Table 1: Properties of Zinc and Sulphur Element Zinc Sulphur Number of moles, mole 0.365 Mass, g 23.3 16.3 Number of atoms, atom 3.06 x 1023 Dmitri Mendeleev found elements known as Element X and Element Y. He found that element X and element Y are one of the transition element and non-metallic element respectively in Periodic Table. Identify Element X and Element Y according to their groups from Periodic Table. I Using the information from Table 1, i) Calculate the mass of atom X by assuming the number of mole similar with Zinc. Calculate the number of atom Y by assuming the mass of atom Y is similar with 16.3 g of sulphur. ii)
Table 1: Properties of Zinc and Sulphur Element Zinc Sulphur Number of moles, mole 0.365 Mass, g 23.3 16.3 Number of atoms, atom 3.06 x 1023 Dmitri Mendeleev found elements known as Element X and Element Y. He found that element X and element Y are one of the transition element and non-metallic element respectively in Periodic Table. Identify Element X and Element Y according to their groups from Periodic Table. I Using the information from Table 1, i) Calculate the mass of atom X by assuming the number of mole similar with Zinc. Calculate the number of atom Y by assuming the mass of atom Y is similar with 16.3 g of sulphur. ii)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 111CP
Related questions
Question
100%
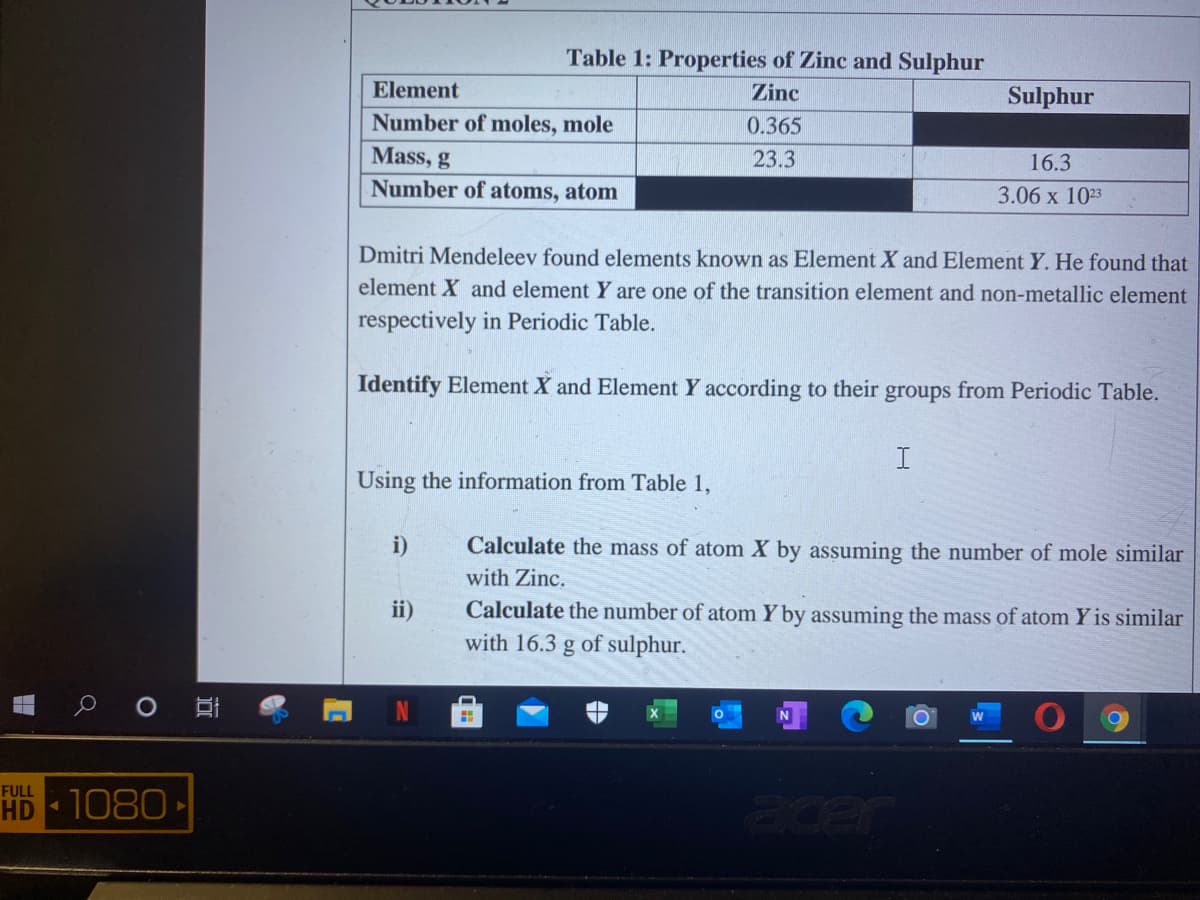
Transcribed Image Text:Table 1: Properties of Zinc and Sulphur
Element
Zinc
Sulphur
Number of moles, mole
0.365
Mass, g
23.3
16.3
Number of atoms, atom
3.06 х 103
Dmitri Mendeleev found elements known as Element X and Element Y. He found that
element X and element Y are one of the transition element and non-metallic element
respectively in Periodic Table.
Identify Element X and Element Y according to their groups from Periodic Table.
Using the information from Table 1,
i)
Calculate the mass of atom X by assuming the number of mole similar
with Zinc.
Calculate the number of atom Y by assuming the mass of atom Y is similar
with 16.3 g of sulphur.
ii)
FULL
HD 1080 ►
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning