Q: ): In the space provided below please give the detailed mechanism for the following transformation.…
A: The reaction given is,
Q: Draw the complete electron pushing mechanism for the following transformations
A:
Q: (SYN) Draw the alkyne that, when treated with diazomethane and irradiated with ultraviolet light,…
A: The product has a cyclopropene ring with one methyl and one propyl substituents on the double bonded…
Q: Draw the mjor product of both question with mechanism asap
A: Given structure of reactants.
Q: Need help. Please draw out mechanism on how to get the product.
A:
Q: (SYN) Which solvent-acetone or tert-butyl alcohol-would be better to use to carry out the reaction…
A: E1 reaction is a unimolecular elimination reaction, which involves the removal of an HX substituent…
Q: Draw the mechanism for the following reaction with arrows and include all intermediates. Include…
A: The F-NR2 attacks on the triple bond of the compound and results in the formation of an allene…
Q: Q2. Complete the following reaction. Show mechanism CI
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Draw the products of attached reaction by following the curved arrows.
A: The products of attached reaction can be drawn as
Q: Bonus: What anion is a more reactive nucleophile? Why? Must draw a picture! VS.
A:
Q: Draw the complete, detailed mechanism for the reaction: conc HCI ?
A: In rection of alkene , at first, alkene donate electron to H+ and carbocation intermediate is…
Q: (SYN) How would you synthesize the compound shown here if, as your starting material, you may use…
A: PCC- It is Pyridinium Chloro chromate. It is used as good oxidizing agent. It is used in oxidation…
Q: alkyl halide is most likely to A undergo the which forms a carbocation intermediate. reaction…
A: There are basically two types of substitution reactions ;- SN1 (nuclephilic substitution…
Q: The attached reaction does not afford the major product that is given.Explain why this is so, and…
A:
Q: Complete the following reaction scheme , Complete the following reaction scheme
A:
Q: Draw a mechanism for the reaction of methylamine with formic acid. In the box to the left, draw any…
A:
Q: Which nucleophiles will add to the following conjugated enone at the A carbon? Which will add at…
A: The compound given is,
Q: n two of uiese Teaction sequences will this transformation?
A:
Q: What is the curved arrow notation for the formation of the following alkene?
A:
Q: Problem: Consider the addition of HCl shown below. (a) Draw the arrows for the first step of the…
A: Addition of HCl to the alkene result in the formation of haloalkane. In the first step carbocation…
Q: Draw the complete, detailed E1 mechanism for each of the following reactions, and show all resonance…
A: E1 mechanism follows through two steps 1) In the first step by eliminating leaving group (halide…
Q: alkene to an >> reaction
A:
Q: Draw the complete, detailed mechanism for each of the following reactions and predict the major…
A: Note - Since you have asked multiple question, we will solve the first question for you. If you want…
Q: Draw the curved arrow mechanism for the following reaction:
A: Reaction mechanism;
Q: Consider the mechanistic step shown below. Which of the four characteristic patterns of arrow…
A: We have to predict: Arrow pushing pattern Place for Head of curve arrow.
Q: Draw the major product of the following reaction and enter its InChl code in the space provided.…
A: This is a nitration reaction, in this reaction nitronium ion generated from the nitrating mixture…
Q: Draw the detailed mechanism for the following:
A: In the first step- electrocyclic ring opening occur
Q: The reaction shown here is an example of the Favorskii reaction, which involves an R¯ leaving group…
A: The Favroskii reaction involves the rearrangement of a ketone with the alpha halogen substituent to…
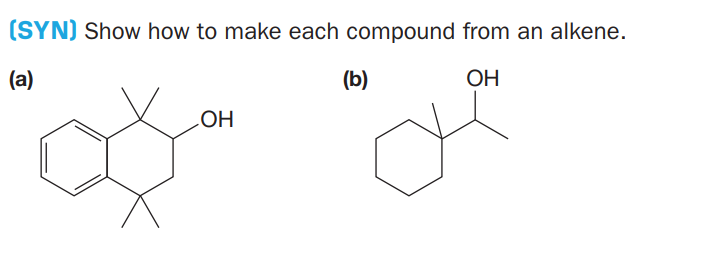
Q: Problem: Show how the following compound can be produced from an alkene. Include the alkene, the…
A: Simmons–Smith reaction: The Simmons–Smith reaction is a cheletropic reaction involving an organozinc…
Q: (SYN) Show how to carry out the transformation at the right, and draw the complete, detailed…
A: The given compound contains 1˚ and 3˚ alcohol group as well as phenol group. Among these, only 1˚…
Q: (SYN) Show how each epoxide can be produced from an alkene. (a) (b) (c)
A: (a)
Q: Identify the product of the following one-step sequence. Note that this is a reaction that you have…
A:
Q: (SYN) Show how to carry out each of the following transformations. (a) (b) Ph. (c) (d) ? ?
A: (a). The acidic anhydride is first hydrolyzed to form 2 moles of benzoic acid which is treated with…
Q: (SYN) Show how to carry out each of the following syntheses by first converting the alcohol into a…
A: (a) Given reaction is In the above reaction, the alcohol can be converted into sulfonyl chloride bu…
Q: Draw the complete, detailed mechanism for the following reaction.
A:
Q: BuLi
A: Organolithium is a reagent that acts as a base or nucleophile in the reaction as per the given…
Q: (SYN) Show how you would carry out each of the following transformations.
A: Electron donating groups activate the benzene ring towards electrophilic substitution reaction and…
Q: (SYN) Show two different syntheses for the compound shown here, one using (CH3)2CULI as a reagent…
A:
Q: mechanism for the following reaction.
A: This is a chlorination reaction, which occurs in presence of light and heat. It contains 3 steps :…
Q: Draw the missing reactant for the following reaction
A: The compound which contains ether and alcohol functional groups attached with the same carbon is…
Q: Draw the complete, detailed mechanism for the following reaction along with the major product. Cl2…
A:
Q: In cach case below select the synthetic procedure/s that could be used to carry out the…
A: Alkene iBH3iiH2O2,OH- antimarkovnikov's addition of H2O (via non-classical carbocation intermediate)…
Q: Complete the following reaction and provide the detailed mechanism H*
A:
Q: (SYN) Propose three different syntheses of the alcohol shown here, each using a different Grignard…
A: Given target compound,
Q: -Br но-
A:
Q: a) Draw a complete, detailed mechanism for the following reaction and label the rate- determining…
A:
Q: Draw the products of attached reaction by following the curved arrows.
A: The products of attached reaction by given the curved arrows can be drawn as
Q: Н.
A: Base accept proton from alpha position to carbonyl compound and form resonating structure ( enolate…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- By following the curved red arrows, draw the product(s) of each of the following reaction steps. Also indicate which species is the electrophile and which is the nucleophile.Draw a mechanism to account for the reaction shown here, which scrambles the isotopic labelingDraw a complete, detailed mechanism for this reaction.

