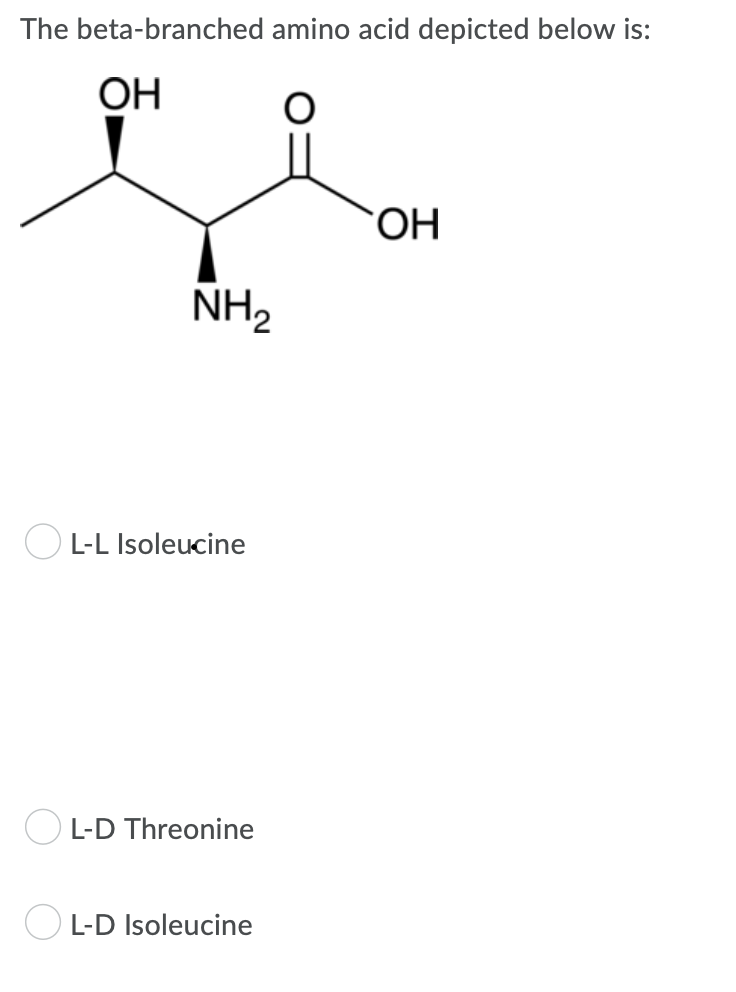
The beta-branched OH amino acid depicted below is: O OH NH₂ OL-L Isoleucine
Q: 10. Why would it be inaccurate to say that solution that maintains a constant pH? KU a buffer is a
A: The pH of blood is approximately 7.4, which is somewhat basic. This pH fluctuates by less than a…
Q: 4. Fill in the table below Pathway Glycolysis Krebs Cycle Electron Transport Chain Number of ATP…
A: Glucose is a carbohydrate that is catabolized by glycolysis into two molecules of pyruvate. Pyruvate…
Q: Identify the types of linkages in each of the three disaccharides. Only place three of the linkages.
A: Disaccharides are consist of two monosaccharides units linked by glycosidic bonds. The type of…
Q: Does TGF-β treatment cause cells to grow more or less in the soft-agar assay? (a) More, (b) Less
A: Anchorage-independent growth, which is a hallmark of carcinogenesis, is the capacity of transformed…
Q: 22. The blood group by the MN system depends on the combi- nation of the codominant genes CM and CN.…
A: For the blood grouping by MN system, According to hardy Weinberg equation, a gene that exists as two…
Q: Which answer is correct (a or b) for the question about why your hands and feet get wrinkled after…
A: In a water molecule, two hydrogen atoms form single polar covalent bonds with an oxygen…
Q: What are site-recombinases? Describe in detail how cre- recombinase can be used to decipher the…
A: Genetic recombination is the transfer of genetic material between organisms, resulting in offspring…
Q: Which of the following statements regarding gluconeogenesis is NOT true: It utilizes all of…
A: Gluconeogenesis refers to the process of synthesis of glucose from non-carbohydrate precursors. It…
Q: What positive impact can negatively charged amino acid in a protein have in the body? Include an…
A: Answers for the following question are :
Q: how to read this table? like for example, just by looking at the table, it was said that leptin has…
A: Introduction: Separation is brought about through the molecular sieving technique, based on the…
Q: Find the connection among the words below and choose the letter of the word which is different. O A.…
A: Gel electrophoresis is the technique used to identify, quantify, and separate nucleic acids. It…
Q: Identify (1) the group where the lipid belongs, and (2) determine whether the lipid is saponifiable…
A: The given structure is of Cortisol also known as hydrocortisone. A saponifiable lipid is part of the…
Q: TAG
A: The conversation of one molecule to its molecular level is called oxidation.i The answer to the…
Q: II. Know your macromolecules. 1. Match the component molecule or building blocks below to their…
A: Biomolecules are carbon-based organic compounds produced by living organisms. They are primarily…
Q: epinephrine/ glucagon
A: The bioactive molecules which helps to catalyze the product in faster rate is called enzyme. The…
Q: What are the x and y values that you have input in the excel? I thought the x would be from the…
A: Proteins are composed of amino acids, coomassie brilliant blue dye binds to basic amino acids of the…
Q: In CO2 assimilation of C4 plants, the acceptor molecule is oxaloacetate. Group of answer choices:…
A: In C4 plant , CO2 assimilation occur via C4 cycle, also known as Hatch- Slack pathway.
Q: QUESTION 2 CH3COO-C₂H5 + H₂O CH3COOH + C₂H5OH Ethyl acetate Acetate Ethanol Ethyl acetate, acetate…
A: Ethyl acetate, acetate and ethanol were added to water solution in concentrations of 0.3M and left…
Q: essential amino acid amino acid side group/R-group peptide bond polypeptide receptor…
A: Amino acids contribute majorly to the making of proteins in the human body. They are important for…
Q: Discuss briefly but concisely the importance of the following metabolic pathway. E. Glycogenolysis…
A: These are the different pathways for carbohydrate metabolism. Glycogenolysis is the pathway for…
Q: The G-proteins associated with GPCRS are made up of three subunits. In order to become active they…
A: The G protein coupled receptors are a classes of cell surface receptors. They are associated with G…
Q: Give the positive result for the test of pyrimidines.
A: The nitrogenous bases of nucleic acids are classified into two. They are, the purines and…
Q: Determine the OPTIMUM pH for the enzyme reaction using the software simulation: (HINT: Run the…
A: Enzymes are the biocatalyst which increases the rate of reaction without itself being changed in…
Q: D.3 Loading Samples and Running the Gel Use special gel loading tips or a micro-syringe to load…
A: Generally when performing western blotting the volume loaded in the well is 20 uL. Since your…
Q: Determine the class of lipid (see attached image for choices) shown by the following structures and…
A: Lipids are a class of biomolecules that are nonpolar in nature. These are hydrophobic in nature and…
Q: Which of the following is an inhibitor of gluconeogenesis? Group of answer choices: -ATP -ADP…
A: Gluconeogenesis is the process of Synthesis of glucose from lactose, pyruvate, glucogenic aminoacids…
Q: Synthesis of a protein from amino acids Formation of a triacylglycerol from glycerol and fatty acids…
A: Anabolic pathway:- involved in synthesis of compound. This process consume energy therefore is…
Q: Discuss briefly but concisely the importance of the following metabolic pathway. H.B-oxidation of…
A: Introduction: Fatty acids from the diet or through degradation of triglycerides stored in the…
Q: Calculate the unknown concentration of the PROTEIN B with an absorbance value of A358 given the…
A: Tp calculate the unknown concentration of protein B from absorbance value, we first need to plot a…
Q: Discuss the central role of glutamate in nitrogen metabolism in both muscle and the liver.
A: Many of the α-amino acids' amino groups are accumulated in the liver as the amino group of…
Q: I need Plant Physiology Help Immediately Please If 2 molecules of phosphoglycolate are produced what…
A: Introduction: Calvin cycle consists of a series of reaction that reduces carbon dioxide to produce…
Q: What is the number of ATP molecules that will be produced from the beta oxidation of a 22 carbon…
A: A fatty acid molecule is a long chain carboxylic acid. A saturated fatty acid is one that lacks…
Q: The mannose 6-phosphate (M6P) receptors are crucial for delivering lysosomal proteins to lysosomes,…
A: The process of vesicular transport is an important mechanism for exchange of proteins and lipids…
Q: How many molecules of NADPH are needed to fix 1 molecule of carbon dioxide? Group of answer…
A: Carbon dioxide is fixed during the photosynthesis process. In photosynthesis, the carbon dioxide is…
Q: Prepare a 75 ml of a 10% (w/v) FeCl3 solution. How many grams of FeCl3 must be weighed out and…
A: Given Values: Volume of FeCl3 solution = 75 ml Concentration of the FeCl3 = 10% (w/v)
Q: 6. Explain briefly the importance of enzyme inhibition in biochemistry.
A: An enzyme is a protein that catalyzes biochemical reaction. It converts substrate into product. A…
Q: KM is determined by measuring the reaction velocity of two enzymes (X and Y) at different…
A: Enzymes are usually protein molecules which catalyzes a biochemical reaction by decreasing its…
Q: The mannose 6-phosphate (M6P) receptors are crucial for delivering lysosomal proteins to lysosomes,…
A: KDEL is a target peptide sequence in mammals and plants located on the C-terminal end of the amino…
Q: escribe how mammalian porphyrins are synthesized from glycine and succinyl coenzyme A ?
A: Porphyrins are crucial for mammals as they control harm biosynthesis. They can move through…
Q: In receptor mediated endocytosis, the receptors (such as the low-density lipoprotein, LDL, receptor)…
A: In receptor mediates endocytosis, Receptors are expressed on the plasma membrane. The receptor bind…
Q: 10.12 2. What is the clinical significance of ALT?
A: Alanine aminotransferase, usually referred to as ALT, is an enzyme that is concentrated primarily in…
Q: In cyclic photophosphorylation, NADPH is produced. True False
A: Photophosphorylation is the conversion of ADP to ATP that occurs in the chloroplasts of cells. The…
Q: For the following three carbohydrates, draw the hawort projection and chair structure
A: D-Glucose, D-Allose, and D-Idose are aldohexoses. These are the monosaccharides. Haworth projection…
Q: A class of animals called ruminant animals that includes grassers (e.g., cows, zebras, giraffes,…
A: Introduction: An adaptation is something that is concerned with the structure or behavior of an…
Q: Spectroscopic/ Chromatogrphic Method olorimetry V Spectroscopy Spectroscopy aman Spectroscopy…
A: Introduction: An analytical technique is applied throughout the industry, medicine, and all the…
Q: Consider the complete oxidation of one mole of simple TAG containing lignoceric acid residues…
A: Fatty acids are degraded through the process of beta-oxidation in the mitochondrial matrix. Each…
Q: The graph below shows the O₂-binding curves for myoglobin (Mb) and hemoglobin (Hb). a. 100 80 60 40…
A: Introduction: Oxygen binding is the binding of an oxygen molecule to a specific functional protein…
Q: Why doesn’t the brain utilize fatty acids for energy
A: The oxidation of fatty acids provides around 95% of the energy derived from fat. Free fatty acids…
Q: 1. Breaking of disulfide bonds in albumin to produce -SH is most likely caused by?* Acetic acid…
A: Specific biological tests can be performed to identify specific biomolecules like nucleic acids,…
Q: Which of the following statements is TRUE about cholesterol biosynthesis? NADPH is needed to form…
A: Cholesterol is a type of lipid or sterol. Cholesterol is an important constituent of the plasma…
please choose one of the three options
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- Define the following terms: a. reducing sugar b. alditol c. enediol d. acetal e. ketalWhat following compound can be found with a tyrosyl unit? A) Glycosyl phosphatidyl inositol B) Sphingosine 1 -phosphate C) Glycogen D) Serotonin E) S-glutathioneDefine the following terms: a. arginosuccinate b. Krebs bicycle c. N-acetylglutamate d. BH4 e. taurine
- All of the following are repeating units of glycosaminoglycans EXCEPT: A. hyaluronate B. heparin C. hemoglobin D. keratin sulfate E. chondroitin 6-sulfateDefine the following terms:a. fatty acidb. monunsaturated fatty acidc. polyunsaturated fatty acidd. saturated fatty acide. acyl groupClassify each of the following sugar pairs as enantiomers, diastereomers, epimers, or an aldose–ketose pair. a. D-erythrose and D-threose b. D-glucose and D-mannose c. D-ribose and L-ribose d. D-allose and D-galactose e. D-glyceraldehyde and dihydroxyacetone
- Butanoic acid (C3H7COOH) is described as a weak acid. Define the term weak acidAldehyde sugars are also referred to as _________________.Relate the molecular properties to physicochemical properties of the following lauric acid stearic acid oleic acid methyl laurate methyl oleate a-tocopherol hexadecane cholesterol estrogen





