STRUCTURE: H (c) (d) H CH3 (b) O-H (a) (e) H H (f) For the IR spectrum shown below*, identify the structural features which cause each of the major IR signals. 100 D 4000 3000 2000 1500 1000 MAVENUERI
STRUCTURE: H (c) (d) H CH3 (b) O-H (a) (e) H H (f) For the IR spectrum shown below*, identify the structural features which cause each of the major IR signals. 100 D 4000 3000 2000 1500 1000 MAVENUERI
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter4: Nomenclature
Section4.1: Naming Binary Compounds
Problem 5RQ
Related questions
Question
Please answer the following related to
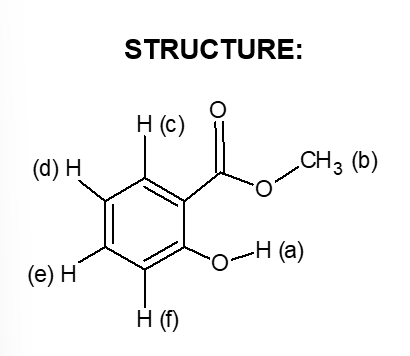
Transcribed Image Text:STRUCTURE:
H (c)
(d) H
CH3 (b)
O-H (a)
(e) H
H (f)
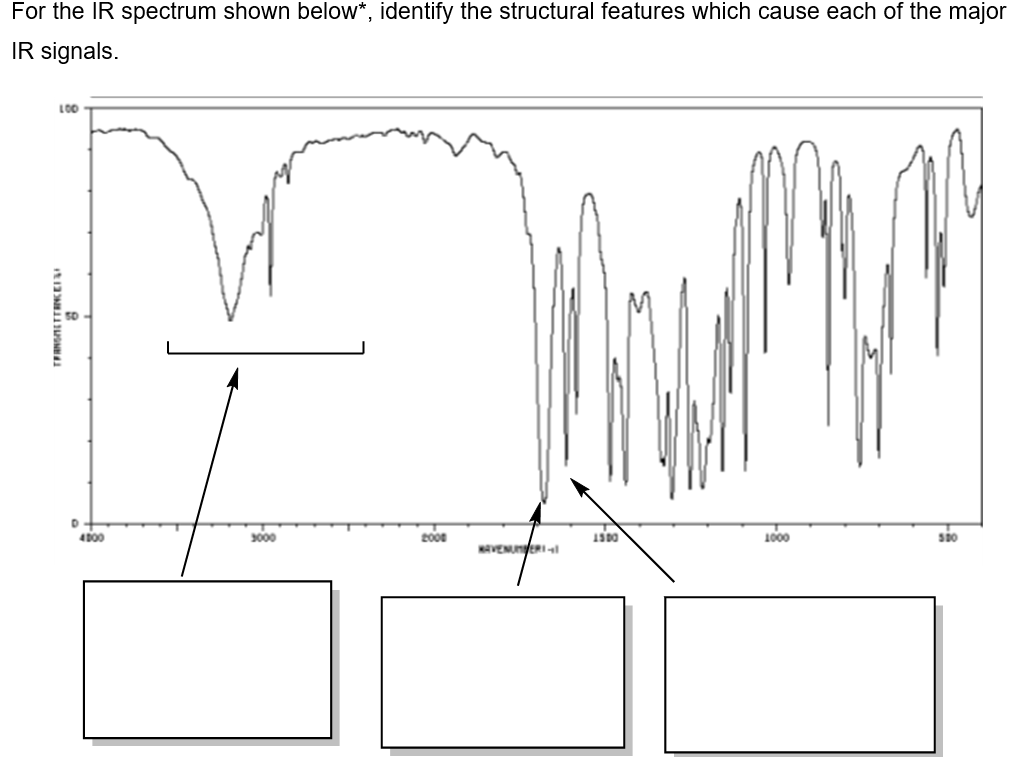
Transcribed Image Text:For the IR spectrum shown below*, identify the structural features which cause each of the major
IR signals.
100
D
4000
3000
2000
1500
1000
MAVENUERI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps with 1 images

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning