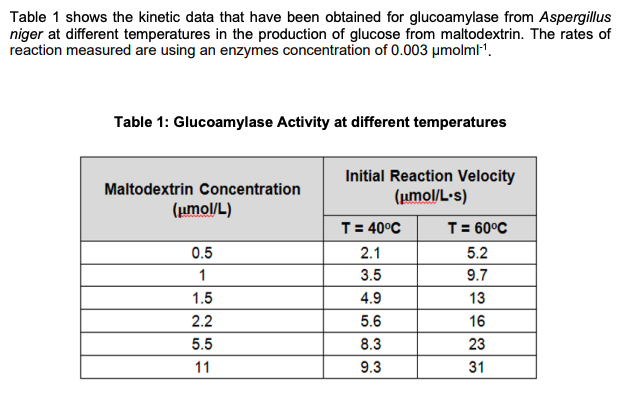
Table 1 shows the kinetic data that have been obtained for glucoamylase from Aspergillus niger at different temperatures in the production of glucose from maltodextrin. The rates of reaction measured are using an enzymes concentration of 0.003 μmolml-¹. Table 1: Glucoamylase Activity at different temperatures Maltodextrin Concentration (μmol/L) 0.5 1 1.5 2.2 5.5 11 Initial Reaction Velocity (μmol/L.s) T = 40°C 2.1 3.5 4.9 5.6 8.3 9.3 589 T = 60°C 5.2 9.7 13 16 23 31
Q: 4A. Write out the chemical equations (structures are not necessary) for the two steps in the…
A: Amino acyl tRNA synthetase is the enzyme that catalyzes the transfer of amino acid to the specific…
Q: Which of the following is incorrect about chymotrypsin, trypsin, and elastase? a. They catalyze the…
A: Introduction: Chymotrypsin is a digestive enzyme present in our bodies which is secreted by the…
Q: Sphingolipids serve what function in biological systems? A) energy storage B) cell membrane…
A: Sphingolipids belong to a different category of lipids. Sphingosine, 18-carbon amino alcohol,…
Q: Enzyme kinetics uses math to quantify all of the following, except: a. An enzyme's substrate…
A: Enzymes are biocatalysts that facilitate the biochemical reactions by decreasing the activation…
Q: escribe using two named examples how recombinant genes have been used to produce commercially…
A: Introduction: Recombinant DNA technology is a molecular biology methodology involving DNA production…
Q: Which of the following is incorrect about the rate of an enzyme-catalyzed reaction? O a. It does not…
A: The rate of an enzyme-catalyzed reaction depends on certain factors and one of them is concentration…
Q: In the net reaction of glycolysis, which of the following is a term on the product side? a. 2 NADH…
A: Glycolysis is a metabolic pathway during which glucose molecule splits into pyruvate molecules…
Q: Fatty acids are ______ at physiological pH. a. charged b. uncharged c. very cheap d. hydrophobic e.…
A: Fatty acids are important micromolecules which combine together to form lipids in plants, animals…
Q: Macronutrients provide us the energy to live, move, and function. Evaluate the sentence above,…
A: Bioenergetics refers to the study of living system and it's energy transformations within and among…
Q: 9. Starting as a polysaccharide in a spaghetti dinner, trace the path that energy and biomass make…
A: We all know that both mass and energy is conserved. So whatever the mass of the polysaccharide was…
Q: Cholesterol-lipid microdomains are often referred to as membrane rafts.
A: True.
Q: Experiment #1: The Effect of Concentration on Reaction Rate Number of Drops of Enzyme 2 4 6 Reaction…
A: During a biochemical reaction, substrate molecules get converted into product according to the…
Q: Match the following terms with the appropriate type of movement across a cell diffusion active…
A: Molecules are transported across the plasma membrane through mechanisms such as passive transport…
Q: The structure shown below is a * H₂C-O HC-0 H₂C-O Fatty acid O Fat O Cholesterol O Carbohydrates MA…
A: Biological lipids are a chemically diverse class of organic compounds that are either insoluble or…
Q: wre these products right? please clearly show what the products will be
A: Waxes are simple lipids composed of fatty acid esters and long-chain alcohols. Beeswax, lanolin are…
Q: COO I CH₂ HC-COO HO-CH COO COO™ 1 CH₂ CH₂ COO 11 C-H COO
A: NOTE:- kindly repost 1st reaction ,as the image is blur, thus couldn't see the picture clearly.…
Q: We are outnumbered by the bacteria in our colon. Why don't they typically make us sick? a) They…
A: The human microbiome helps to maintain the intestine linings and prevents the growth of pathogenic…
Q: Family B Generation 1 Female 1 Absent Male 1 Morton's Toe Generation 2 Female 1 Morton's Toe married…
A: The pedigree chart will clearly show the occurrence of any trait or disease in different…
Q: Number 1 question answer. Thank you. no need for a long explanation.
A: Metabolism is a process that involves various enzyme catalyzed biochemical reactions, which…
Q: 10. You have developed a transcription assay that recapitulates the in vivo regulation of the…
A: As given in the question, two additional transcription factors (Click and Clack) were identified in…
Q: Suppose a gene has the sequence ATGGGTTATCGCGAGTAC. A point mutation changes the gene to read…
A: Gene is a segment of DNA that codes for a protein. It transcribes to mRNA which then translates to…
Q: what is the N and C terminal amino acid residues of
A: A protein is a sequence of amino acids when we draw the structure of peptide or protein…
Q: What is the structure of the steroid nucleus found on all sterols? A. It consists of five fused…
A: The structure of the steroid nucleus found on all sterols is described in next step.
Q: which step of the polymerase chain reaction takes place at 98 degrees?
A: It is an enzymatic process by which specific segments of DNA can be amplified by using a primer,…
Q: Draw the condensed structural formulas for the products of the reaction of which is catalyzed by…
A: Enzymes are the biocatalysts that speed up biochemical reactions. These are generally proteins. AST…
Q: Need assistance
A: Hi, thank you for posting the question on Bartleby. As per the guidelines, we can answer only the…
Q: How many net molecules of nucleoside triphosphate (ATP and equivalent molecules) are produced by…
A: As you have posted multiple unrelated questions we are supposed to answer only the first question…
Q: Which of the following is incorrect when comparing myoglobin and hemoglobin? a. Myoglobin and…
A: Hemoglobin and myoglobin are both oxygen-binding proteins. Hemoglobin transports oxygen from the…
Q: Which of the following is an achiral amino acid? alanine arginine proline glycine Previous Page Next…
A: Q1 two categories represent a classification of all lipids based on their molecular structure is…
Q: Choose 3 words and make them a sentence. Repeat this structure 2 times please
A: Proteins having additional sugar chains are found in glycoproteins. Lipids with linked carbohydrates…
Q: Which properties of a fatty acyl chain cause the Tm to be lower? 1. shorter hydrocarbon chain length…
A: Fatty acids are the simplest form of lipids, it can either be saturated or unsaturated and can be…
Q: ... A(n) binds to a receptor and elicits a biological effect; while a(n) binds to a receptor but…
A: Binding of certain molecules at their receptors involves the eliciting of response that can be…
Q: Which of the following is/are true about the primary structure of a protein? It does not include…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: 1. Assign IUPAC names to each of the following small peptides. a. Gly-Ala-Leu b. Gly-Tyr-Ser-Ser C.…
A: As a general rule of writing peptides, the N-terminal is at the left and the C terminal is at the…
Q: Question 21 An example of an antibiotic useful in treating tuberculosis would be: 01) penicillin O2)…
A: Antibiotics are substances that can control or kill bacterial growth. They are useful in treating…
Q: How many bypassed reactions does gluconeogenesis have? 10 2 3 6 1
A: The synthesis of glucose from the non-carbohydrate precursors is called gluconeogenesis. Major…
Q: Which of the following sets corresponds to nonpolar amino acids? a. Met, Ile, Glu b. Ala, Leu, Trp…
A: Amino acids are α-amino carboxylic acids that are the basic building blocks of proteins. Amino acids…
Q: Which of the following is incorrect? a. The overall shape of an entire polypeptide including all its…
A: Proteins are polypeptides or polymers of amino acids. Amino acids are the building blocks of the…
Q: Which of the following is/are epimer/s of glucose?* A. Galactose only B. Mannose only C. Fructose…
A: Carbohydrates are polyhydroxy aldehydes or polyhydroxy ketones, or compounds that can be hydrolyzed…
Q: What metabolic substrate is produced from the carbon atoms of each of the following amino…
A: Amino acids are organic molecules which act as the building blocks of proteins. During the…
Q: Which of the following is incorrect about the peptide Val-Leu-Ala-Ser-Gly? It has a net charge of…
A: H3+N-Val-Leu-Ala-Ser-Gly-COO- a) We have 1 positive charge at the N-terminal and one negative charge…
Q: The sequences of four DNA molecules are given bel ii. TTTCCCGGGAAA AAAGGGCCCTTT iv. GCCGGATCCGGC…
A: DNA molecules are the nucleic acids that contain sugar, phosphate and nitrogenous base. Sugar is…
Q: Which of the following is incorrect? a. Without an enzyme, reaction rate can be increased by…
A: Enzymes are the catalysts of biochemical reactions that increase the rate of reaction. Enzymes have…
Q: can be broken down by amylase.* A. glycogen B. amylopectin C. catalase D. amylase
A: Glycogen is a homopolysaccharide composed of glucose units that are linked through glycosidic…
Q: An enzyme catalyzes a reaction at a velocity of 20 micromole/min when the concentration of substrate…
A: The name of this metabolic pathway is derived from the citric acid (a tricarboxylic acid, often…
Q: How does the hormone get from its secretory gland to its target tissue? • Are steroid hormones…
A: Since there are multiple questions in the image, I will explain the entire signaling cascade.…
Q: The activity of an enzyme is studied with and without an inhibitor. Specifically, the initial rates…
A: With the following details provided you are first supposed to plot a graph between substrate…
Q: List ALL of the substrate-level phosphorylation steps in cellular respiration and the enzymes that…
A: Substrate level phosphorylation is the metabolic process by which nucleotide tri phosphate ATP or…
Q: Which of the following is incorrect about the G protein signaling pathways? a. The binding of the…
A: The guanine nucleotide-binding protein (G-protein) cell signaling pathway is involved in many…
Q: Which of the following statements concerning oxidative phosphorylation is false? Group of answer…
A: The free energy released from oxidation of glucose to carbon-dioxide is stored in the form of…

Step by step
Solved in 2 steps with 1 images

- Table 1 shows the kinetic data that have been obtained for glucoamylase from Aspergillus niger at different temperatures in the production of glucose from maltodextrin. The rates of reaction measured are using an enzymes concentration of 0.003 μmolml-1. Outline and describe the reaction scheme of the production of glucose from maltodextrin by using glucoamylase.Calculate the mass of invertase (in mg) and concentration of invertase (in mM) contained in a 25.0mL sample of yeast extract that has 3,000 total units of activity, assuming that pure invertase has a specific activity of 1,000 units/mg with a mass of 270kD.For the following aspartase reaction in the presence of the inhibitor hydroxymethylaspartate, determine Km and whether the inhibition is competitive or noncompetitive. You have to plot thegraph on the graph paper and also by using excel.[S] V, No Inhibitor V, Inhibitor Present(molarity) (arbitrary units) (same arbitrary units) 1 x 10-4 0.026 0.0105 x 10-4 0.092 0.0401.5 x 10-3 0.136 0.0862.5 x 10-3 0.150 0.1205 x 10-3 0.165 0.142
- Conversion of F1,6BP to GA3P and DHAP by aldolase is striking in that it is strongly unfavorable when all species are at 1 M (∆G°’ = +22.8 kJ/mol) but favorable when the species are at their physiological conditions (∆G = -5.9 kJ/mol). Based on ∆G°’ and ∆G of aldolase and TIM, and using a reasonable physiological value of 15 mM for the concentration of F1,6BP, calculate the concentrations of GA3P and DHAP under physiological conditions (use T = 37°C). Why is the reaction so much more favorable under these conditions than when all species are at 1 M?The standard free energy change for the reaction catalyzed by phosphoglucomutase is -7.1kJ/mol, (a) calculate ΔG at 37°C when the concentration of glucose-1-phosphate is 1-mM and the concentration of glucose-6-phosphate is 25-mM, (b) Is the reaction spontaneous under these conditions?The KM for the reaction of chymotrypsin with N-acetylvaline ethyl ester is 8.8 × 10−2 M, and the KM for the reaction of chymotrypsin with N-acetyltyrosine ethyl ester is 6.6 × 10−4 M. (a) Which substrate has the higher apparent affi nity for the enzyme? (b) Which substrate is likely to give a higher value for Vmax?
- Calculate the standard free-energy change of the reaction catalyzed by theenzyme phosphoglucomutase, given that, starting with 20 mM glucose 1-phosphate and no glucose 6- phosphate, the final equilibrium mixture at 25 °C and pH 7.0 contains 1.0 mM glucose 1-phosphate and 19 mM glucose 6-phosphate. Does the reaction in the direction of glucose 6-phosphate formation proceed with a loss or a gain of free energy?The following initial-rate data were obtained on the rate of binding of glucose w ith the enzyme hexokinase (obtained from yeast) present at a concentration of 1.34 mmol dm-3. What is (a) the order of reaction with respect to glucose. (b) the rate constant?[C6H12O6]/(mmol dm-3) 1.00 1.54 3.12 4.02vo/(mol dm-3 s-1) 5.0 7.6 15.5 20.0The standard reduction potential for ubiquione (A or coenzyme Q) is .045 V, and the standard reduciton potential (E) for FAD is -0.219 V. Using these values, show that the oxidation for FADH2 by ubiquinone theoretically liberates enough energy to drive the synthesis of ATP. Faraday constant =96.48KJ/Vol delta G' standard for ATP Synthesis is +30.5 KJ/mol R=8.314 J/mol K=1.987 cal/mol K
- The enzyme β-methylaspartase catalyzes the deamination of β-methylaspartate. For this aspartate reaction in the presence of the inhibitor hydroxymethylaspartate (3.8 M), determine KM and whether the inhibition is competitive or noncompetitive (KI = 1.0 M). [S], M V w/o inhibitor, M/s V w/ inhibitor, M/s 1x10-4 0.0259 0.0098 5x10-4 0.0917 0.040 1.5x10-3 0.136 0.086 2.5x10-3 0.150 0.120 5x10-3 0.165 0.142 In the ABSENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= __________ (1[S])(1[S]) + __________, and the KM is __________ M. In the PRESENCE of inhibitor: The Lineweaver-Burke equation is 1V=1V= ____________ (1[S])(1[S]) + ___________, and the KM is ___________ M. The type of inhibition is ____________. Round-off all answers to two (2) significant figures.The Keq (25C) of the reaction below is 635.67. Fructose 1,6-biphosphate <-->fructose -6-phosphate + Pi. a) What is the standard Gibbs free energy change for this reaction? b) if the concentrationof fructose 1,6 biphosphate is adjusted to 0.85 M and that of fructose 6 phosphate and phosphate adjusted to 0.055 M, what is the actual free energy changeThe enzyme, fumarate, has the following kinetic constants: k 1 k 2 k -1 where k 1 = 10 9 M -1 s -1 k -1 =4.4 x 10 4 s -1 k 2 = 10 3 s -1 a. What is the value of the Michaelis constant for this enzyme? b. At an enzyme concentration of 10 -6 M, what will be the initial rate of product"

