Q: a) Calculate the pH after 0.0100 mol of H+ ions are added to 0.100L of buffer containing 1.00M…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: Consider the reaction. 2A(g) B(g) Kp = 2.48 x 10-5 at 500 K If a sample of A(g) at 4.50 atm is…
A: P(A) = 4.50 atmT = 500 KKp = 2.4810-5Pressure of B at equilibrium =?
Q: lease be very confident in your answer
A: Hybridization is mixing and recasting of atomic orbitals to form the new orbitals with equal energy…
Q: Which of the following compounds would have the highest Rr? The lowest Rf? - CH 3 OH వి CH3
A: The objective of the question is to find the compounds from the given option that would have the…
Q: Which of the following molecules would exhibit the property listed: CH,CH,COCH,CH, A.…
A: Given are organic compounds. Compound A is alkane.Compound B ester. Compound C is ketone.Compound D…
Q: Provide the IUPAC name of the product, if any forms from the reaction: in name ex…
A: Carbonyl compounds can be reduced into alcohols. The aldehyde reduces into primary alcohol and the…
Q: Which Newman projection represents correct conformation for the following compound: HO H A HO CH3 H…
A: In order to better visualize different conformations of a molecule, it is convenient to use a…
Q: What is the configuration of the chiral center of the following molecule? OR ○ S Meso OH
A: According to Cahn-Ingold-Prelog rule-1) More atomic number having more priority.2) If first atom is…
Q: Identify how many C (CARBON) atoms have sp hybridization for the compound below: :0: N: A B C 0…
A: Hybridization is defined as the intermixing of atomic orbitals which results in the formation of new…
Q: For the compound below please identify the position of equilibrium (which chair conformation is more…
A: Given is structure of (1R,2S)-1,2-dichlorocyclohexane.The more stable chair conformation is one in…
Q: ∆Gº for the reaction, 2HBr(g)⇄ H2(g)+Br2(g), is 33.3 kJ.
A: The objective of the question is to understand the implications of the given standard Gibbs free…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. OH Na2Cr2O7 H2SO4, H2O Q
A:
Q: What type of linkage is between these monosaccharide derivatives? CH3 C=0 CH₂OH H NH H OH H OH H H H…
A:
Q: Give detailed Solution with explanation needed....don't give Handwritten answer
A: Option E is correctExplanation:calculate the total number of valence electrons in ClO₃⁻ Chlorine…
Q: At equilibrium, the concentrations in this system were found to be [N_{2}] = [O_{2}] = 0.3Mand[NO] =…
A: The objective of the question is to find the final concentration of NO after equilibrium is…
Q: Draw the starting structure that would yield this product under these conditions. 1. Br2 (1 equiv),…
A: This is an example of Hell Volhard Zelinsky (HVZ) Reaction
Q: Consider the two containers below. How does the pressure in these containers compare? A B T = 200K…
A: We can compare the pressures in the two containers, we can use the ideal gas law, which states:…
Q: Highlight the most acidic of the H atoms shown in each molecule below.
A: Given are organic compounds.Acidity is defined as the ability of a molecule to release protons.If a…
Q: May you explain the H NMR and the C NMR please. If my structure is right please. Thanks
A: Your structure is absolutely correct.!! Congratulation!! The NMR and the FTIR have been analyzed…
Q: The Knoevenagel condensation reaction is a variant of the aldol reaction. A major drawback of the…
A: The Knoevenagel condensation reaction is a version of the aldol reaction in which an aldehyde or…
Q: raw the product of this reaction. Ignore inorganic byproducts. Br2 (2 equiv) Q
A: The objective of the question is to find the product of the given reaction in which substituted…
Q: 2. Below is the structure of the drug Odevixebat, which is used for treating decreased bile…
A: We have to choose the functional groups.
Q: 11. eauxture. Important: remember to riase and wipe off the mixtures so as not to contaminate them.…
A: The objective of the question is to draw the structures of methyl stearate and propylene glycol and…
Q: How many grams of lead azide will dissolve in 500 mL of 100 M Pb(NO3)2 solution? Ksp=3.4×10^-14
A: The objective of the question is to calculate the amount of lead azide that will dissolve in a given…
Q: The number of moles of CH4 in 6.00 q is
A: Given: Mass of CH4 = 6.00 gRequired: Moles of CH4 = ?
Q: Please show the major products with stereochemistry where needed
A: A reaction is a process by which a reactant is converted into a product. In this process, functional…
Q: Reaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0 °C, followed by treatment…
A: The objective of the question is to understand the chemical reaction that occurs when p-nitroaniline…
Q: Nitesh
A: The objective of this question is to calculate the change in Gibbs free energy (ΔG) for a reaction…
Q: Macmillan Learning Pressure (atm) Mass (g) Syringe Volume (mL) Temperature (°C) 1.05 0.127 9.10 21.5…
A: Generally, the volume of a gas and the number of moles are directly proportional to each other. This…
Q: Draw the correct aromatic organic product for the following reaction. (If no reaction, draw the…
A: Isopropyl benzene contains one benzylic hydrogen. So it will undergo oxidation reaction when treated…
Q: To synthesize the heavier transuranium elements, a nucleus must be bombarded with a relatively large…
A: Answer:When the nucleus of an atom is changed by the bombardment of smaller particles then such…
Q: Do not use chat gpt
A: Please find the attachment for detailed solution.Explanation:Please find the attachment for detailed…
Q: Draw the product of this reaction. Ignore inorganic byproducts. Br2 (2 equiv) Drawing Q
A: In the reaction given above, reaction between alkyne and bromine (Br2) is given. In this reaction,…
Q: I and II are: I FL constitutional isomers. O enantiomers. O identical. O diastereomers. O not…
A: Isomers are compounds that have same molecular formula but different structural formula.…
Q: 4. 915) A saturated solution of iodine in water contains 0.33 g iodine /L. In a potassium iodide…
A: The equilibrium constant (K) of a reaction is used to show the relationship between the…
Q: The ionic radii of element E and a different metallic element, M, are shown in the following table:…
A: The objective of the question is to determine whether the lattice energy of MO would be less than,…
Q: 8.31d KMnO4, NaOH Cold ? Modify the given carbon skeleton to draw th enantiomers. Note: you can…
A: Structure of product of the given reaction is to be determined.
Q: Question 6 Propose a chemical structure for the name below. Make sure to clearly define the…
A: From the given name, dien-one means there are two C=C and one C=O. 2,5-dien-1-one means there are…
Q: The equilibrium constant Kp = (9.180x10^-1) for the following reaction in the ga phase at a given…
A: Given:Reaction: Equilibrium constant, Kp = 9.180x10-1 =0.918Initial pressure of H2O= 3.06x10-1 bar…
Q: 11. How many electrons are in the p-orbital of the O atom in the following molecule? A. O B. 1 C. 2…
A: During orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals known…
Q: A Ca(OH)2 tablet has 360 grams of Calcium. What is the mass of tablet in milligrams? My Ca=40.078…
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: For the compound below please choose the correct set of chair and flipped chair conformations: α…
A: The cyclohexyl ring goes into rapid inversion which is also called topomerism, ring reversal or…
Q: Provide the IUPAC name of the product, if any forms from the reaction: in name ex…
A: The given reaction is CH3-CH2-CH2-C-H + H2 Pt. This is a hydrogenation reaction, where hydrogen is…
Q: I am needing help with a through c please.
A: Detailed solution is provided below.Explanation:
Q: • Identify the type of hybridization (sp, sp², sp³) for all N (NITROGEN) atoms of the following…
A: The process of intermixing of atomic orbitals that are similar in energy, orientation and shape to…
Q: Retrosynthesis Practice jay th Ph Aldol Condensation ? wittig ? ? + טרי
A: We have to predict the starting materials using retrosynthesis.
Q: Please don't provide handwritten solution ...
A: The major product of the reaction is product-A.Explanation:
Q: please help this is my 4th time posting this and i havent gotten a correct answer. Please actually…
A: Please find the attached image.Details of axesX-axis: 1/S (reciprocal of the substrate…
Q: Indicate whether or not the hydrocarbons on the list react with 1) ammoniacal solution of AgNO3 or…
A: Out of the listed hydrocarbons, only alkynes with a terminal alkyne group (-C≡CH) react with…
Q: Propose a synthesis for lactone B starting from hydroxy-aldehyde A. Show the required reagents and…
A: The objective of this question is to draw a multi-step synthesis pathway to achieve a target…
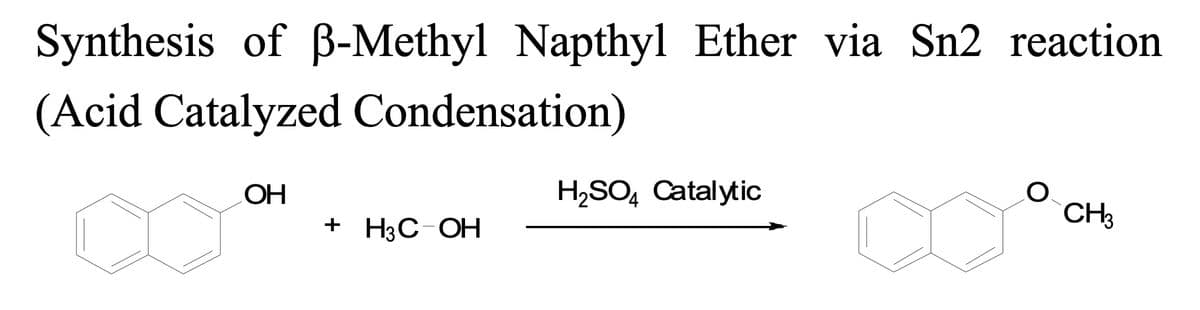
In the Acid Catalyzed Condensation Synthesis of Methyl beta- Napthyl Ether experiment, the following reaction is performed. Draw a detailed arrow pushing mechanism for this reaction?
Step by step
Solved in 1 steps with 1 images

- Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answer) Which of the following scientific statements are true and which are false? 1- [ ] E1 describes an elimination reaction in which the rate-determining step does not involve the base. 2- [ ] Rate of formation of carbocations follow the order 3˚ > 2˚> 1˚> CH3+ 3- [ ] Cyclohexanol is oxidized to ketone using Jone's reagent. 4- [ ] Symmetric ethers are generally synthesized by dehydration of alcohols. 5- [ ] The SN2 reaction occurs with inversion of configuration.The following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH. Explain. CH3CO2CH3 > CH3CO2CH2CH3 > CH3CO2CH(CH3)2 > CH3CO2C(CH3)3
- The formation of Br2 from NBS first involves the reaction of NBS with HBr to form an iminol intermediate and molecular bromine. The intermediate then undergoes acid-catalyzed tautomerism to form succinimide, the byproduct of the reaction. Propose a curved-arrow mechanism for the conversion of NBS into succinimide that also accounts for the formation of Br2.Explain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.Show the reaction mechanism for the formation of diethyl ether from ethanol in concentrated sulfuric acid: a. SN2 b. SN1
- Arrange the following compounds in their ease of undergoing an SN2 reaction: a. CH3CH2CH2-Cl b. (CH3)2CH-Br c. (CH3)3C-Cl d. CH3-Brn-Pentanol (CH3CH2CH2CH2CH2OH) and 2-methylbutan-2-ol (CH3CH2C(CH3)2OH) are converted to their corresponding alkyl chorides on being reacted with hydrogen chloride. (a) Write out an equation for each reaction (b) Assign each the appropriate symbol (SN1 or SN2) (c) Write a suitable mechanism for each reactionShow the relationship between E2 Reactions and Alkyne Synthesis ?
- Explain the following result. Although alkenes are generally more reactive than alkynes toward electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.Devise a synthesis of (E)-1-phenylhex-1-ene (CH3CH2CH2CH2CH = CHPh) using hydrocarbons having ≤ 6 C’s and a Suzuki reaction as one of the steps.Give at least two methods for the synthesis of 2-methylpropane. What happens if it is treated with: a) HNO3 dilute, t °C, p; b) Br2/t °C; c) H2SO4 conc. In the case of the reaction with HNO3 dilute, t °C, p the mechanism is required.



