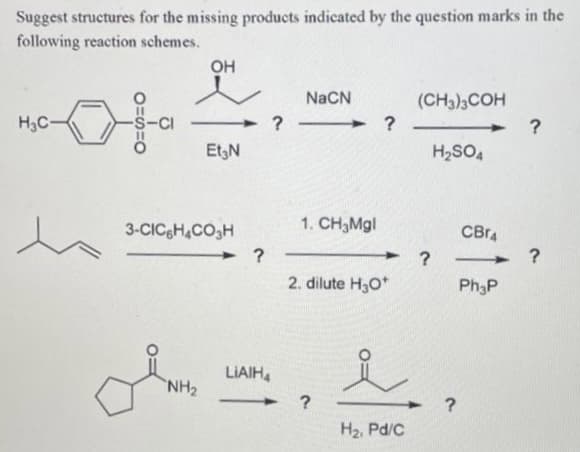
Suggest structures for the missing products indicated by the question marks in the following reaction schemes. Он NaCN (CH3)3COH H3C- Et3N H2SO4 3-CICH,CO,H 1. CH3MG| CBr4 2. dilute H30* Ph3P LIAIH, NH2 H2, Pd/C
Q: Give the major organic product(s) of each of the following reactions. If none is predicted, write…
A: We need to determine the product formed when bromine reacts with toluene.
Q: plete the synthetic sequences by drawing products/substrates/reagents in empty spaces in reactions…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: Provide answers to sections (a) to (c) for each of the organic compounds represented below. (a)…
A: Basic concepts of Organic chemistry.
Q: CH,0 CH,CH OH C.
A: 1) Hydroboration- Oxidation - The addition of borane followed by oxidation is known as the…
Q: OH CI CF 3 1) Na H 2) NaNH, NH3 NO₂ ? ?
A:
Q: Not all tertiary haloalkanes undergo S,1 reactions readily. For example, the bicyclic com- pound…
A:
Q: KMNO4, hot -CH2-CH2-CH H*
A:
Q: KMNO4, hot -CH2-CH2-C-H H*
A: In this question, we want to draw the major products. You can see details Solution below.
Q: 1. Sn, HCI A 2. OH" HNO3 Cl2 H2SO4 FeCl3
A: A sequence of reactions of electrophilic substitution, which is to be accomplished.
Q: What is the final product of the following reaction sequence? Give structural details of all…
A: 1) Removal of most acidic H by NaH.
Q: The reaction of 1-iodopropane with potassium thiocyanate (KSCN) in certain solvents results in the…
A: A. 1-Propene undergo addition reaction with H-SCN via carbocation formation. Due to greater +I and…
Q: Br CH3 CH3CHCHCH2ĊHCH3 Br OH 1. Hg(OAc)2, H2O CH3CH2CH2ĊHCH3 2. NABH4 ÇI HCI CH3CHCHCH,CH2CHCH3 CH3…
A:
Q: -CH2-CH2-C-H KMNO4, cold dilute
A: In this question, we will draw the major product for the given Reaction. You can see, details…
Q: In the synthesizing of cis-2-hexene as a product consist of mixing of 1-pentyne and an alkyl halide.…
A: We have to predict the Reagent and reaction involved.
Q: Gee twee moontlike [1,2]-herrangskikkings- reaksieprodukte waar h alkielgroep migreer vanaf die…
A: The reaction given is,
Q: Chemical Engineering 7) Propose multistep syntheses for the following molecules. i EtO OEt MeO OMe
A: Note - Since the given question is a multiple question, hence I solved first question according to…
Q: A2. Complete the following by writing the structure of the principal organic product(s) for each…
A: According to the guidelines prescribed by bartleby, we can answer a maximum of three subparts.…
Q: 1. Sn, HCI A 2. OH HNO3 Cl2 H2SO4 FeCl3
A:
Q: CH3 1. Hg(OAc)2, H₂O 2. NaBH4 1. LDA 2. PhCH₂Br ? ?
A: Here we are required to predict the product of the reaction
Q: (7) Complete the following outlined reactions by providing the major organic product expected in…
A: We have to carry out the given transformation.
Q: in the reaction below. R SR DƏSn SI YU NH2 HO, HX + NEN What functional-group transformation occurs…
A: A) The given reaction is a transformation from benzoic acid to aniline. The functional group…
Q: Provide the major monobrominated product of the reactions below Br₂, hv i) Br₂, hv ii) In the…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: 6. In an organic chemistry lab, chemists were attempting to convert compound I into compound II via…
A:
Q: Which of the alkenes below is the most reactive towards acid-catalyzed hydration?
A: Markovnikovs rule is applied to acid catalysed hydration of alkene.
Q: H2, Pd/C ELOH
A:
Q: Question is attached
A: These are the organic reaction, in which from two or more reactants it gives now product by breaking…
Q: The discoverer of the dihydroxylation reaction, prof. K. Barry Sharpless, is blind on one eye.…
A:
Q: 2. Write suitable detailed mechanisms to account for the formation of the products in the following…
A:
Q: Complete the Reaction below by providing the major product and write what the type of reaction is…
A:
Q: KMNO4, cold. -CH2-CH2-C-H dilute
A: The question is based on the concept of organic reactions. we have to identify the Product formed…
Q: (i) CH3-CEC-CH3 + H2 Lindlar CH3OH А В H2SO4
A: Below attached file showing the details answer. All the best.
Q: 2NaOH O Na HCI O-H O-O, H2O, 0-5°C D
A:
Q: 3 Give mechanisms for the acid-catalyzed and base-promoted alpha-halogenation ofketones. Explain why…
A: The mechanisms for the acid-catalyzed alpha-halogenation of ketones is,
Q: Provide the structure of the major organic product of reaction 1 and the major organic product after…
A: Concept introduction; When alcohol treated with thionyl chloride in presence of base OH group is…
Q: Propose a structural formula for compound D and for the product, C,H,,0, formed in this reaction…
A: solution;- Salfonium salt react with a strong base gives sulfur ylide and also gives desirable…
Q: What is the chemoselectivity issue in the following reaction? You may wish to consult the datasheet…
A: GIVEN:-
Q: Alkenes are more reactive than alkynes toward addition of electrophilic reagents (i.e., Br2, Cl2, or…
A: A question based on electrophilic addition reactions, which is to be accomplished.
Q: The Strecker reaction is a three component reaction (aldehyde/ketone, amine, and cyanide) used to…
A: Here we are required to show the mechanism for the synthesis of phenylalanine using strecker…
Q: CEN O OCH3 1. LDA 2. CH3Br 1. LDA 2. CH3CH₂Br ? ?
A:
Q: Consider the following schematic diagram. OH OH Reagent II LOCH; Reagent I H,C- OCH3 H,C. Compound M…
A: In this question, given the compund we have to identify the reagent for suitable for the reaction.
Q: What are the names of the compound A, B, C, D, E, F, G & H. Hence suggest structures for compound C…
A:
Q: 9. H3C-C -CI + NaBr
A:
Q: (c) CH3-CECH 2C12
A: Alkyne on addition with 1 equivalent Cl2 form alkene and 2 equivalent Cl2 form alkane.
Q: Choose the best reagent(s) from the list provided below for carrying out the following reaction…
A:
Q: Will all the reactions below proceed to completion? Provide an explanation. CH3CH2Br + NaOCH3 CH3B +…
A:
Q: N CH3 CH3 1. NBS, AIBN 2. NaOH ?
A:
Q: 2. Devise reactions schemes to carry out the following multi-stage processes to make ethyl ethanoate…
A: Interpretation - To make ethyl ethanoate from using the ethanol as the organic reagent - To make…
Step by step
Solved in 2 steps with 2 images

- can i get help drawing out actual structures including the nucleophilic addition of Cy2NH to parafomaldehyde and its hemiaminal intermidiate and the condensation step when it is displaced by terminal alkyne forming allene, also what is dioxane getting rid of as the solvent, thanksFill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.Help with the following ochem reaction scheme... Consider 3,4-dimethylpiperidine being subjected to the below reaction scheme: step 1) CH3I (excess) step 2) NaOH, heat step 3) CH3I (excess) step 4) NaOH, heat Provide the bond line structures for the major organic product obtained in each step and then discuss the regiochemistry for step 2.
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- The reaction of 1-iodopropane with potassium thiocyanate (KSCN) in certain solvents results in the formation of two isomeric products, propylthiocyanate and propylisothiocyanate (see scheme below), via the SN2 reaction mechanism. Attempts to prepare a similar mixture of these same isomeric products (propylthiocyanate and propylisothiocyanate) starting from 1-propene is illustrated below. Despite the strong acidity of thiocyanic acid (recall pKa = 1.1), this addition reaction does not lead to either of the products indicated. Based on your knowledge of alkene addition reactions, explain this experimental result.Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?Complete and predict the regioselectivity of the following pericyclic reaction: Explain your answer using FMO principles
- EXPLAIN BY WORDS IN ORDER FOR ME TO UNDERSTAND THE GENERAL REACTION SCHEME. IN THIS CROSS-COUPLING REACTION WHICH ORGANOHALIDE (R-X) REACTS WITH ORGANOBORANE [R1B(OR)2].Give the major organic product(s) of the reaction shown below. Be sure to show all stereoisomers formed, and indicate which isomers are formed in equal and unequal quantities.Give the condensed structure of the product/s for the reactions below and indicate for each the mechanism involved in the formation of such product/s as Sn1, Sn2, E1, E2. Please note that it must be in condensed structure: 1. BrCH2CH2CH2Br + Mg (ether)

