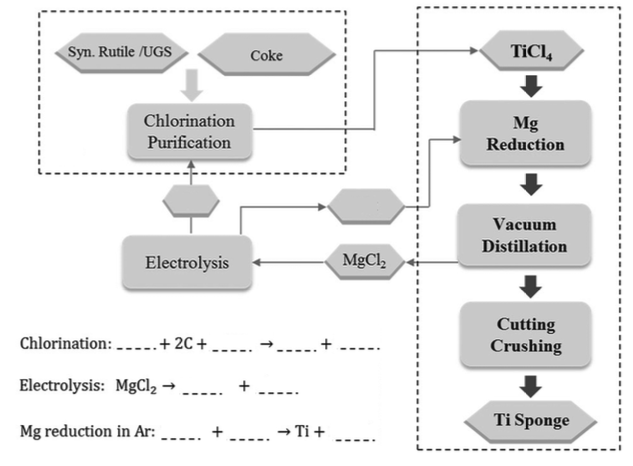
Syn. Rutile /UGS Chlorination Purification Electrolysis Chlorination: ____. + 2C+ Electrolysis: MgCl₂ → Mg reduction in Ar: ‒‒‒- Coke ---.. → Ti + MgCh ---- TICI Mg Reduction Vacuum Distillation Cutting Crushing Ti Sponge
Q: Draw the product of this reaction. Ignore any counter ion or inorganic byproducts. CH3COCI NH₂
A: It is based on the reactivity of amine Here we are required to find the product of the reaction.
Q: Equilibrium 1. write the equilibrium expression for the following reaction 4 NH3 (g) + 7 O2 (g) = 4…
A:
Q: Look at the oxygen atom labeled with the number 9. What functional group is this oxygen atom a part…
A: Functional group is an atom or group of atoms that define the chemical nature and reactivity of a…
Q: how many grams of AsCl3 are produced if 3.9363g of AsF3 are allowed to react with excess C2Cl6.
A: Weight of AsF3 = 3.9363 gm-> C2Cl6 is in excess.
Q: Question 2 A compound A; containing only carbon, hydrogen and oxygen is found to be 78.30% C, 13.04%…
A: Compound A; containing only carbon hydrogen and oxygen% of C = 78.30 % = 78.30 g% of H = 13.04 % =…
Q: 5. The pH of a 0.002 50 mol/L solution of benzoic acid is 3.65. Calculate the K for benzoic acid.
A:
Q: ) What is the pH before any titrant has been added? What is the pH at the midpoint of the titration?…
A: Given,pH vs volume of strong monoprotic base curve Volume of monoprotic acid =20.0 mLconcentration…
Q: Select all the molecule pairs that are enatiomers to eachother A) O E) II... || *** G) None of the…
A: Enantiomers can be defined as one of two different isomers of a compound which is the mirror image…
Q: OH H₂SO4, A Grogand Regt 1) 2 equiv MgBr 2) H₂O
A:
Q: In the process 92235U + 01n → 52137Te + 4097Zr + 201n, what can the two neutrons at the end do?
A: This is a nuclear fission reaction. A nuclear fission reaction takes place when a neutron is…
Q: Based on the experiment described below determine the Michaelis constant of the enzyme involved to…
A: We are given a Lineweaver Burk plot and we have to determine the Michaelis constant of the enzyme…
Q: 10.6 moles of an ideal gas occupy 36.5L at a certain temperature and pressure. What volume would…
A: The ideal gas equation is written as:PV=nRTHere P, V, n are the pressure, volume, and number of…
Q: Naphthalene is a white volatile, solid polycyclic hydrocarbon used in moth and repellents. Looking…
A: For aromaticity, any compound must satisfy these four condition:The molecule must be cyclic.The…
Q: what is the form of this ring structure? the left monosaccarhide name and the right monosaccharide…
A: In this question we are given a disaccharide.We have to answer the form of the ring structure and to…
Q: Suppose you wanted to increase the buffer capacity of the formic acid/sodium formate buffer solution…
A: In this question we have to found what we have to do to increase buffer capacity.
Q: The active ingredient of Benadryl© Chesty Forte Cough Liquid is Guaiphenesin an expectorant drug…
A: It is based on the concept of concentration Here we are requires to find the volume of the cough…
Q: Please predict the product for each of the following reactions. Make sure to clearly indicate the…
A: LiAlH4 acts as hydride ( H-) donor. When LiAlH4 reacts with epoxide, it donates H- to the carbon…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction entropy…
A: The entropy of a reaction (S) is the difference between the sum of the entropies of the products and…
Q: Assign the following alkene molecules as being cis or trans, E or Z isomers.
A: A alkene compound is denoted by two types of process. These two process are…
Q: 7) Which of the following sets of reactions accomplishes the synthesis shown below? H3C-C=CH A. 1.…
A:
Q: Part A Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the…
A: This is an example of electrophilic isomerization of alkene.In this reaction, the double bond of an…
Q: Compound F may be synthesised by the method attached: When 2-chloropropane treated with NaOH and…
A:
Q: 1. Determine the Empirical Formulas from the % Composition data. Compound A: Compound A has a %…
A: Empirical formula of compound A and B is to be determined using the given mass percent composition.
Q: CI H CI
A:
Q: Draw the structures of the conjugate bases of the following acids: COOH | H-C-H NH (a) COO I CH ||…
A: It is based on the concept of acid-Base. Here we are required to find the structure of conjugate…
Q: A medical technician is working with the four samples of radionuclides listed in the table below.…
A: we have to complete the given table with respect to the radioactivity of the given samples
Q: Convert between mass and moles of a compound. Use the molar mass of iron(II) nitrite, 147.9 g/mol,…
A: It is based on the mole concept Here we are required to find the moles of iron nitrite present in…
Q: H₂N Prostaglandin (PGE₂) -OCH3 OH
A: A functional group is a substituent or group of atoms in a molecule that causes the molecule's…
Q: Molecule CH3CH₂CH3 CH3CHO CH3CH₂OH Type Alkane Aldehyde Alcohol Boiling point (°C) -42 +21 +78
A:
Q: 4. a. The ¹H NMR spectrum of a compound with the molecular formula C-H₁0O2 is shown below (integral…
A: The concept of this question is based on organic spectroscopy which includes HNMR, C13 NMR, UV and…
Q: Choose the resonance structure that results from the arrow pushing scheme below:
A:
Q: Which of the following molecules produced the 1H NMR spectrum shown below?
A: Chemical shifts from the given 1H-NMR spectrum.1. 7.2 ppm (2H, d)2. 7.4 ppm (2H, d)3. 5.1 ppm (1H,…
Q: A solution of ethylamine (CH3CH₂NH2; K₁ = 4.30 x 10-4) produces a pH = 12.67. What is the %…
A:
Q: A 1.25 gram sample of solid calcium carbide (CaC2) reacts with water to produce acetylene gas (C2H2)…
A:
Q: Hello, I have a very important question to ask you, if you can help me I would be very happy. My…
A: A question based on IR spectroscopy of organic compounds. A comparative IR spectra of 2 samples is…
Q: 1. A student prepares a 100 mL aq containing a small amount of (NH4)2SO4 and a second 100 mL…
A: 1) Given question related to mixing of two inorganic salt solutions. “Since you have posted multiple…
Q: Hi, I need to create a formula utilizing the tols that are at the bottom of the picture. In order to…
A: K2CrO4 on dissociationK2CrO4 <===> 2K+ + CrO4-Molarity of K2CrO4 = 0.122 M = 0.122 mol/L
Q: When ammonia (NH3) is allowed to flow past copper(II) oxide at a high temperature nitrogen gas (N2)…
A: From the given equation and given data we have to estimate the amount of nitrogen .
Q: What is the pH of a 0.052 M sodium acetate (NaC2H302) solution? (Ka= 1.8x10-5) for C2H4O2 13:58
A: The concentration of sodium acetate is .The Ka for acetic acid is .To find the pH of sodium acetate.
Q: In the context of chemical reactions, explain the concept of kinetic versus thermodynamic control…
A: The concept of kinetic versus thermodynamic control in chemical reactions explores the factors that…
Q: НО. OH Br Br
A: To convert 2,3-dimethoxy-1,5-diol into 1,4-dibromo-2,3-dimethoxybutane, several steps are involved.…
Q: What type (redox, acid-base, precipitation) are the reactions below? For the redox reactions,…
A: The reaction in which both oxidation as well as reduction takes place simultaneously is called…
Q: Consider a chemical reaction where a gaseous reactant A reacts with a solid reactant B to form a…
A: In this response, we explore the factors influencing the reaction rate of a chemical reaction…
Q: Calculate the enthalpy reaction for CO(g)+2H2->CH3OH(g)
A:
Q: 6910m to mile 79.0 F to C -15Degrees F to K please show how to do these using map solution…
A: Meter (m) to miles (mile): To convert meters to miles, conversion to kilometers is…
Q: EtO. JoJooziom O PPh3
A: To draw the product and mechanism of the given organic reaction.
Q: 1. Circle and name 4 (and only four) of the functional groups in the molecule below. You must do…
A: The given structure is shown belowWe have to identify and name four functional groups present on…
Q: Arrange the following in order of increasing vapour pressure at room temperature. CH3CH₂OCH₂CH3…
A: Vapour pressure is a pressure exerted by a vapour on the liquid surface.When the vapour pressure…
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, she fills…
A: The relationship between ΔG,ΔH & ΔS is ΔG=ΔH −TΔS For spontaneous reactions, ΔG < 0
Q: List the given compounds in order of increasing acidity (lowest first). Give a reason for your…
A: In chemistry, acid means the compound which is able to donate H+ ions.Now which compound donate H+…
Complete the reactions below
Step by step
Solved in 3 steps

- In Fehling's reaction heat is ?a)Reducerb) Catalyserc)Substrate explain the answerIn the video "Sacrificial Anode Materials Demo", which among Zn, Al, Mg served as as i) sacrificial anodes? and ii) as noble coatings?TRUE OR FALSE. 1The color of permanganate solution renders impossible accurate readings at the lower meniscus.2Permanganate-oxalate reactions are run at reduced temperatures.3The permanganate method is always carried out in a highly acidic medium.4Sulfuric acid in permanganate titration prevents the formation of reduction products.5REDOX reaction involves the sharing of electron/s from one species to another.
- For Redox Analysis of Iron... ~0.02 M of potassium dichromate is prepared as titrant for dried unknown. ~.3 g of sample, 50ml of 6M HCl, 20mL SnCl2, 10 mL of saturated HgCl2 solution, 60 mL of 3M H2SO4, 15mL of concentrated H3PO4, 100mL DI water. HCl (+ heat in fumehood until samples dissolves), SnCl2 (added with pipet until solution changes from yellow to colorless/light green with 3 drops in excess), HgCl2 (after cooling), H2SO4, H3PO4, DI Water, is added to the sample solution with 8 drops of diphenylamine sulfonate indicator then titrated with the potassium dichromate to a violet blue endpoint. 4. Why is it necessary to carry out the reduction of iron and then the titration, before going on to the next sample 5. If you look carefully, there are pieces of tin metal on the bottom of the SnCl2 reagent solution. Why is it there?For Redox Analysis of Iron... ~0.02 M of potassium dichromate is prepared as titrant for dried unknown. ~.3 g of sample, 50ml of 6M HCl, 20mL SnCl2, 10 mL of saturated HgCl2 solution, 60 mL of 3M H2SO4, 15mL of concentrated H3PO4, 100mL DI water. HCl (+ heat in fumehood until samples dissolves), SnCl2 (added with pipet until solution changes from yellow to colorless/light green with 3 drops in excess), HgCl2 (after cooling), H2SO4, H3PO4, DI Water, is added to the sample solution with 8 drops of diphenylamine sulfonate indicator then titrated with the potassium dichromate to a violet blue endpoint. 1. What is the purpose of the phosphoric acid in this experiment? Please be specific. 2. How does the Diphenylamine sulfonate indicator work? What chemical reaction causes the color change? How is an indicator chosen in a redox reaction?For Redox Analysis of Iron... ~0.02 M of potassium dichromate is prepared as titrant for dried unknown. ~.3 g of sample, 50ml of 6M HCl, 20mL SnCl2, 10 mL of saturated HgCl2 solution, 60 mL of 3M H2SO4, 15mL of concentrated H3PO4, 100mL DI water. HCl (+ heat in fumehood until samples dissolves), SnCl2 (added with pipet until solution changes from yellow to colorless/light green with 3 drops in excess), HgCl2 (after cooling), H2SO4, H3PO4, DI Water, is added to the sample solution with 8 drops of diphenylamine sulfonate indicator then titrated with the potassium dichromate to a violet blue endpoint. 3. How would you prepare a complete anlaysis procedure using KMnO4 as the oxidant instead of K2CrO7? Include sample and solution preparation, approximate weights of samples and reagents, procedure and chemical reactions. In particular what is the purpose of the Zimmerman Reinhardt Reagent?
- Many applications of electrochemistry have been in chemical analysis.Suppose a Fe|Fe2+ concentration cell is set-up in which the [Fe2+] = 1.0 x10¯2 M in one compartment but the concentration in the othercompartment is unknown. The Ecell was measured at 0.024V when thehalf-cells were connected.(i) Without doing a numerical calculation, is the unknown more orless concentrated than the 1.0 x 10¯2 M solution? Justify youranswerMany applications of electrochemistry have been in chemical analysis.Suppose a Fe|Fe2+ concentration cell is set-up in which the [Fe2+] = 1.0 x10¯2 M in one compartment but the concentration in the othercompartment is unknown. The Ecell was measured at 0.024V when thehalf-cells were connected.(i) Without doing a numerical calculation, is the unknown more orless concentrated than the 1.0 x 10¯2 M solution? Justify youranswer(ii) Calculate the unknown concentration.(iii) Does your calculated answer agree with your original answer?Why?(c)Ions that react with Ag+ like SCN- can be determined electrogravimetricallyby deposition on a silver working anode:Ag(s) + SCN- ≤ AgSCN(s) + eA silver anode with initial mass of 12.4730 g was used to electrolyse 85.00 mL of asolution of 0.0245 M KSCN. At the end of the experiment, the anode weighed12.5767 g. How long did the experiment take, if a current of 100 mA was used?
- What is chemical reduction (only reduction, not redox) and chemical dechlorination in chemical treatment of hazardous waste? What are their processes?A 100 ml sample of pond water was analyzed using Winkler method to determine its oxygen demand. It took 32.4 ml of 0.0100 M Na2S2O3 to react with Iodine. Calculate the oxygen for this pond water.Addition of both Bi3+ and Sn2+ by the method by studying the cathode potential to be made for molding of those in a buffered solution to 0.100 M and pH 1.5. Find the concentration of the more difficult-to-make species or the more easily-reducible species. Sn2+ + 2 e- ↔ Sn (k) E0 = - 0.136 V BiO+ + 2 H+ + 3 e- ↔ Bi (k) E0 = 0.320 V A. 3.2x10-2MB. 2.33 x 10-22MC. 1x10-6MD. 6.2x10-22M



