Suppose 2.0 mol of an ideal gas is taken from a volume of 3.5 m to a volume of 1.7 m3 via an isothermal compression at 35°C. (a) How much energy is transferred as heat during the compression, and (b) is the transfer to or from the gas? (a) Number Units (b) Use correct number of significant digits; the tolerance is +/-2%
Suppose 2.0 mol of an ideal gas is taken from a volume of 3.5 m to a volume of 1.7 m3 via an isothermal compression at 35°C. (a) How much energy is transferred as heat during the compression, and (b) is the transfer to or from the gas? (a) Number Units (b) Use correct number of significant digits; the tolerance is +/-2%
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 54P: Two moles of a monatomic ideal gas at (5 MPa, 5 L) is expanded isothermally until the volume is...
Related questions
Question
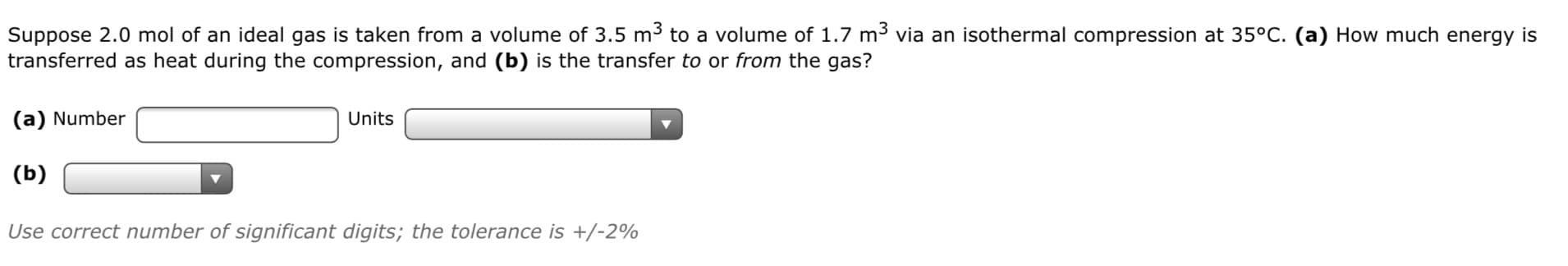
Transcribed Image Text:Suppose 2.0 mol of an ideal gas is taken from a volume of 3.5 m to a volume of 1.7 m3 via an isothermal compression at 35°C. (a) How much energy is
transferred as heat during the compression, and (b) is the transfer to or from the gas?
(a) Number
Units
(b)
Use correct number of significant digits; the tolerance is +/-2%
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning