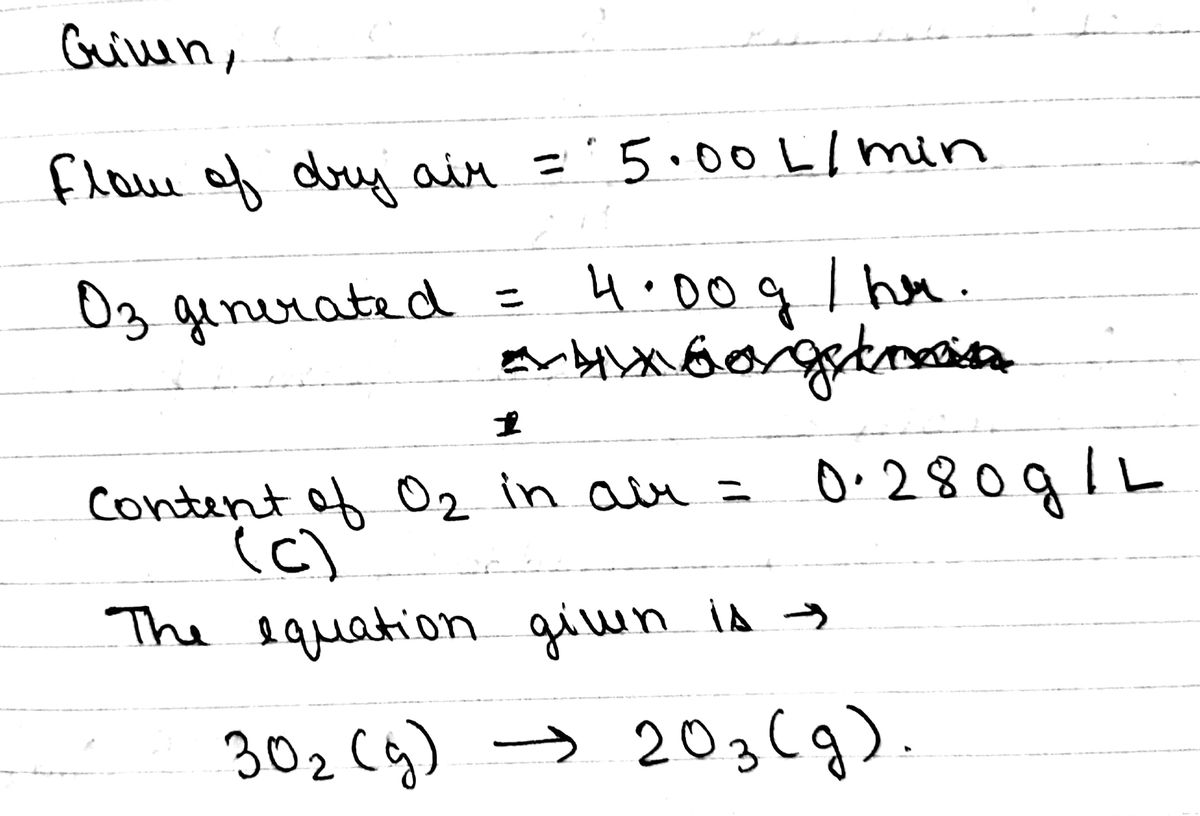
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div
