THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THC
THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THC
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter16: Amines
Section: Chapter Questions
Problem 16.38P
Related questions
Question
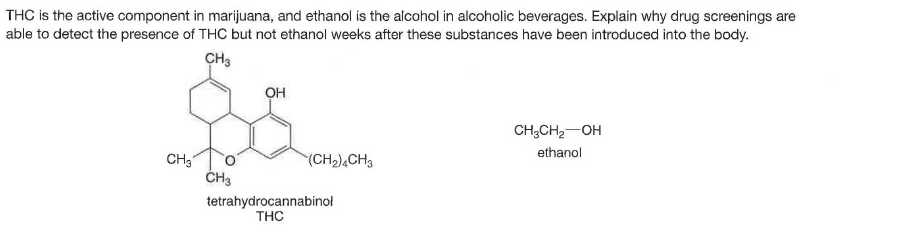
Transcribed Image Text:THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are
able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body.
CH3
OH
CH;CH2-OH
ethanol
CH3
ČH3
(CH),CH3
tetrahydrocannabinol
THC
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning