The graph below shows the O₂-binding curves for myoglobin (Mb) and hemoglobin (Hb). a. 100 80 60 40 20 0 0 20 40 60 80 100 pO₂ (torr) a. Use the graph to determine the Kd of myoglobin for O₂ (show your approach). Write that in the provided space. b. What is the difference between myoglobin and hemoglobin that cause the O2- binding curves to so be different? Answer in the provided space. YO?
The graph below shows the O₂-binding curves for myoglobin (Mb) and hemoglobin (Hb). a. 100 80 60 40 20 0 0 20 40 60 80 100 pO₂ (torr) a. Use the graph to determine the Kd of myoglobin for O₂ (show your approach). Write that in the provided space. b. What is the difference between myoglobin and hemoglobin that cause the O2- binding curves to so be different? Answer in the provided space. YO?
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter11: The Blood
Section: Chapter Questions
Problem 1SQE
Related questions
Question
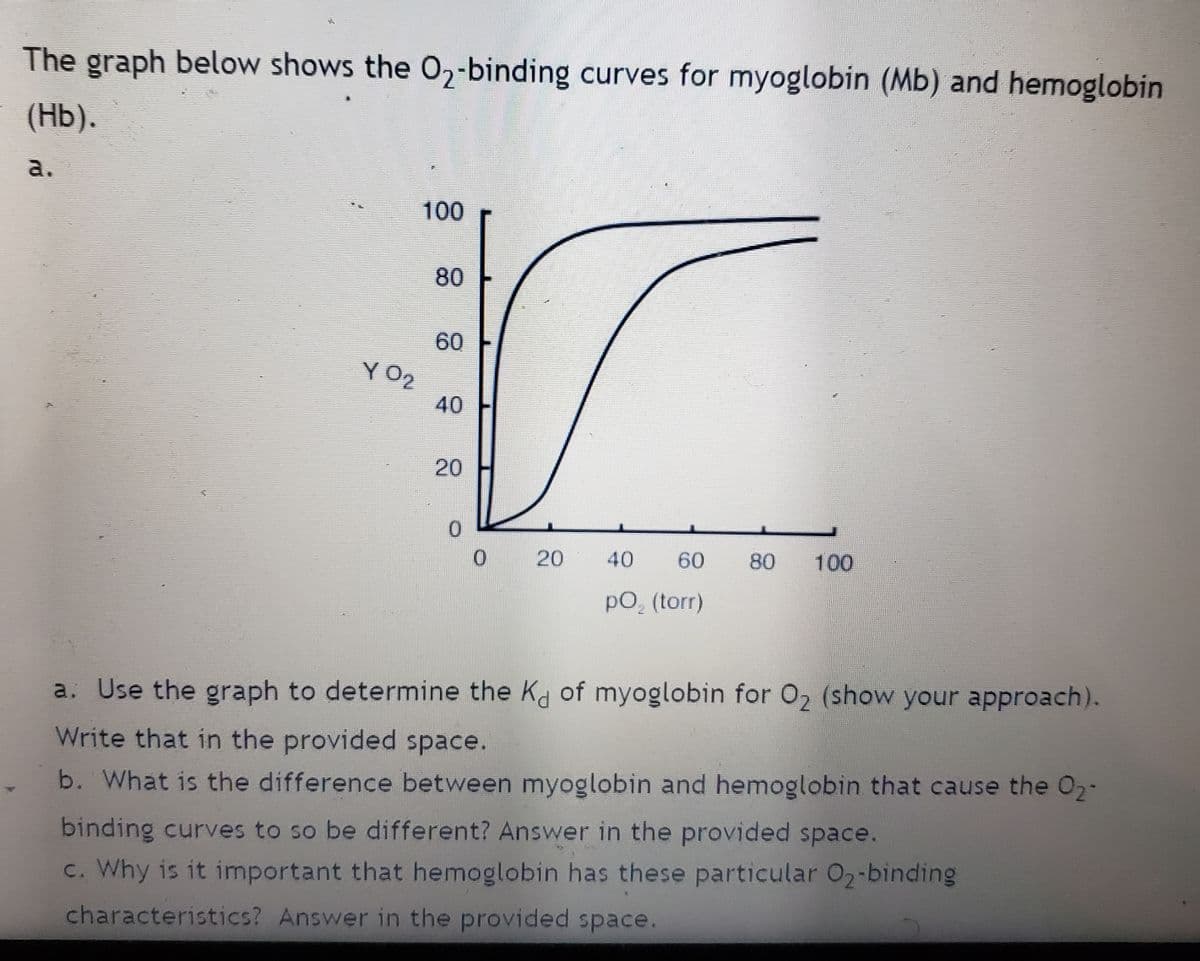
Transcribed Image Text:The graph below shows the O₂-binding curves for myoglobin (Mb) and hemoglobin
(Hb).
a.
100
80
60
40
20
0
0
20
40 60 80 100
pO₂ (torr)
a. Use the graph to determine the Kd of myoglobin for O₂ (show your approach).
Write that in the provided space.
b. What is the difference between myoglobin and hemoglobin that cause the O₂-
binding curves to so be different? Answer in the provided space.
c. Why is it important that hemoglobin has these particular O₂-binding
characteristics? Answer in the provided space.
YO₂
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning


Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning
