The carbon skeletons of many amino acids can be used to replenish the intermediates of the citric acid cycle. Select one: True O False Which of the following compound is a positive allosteric modifier of the enzyme pyruvate carboxylase? Select one: a. Biotin O b. Oxaloacetate O c. Fructose-6-P O d. ATP Oe. Acetyl-CoA Which of the following compounds serve as a primary link between the TCA cycle and the urea cycle? Select one: O a. succinate b. fumarate O c. oxaloacetate O d. malate Oe. citrate A specific inhibitor for succinate dehydrogenase is: Select one: O a. Citrate b. Malonate O c. Arsenite O d. Fluoride O e. Cyanide
The carbon skeletons of many amino acids can be used to replenish the intermediates of the citric acid cycle. Select one: True O False Which of the following compound is a positive allosteric modifier of the enzyme pyruvate carboxylase? Select one: a. Biotin O b. Oxaloacetate O c. Fructose-6-P O d. ATP Oe. Acetyl-CoA Which of the following compounds serve as a primary link between the TCA cycle and the urea cycle? Select one: O a. succinate b. fumarate O c. oxaloacetate O d. malate Oe. citrate A specific inhibitor for succinate dehydrogenase is: Select one: O a. Citrate b. Malonate O c. Arsenite O d. Fluoride O e. Cyanide
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter17: Metabolism: An Overview
Section: Chapter Questions
Problem 15P
Related questions
Question
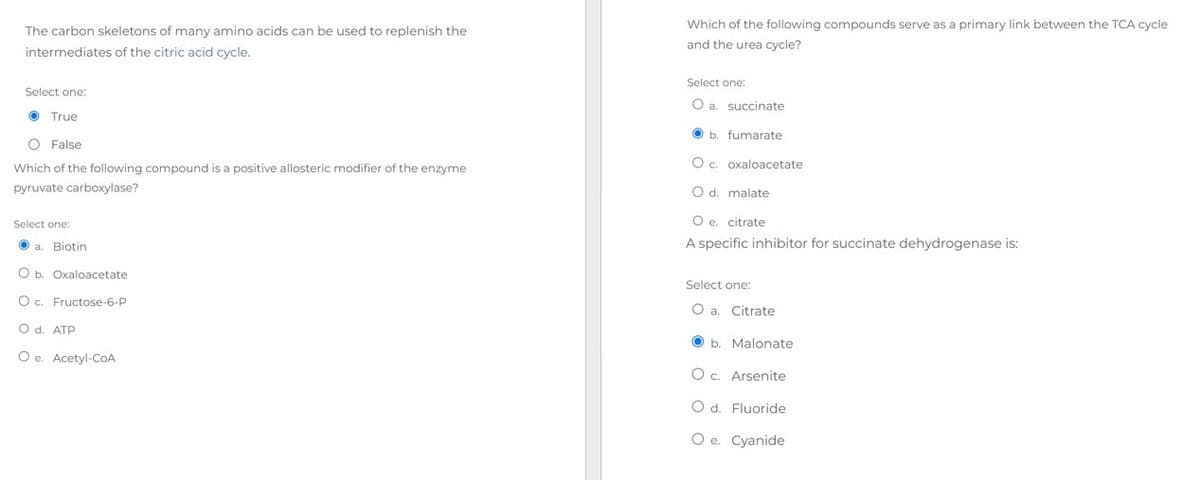
Transcribed Image Text:The carbon skeletons of many amino acids can be used to replenish the
intermediates of the citric acid cycle.
Select one:
True
O False
Which of the following compound is a positive allosteric modifier of the enzyme
pyruvate carboxylase?
Select one:
a. Biotin
O b. Oxaloacetate
O c. Fructose-6-P
O d. ATP
Oe. Acetyl-CoA
Which of the following compounds serve as a primary link between the TCA cycle
and the urea cycle?
Select one:
O a. succinate
b. fumarate
O c. oxaloacetate
O d. malate
O e. citrate
A specific inhibitor for succinate dehydrogenase is:
Select one:
O a. Citrate
b Malonate
O c. Arsenite
O d. Fluoride
O e. Cyanide
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning