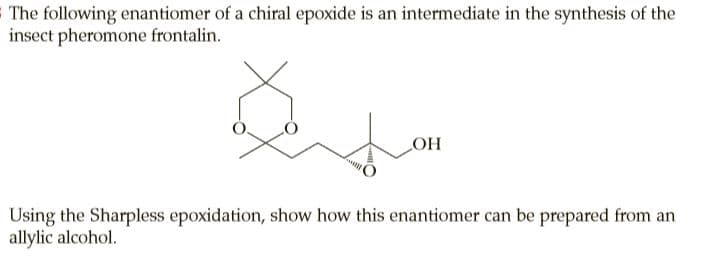
SThe following enantiomer of a chiral epoxide is an intermediate in the synthesis of the insect pheromone frontalin. OH Using the Sharpless epoxidation, show how this enantiomer can be prepared from an allylic alcohol.
Q: HO CH2CH2NH2 The compound is an alkaloid found in ripe cheese. Show how you would synthesize it from…
A: In this question, we have to synthesis the given compound from the starting material benzene and…
Q: Sharpless epoxidation of allylic alcohol X forms compound Y. Treatment of Y with NaOH and C6H5SH in…
A: Two different chiral diethyl tartrate isomers, (+)-DET and (-)-DET are used to indicate the…
Q: 24) Upon azonolysis, compound X produces 2 compounds: Y and Z. can be prepared from the follory…
A:
Q: Sodium Borohydride Reduction 1. Give stereochemical formulas (such as Fischer projects) for all the…
A: Here we have to write structures of all stereo isomers that are fomed in the reduction of following…
Q: HO .CH2CH2NH2 The compound is an alkaloid found in ripe cheese. Show how you would synthesize it…
A: In this question, we have to synthesis the given organic compound from the starting material benzene…
Q: Compound J, C16H16Br2, is optically active. On treatment with strong base, compounds K and L (each…
A:
Q: 14. When an aqueous solution of (R)-2-butanol is treated with a catalytic amount of H2SO4, slow…
A:
Q: Chemistry please provide a mechanism with o-cresol nitration arrows with possible mesomeric,…
A: Here we are required to draw the mechanism for nitration of o-cresol.
Q: Treatment of Compound E with sodium amide and then methyl iodide does not produce the desired…
A: -> NaNH2 is a base hence, it can abstract most acidic hydrogen from compound and generate…
Q: The cis and trans isomers of 2,3-dimethyloxirane both react with −OH to give butane-2,3diol. One…
A: In the given reaction, both cis and trans isomers of 2, 3-dimethyloxirane react with -OH (a strong…
Q: 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is…
A: Since, you have asked multiple question, we will solve the first question for you. If you want any…
Q: A chiral ether of molecular formula C5H100 reacts with hot HI to give a product of molecular formula…
A: Molecular formula of reactant= C5H10O Molecular formula of product= C5H10I2
Q: The sex attractant of the female winter moth has been identified as the tetraene…
A:
Q: Show how you would synthesize the following compounds, starting with benzene or toluene and any…
A: Synthesis of 3-phenylpropan-1-ol:
Q: Compound X has molecular formula C7H14. Hydrogenation of compound X produces 2,4-dimethylpentane.…
A:
Q: Propylene oxide is a chiral molecule. Hydrolysis of propylene oxide gives propylene glycol, another…
A: A. The mechanism of acid catalyzed hydrolysis of (R)-propylene oxide (treatment with H2SO4/H2O)…
Q: 12. When 1-chloronaphthalene is treated with sodium amide, Na+NH2 , in the presence of piperidine,…
A: Here we have to explain the mechanism of reaction when 1- chloronaphthalene react with piperidine in…
Q: When cis-2-decalone is dissolved in ether containing a trace of HCI, an equilibrium is established…
A: Organic chemistry involves reactions of organic molecules. The reactions can show changes in cis and…
Q: 2. The following pyrrole derivative is a drug under development for schizophrenia and it is prepared…
A:
Q: Glucuronic acid is an oxidized derivative of glucose found in animal secretions such as saliva.…
A: First step of this reaction is protonation where glucuronic acid is attacked by H+ ion in the…
Q: 11. Compound A, C9H17N, is an optically inactive alkaloid, containing a tertiary amine that is not…
A: TERTIARY AMINE: It is also known as 3∘ amine. An amine chemical functional group in which nitrogen…
Q: Either enantiomer of the SULFOXIDE below may be prepared from (S)-2-butanol. Propose an efficient…
A: The structure is; The name of the above structure is (S)-2-butanol. The structures of the product…
Q: OMe 11. Me
A: 11. Step 1 : Birch reduction. Step 2 : acid treatment ( double bond isomerization). Step 3 :…
Q: 5) Draw the structure, showing the stereochemistry, of the cyclic products 19 and 20, and the…
A:
Q: The Sharpless epoxidation is used when a single enantiomer product is required. Predict the…
A: Sharpless epoxidation allows the enatioselective epoxidation of allylic alcohols. The oxidizing…
Q: 1.Treatment of 3-methyl-2-butanol with hydrogen chloride yielded only a trace amount of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: HO -CH2CH2NH2 The compound is an alkaloid found in ripe cheese. Show how you would synthesize it…
A: In this question we have to synthesized the given compound from benzene as a starting material. You…
Q: Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B.…
A: Organic transformation.
Q: Sharpless epoxidation of allylic alcohol X forms compound Y. Treatment of Y with NaOH and C6H5SH in…
A:
Q: (b) Draw the structure of compound A and suggest a reaction mechanism for the transformation of A…
A:
Q: OH Br. + Br, + H,O + HBr 1-Bromo-2-pentanol (a racemic mixture) 1-Pentene Treating 4-penten-1-ol…
A: In an organic reaction, a nucleophile attacks an electrophile.
Q: Show how to prepare each alcohol from an organohalogen compound and an oxirane.
A: Alcohol is a very important compound in organic chemistry. It can be prepared by many methods.
Q: Show how you would synthesize the following compounds, starting with benzene or toluene and any…
A: Starting material : Benzene
Q: Give the major product when 2-bromobutanal reacts with acidic aqueous KCN.
A: the major product when 2-bromobutanal reacts with acidic aqueous KCN.
Q: When an aqueous solution of (R)-2-butanol is treated with a catalytic amount of H2SO4, slow…
A:
Q: Show how you would synthesize the following compounds, starting with benzene or toluene and any…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 1) NANH2 2) benzyl chloride/acetone SH
A: When thiol react with a base, it abstracts the proton and then thiol becomes a very good…
Q: 2-Ethyl-1-hexanol was needed for the synthesis of the sunscreen octyl p-methylcinnamate. Show how…
A: (a) Addition reaction of ketones and aldehydes is an aldol reaction and it is reversible reaction.…
Q: A chiral ether of molecular formula C5H10O reacts with hot HI to give a product of molecular formula…
A:
Q: M-C-H AICI3 t. HBr D + [o] room temperature (A)
A:
Q: Each diastereomer, if treated with sodium ethoxide (NaOCH2CH3), undergoes an E2 reaction. One of…
A: In an E2 elimination, the two leaving groups must be antiperiplanar to each other.
Q: Show the Conversion of Alcohols to Alkyl Tosylates with p-toluenesulfonyl chloride (TsCl) in the…
A: Alcohols can be converted to tosylate on treatment with p-toluene sulfonyl chloride (TsCl) in the…
Q: 7. Compound X is an alcohol. Upon oxidation by acidify potassium permanganate, a ketone, Y is…
A:
Step by step
Solved in 2 steps with 1 images

- Ammonia and amines react with epoxides with the same stereospecificity as anionic nucleophiles. Draw a sawhorse or Newman projection formula for the product of the reaction shown, clearly showing the stereochemistry at both chirality centers. What are the Cahn–Ingold–Prelog R,Sdescriptors for these chirality centers in the reactant and the product?The chiral hydroxyl compound P, C7H14O contains a five-membered ring and showspositive result in iodoform test. Compound P and sodium bromide can be produced byrefluxing compound Q with aqueous NaOH solution. Compound P is heated withbenzoic acid in the presence of concentrated H2SO4 to form a sweet-smelling product,compound R. Deduce the structure of P, Q and R. PLEASE PROVIDE CLEAR HAND WRITING AND SOLUTIONSSharpless epoxidation of allylic alcohol X forms compound Y. Treatmentof Y with NaOH and C6H5SH in an alcohol–water mixture forms Z. Identifythe structure of Y and draw a mechanism for the conversion of Y to Z.Account for the stereochemistry of the stereogenic centers in Z. Z hasbeen used as an intermediate in the synthesis of chiral carbohydrates.
- Show how you would synthesize each compound using methylenecyclohexane asyour starting material. If a chiral product is shown, assume that it is part of a racemicmixture.A compound with the formula C8H12 absorbs two equivalents of H2 on Catalina reduction over a palladium catalyst to give compound A. Ozonalysis oxidative cleavage of the unknown compounds gives two compounds B and C.What allylic alcohol and DET isomer are needed to make each chiralepoxide using a Sharpless asymmetric epoxidation reaction?
- Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a. How many stereogenic centers are present in the product?b. Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms two products having different melting pointsand somewhat different solubilities. Draw the structure of these twoproducts. Assign R and S to any stereogenic centers in the products.How are the two products related? Choose from enantiomers,diastereomers, constitutional isomers, or not isomers of each other.When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the structure of the product and indicateits stereochemistry.
- Compound J, C16H16Br2, is optically active. On treatment with strong base, compounds K and L (each C16H14) are formed; K and L each absorb only 2 equivalents of hydrogen when reduced over a Pd/C catalyst. Compound K reacts with ozone to give phenylacetic acid (C6H5CH2COOH), while similar treatment of L gives 2 products. One product, M, is an aldehyde with formula C7H6O; the other product is glyoxal (CHO)2. Draw the structure of compound L.Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral αhydroxy acid A. Identify B and C in the following reaction sequence, and designate the conguration of the enantiomer formed by this route as R or S.Chiral alcohols can be converted to chlorides by several pathways. Which is the best way to make the chloride with retention of configuration?

