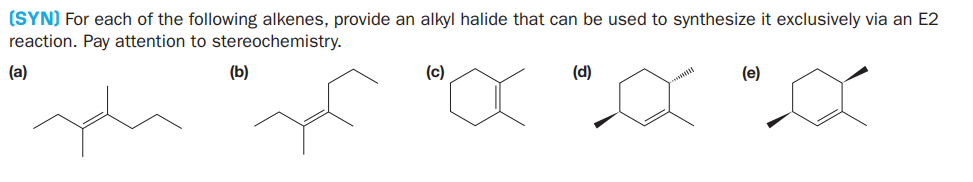
(SYN) For each of the following alkenes, provide an alkyl halide that can be used to synthesize it exclusively via an E2 reaction. Pay attention to stereochemistry. (a) (b) (c) (d) (e)
Q: ): In the space provided below please give the detailed mechanism for the following transformation.…
A: The reaction given is,
Q: Provide specific examples/reaction scheme/mechanism for each of the following: a) Hoffman…
A: Hoffman elimination: When a quaternary ammonium hydroxide is strongly heated it decomposes to yield…
Q: The electrostatic potential maps of benzene and pyridine are shown here. Is the electrostatic…
A: Electrostatic potential map tells us about the charge distribution within a molecule. By knowing…
Q: v.. Using curved arrow notation, propose a detailed mechanism that is plausible for the following…
A: This is an example of formation of bromonium ion followed by intramolecular SN2 reaction.
Q: What is the slow (rate-determining) step in any electrophilic aromatic substitution reaction? Please…
A: Please find below a example of the reaction mechanism
Q: Which reaction in each of the following pairs would you expect to be faster? (i) Write both…
A: The SN1 reactions are also called the nucleophilic substitution reaction where the attacking…
Q: Which of the following could be synthesized utilizing NBS? including curved arrows and intermediates…
A: NBS is N-bromosuccinimide. It is used for bromination. In NBS bromine is attached to nitrogen. It…
Q: In the protonated epoxide shown here, which C-0 bond would you expect to be longer? Why? ?
A: The ring opening reaction of the epoxide in acidic conditions is an example of SN1 reaction. The…
Q: d. Consider the following secondary halides. Changing the leaving group from fluoride to bromide…
A: Halides are good leaving groups. The tendency of the leaving group increases as the size of the…
Q: Explain why A cross couples with the aryl iodide, rather than two molecules of A coupling together…
A: Aryl coupling is commonly a cross-coupling reaction. That means the reaction starts when two…
Q: Given the information below, write out a reasonable mechanism for the reaction. Where not provided,…
A: Mechanism is drawn below--
Q: Predict the product of the reaction between acetic anhydride and each of the following compounds. If…
A: As here question has multiple subparts, so solve first three subparts. If student want complete…
Q: Provide a multi-step synthesis of the target structure on the right from the starting structure on…
A:
Q: (SYN) How would you synthesize the compound shown here using phenylethanal as your only source of…
A: The compound that is to be synthesized is 1,4-diphenylbut-2-ene.
Q: Show the complete mechanism using Curved Arrow Formalism for the reaction of the compound shown…
A: INTRODUCTION: p-chlorobenzaldehyde reacts with EtOH and HCl to give an acetal.
Q: For which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be…
A: We know that SN1, E1 and SN2, E2 mechanisms follow the first and second order kinetics respectively.
Q: A pair of diastereomers is each individually mixed with a strong base, and for both, an E2 reaction…
A: Diastereomous are ------Now if there two inomous undergo e2 -elimination.
Q: The phenyl group, C,H, is known to be an ortho/para-directing group. (a) With that in mind, predict…
A: Para product is majored.
Q: 3) Give a complete, detailed mechanism for the following transformation. C, HCI HCI
A:
Q: (b) ethanol, HCI соон heat
A: Esterification reaction: Carboxylic acid reacts with alcohol in the presence of an acid catalyst to…
Q: provide full and detailed reaction mechanism given the following reactants, products and conditions
A: Given; structure of reactant and products
Q: In each case below select the synthetic procedure/s that could be used to carry out the giving the…
A: Given : structure of reactant and products. Both the reaction are addition of water.
Q: (SYN) Show how you would carry out the synthesis at the right using the starting material given,…
A: Alkynes- Unsaturated hydrocarbon containing carbon carbon triple bond is known as alkyne. Alkynes…
Q: Predict the major product for each of the following reactions:
A:
Q: Predict the main mechanism that occur for the following reaction
A: Nucleophilic substitution reactions the electron-rich nucleophile attacks on a positively charged…
Q: Please be certain to use good curved arrow notation for your mechanism. HYH2O HO,
A:
Q: Please help me, I have the mecahnism good but I am very confused on what the intermediate and…
A:
Q: Show a curved arrow mechanism for the formation of the major product below. major minor The…
A:
Q: What is the curved arrow notation for the formation of the following alkene?
A:
Q: Draw the complete, detailed E1 mechanism for each of the following reactions, and show all resonance…
A: E1 mechanism follows through two steps 1) In the first step by eliminating leaving group (halide…
Q: React
A: Available Reagent is : NaH, THF CH3-Br This reaction is called " Williamson etherification…
Q: Complete the following reactions, clearly indicating regio-and stereochemistry where applicable. In…
A: Aromatic oxidation of the side chain leads to the formation of carboxylic acids in the presence of…
Q: (a) For which aromatic compound do you expect nitration to take place faster: furan or thiophene?…
A:
Q: Complete the following reaction and provide the detailed mechanism NaOH NaOH
A: Intramolecular Aldol condensation- Molecule containing two carbonyls functionalities on reaction…
Q: (SYN) Draw the dienophile that would react with buta-1,3-diene to produce each of the following…
A: The diene and the dienophile can be obtained by performing retrosynthesis of the given Diels Alder…
Q: Provide a reasonable mechanism using curved arrow notation for the following transformation, which…
A:
Q: When the depicted alkyne is treated first with sodium amide and then with the alkyl halide shown,…
A: We have given when alkyne is treated first with sodium amide and then with the alkyl halide. It is…
Q: Draw a detailed mechanism for the following reaction. Please include all lone pairs and charges
A:
Q: Provide the full radical mechanism with all arrows and intermediates shown in the following…
A: We have to predict the mechanism for given reaction.
Q: Identify the product of the following one-step sequence. Note that this is a reaction that you have…
A:
Q: This is a retro synthetic analysis problem, shown here is the final product (initial material is…
A:
Q: (SYN) Show how you would carry out each of the following transformations.
A: Electron donating groups activate the benzene ring towards electrophilic substitution reaction and…
Q: Here is a reaction leading to the formation of a racemic mixture of alcohols as products majors:…
A:
Q: Provide the complete mechanism using Curved Arrow Formalism for the reaction of…
A: Please find the attachment.
Q: In cach case below select the synthetic procedure/s that could be used to carry out the…
A: Alkene iBH3iiH2O2,OH- antimarkovnikov's addition of H2O (via non-classical carbocation intermediate)…
Q: b) T.
A: The question is based on the concept of organic reactions. We have to identify the reagents used in…
Q: For each of the following reactions draw the structure of the major organic product in the box…
A: Hello. Since the question contains multiple sub-parts, the first three sub-parts shall be solved…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

- Can you please help with the following organic chemistry reaction (see attached image) Provide the mechanism involved in the reaction and what the major product(s) would be. Thank youChoose the best reagents to complete the reaction shown below. Should correct, also give it's arrow mechanismPlease provide a detailed mechanism for the following transformation. Also, show the appropriate curved arrows to rationalize the next step. The quality of the electron pushing counts.
- Predict the major, organic product for the following reaction sequence without sterechemistry depicted then predict the stereochemical outcome of the reaction sequence.provide full an detailed reaction mechanism given the following reactants, products, and conditionsWhich of the following carbocations would be to rearrange? Why?
- Draw a detailed mechanism, using arrow formalisms, for the reaction shownbelow. Be sure to include all resonance structures, where appropriate. Use to curved arrow notation, propose a mechanism for the following reaction and state whether it is either SN1, E1, SN2, or E2. Give the IUPAC names of all organic reactants and products.please help with this question. thank you. Propose an efficient synthesis of for each of the following transformation.
- Provide a complete mechanism for the following reaction. Pay close attention to details, including lone pairs, formal charges and the use of curved arrows.Please provide a complete, detailed curved-arrow mechanism for the following reaction.Include ALL lone pairs and formal charges. Using the mechanism and a few words,explain the why the NaH deprotonates at the selected a-position and not the other a-position. Also explain why the indicated alkene is formed in the 3rd step rather than the other possible alkene product.Give a clear handwritten mechanism and product for given below Sn2 reaction..a reaction shows all bonds ,lone pairs and arrows...?

