1 is there a positive or negative entropy change in the first step of histidine synthesis? 2 how many 1-carbon transfer reactions occur in the pathway? 3 how many transamination reactions occur in the pathway ?
1 is there a positive or negative entropy change in the first step of histidine synthesis? 2 how many 1-carbon transfer reactions occur in the pathway? 3 how many transamination reactions occur in the pathway ?
Chapter3: Carbohydrates
Section: Chapter Questions
Problem 7SC
Related questions
Question
1 is there a positive or negative entropy change in the first step of histidine synthesis?
2 how many 1-carbon transfer reactions occur in the pathway?
3 how many transamination reactions occur in the pathway ?
4 in purine and pyrimidine synthesis , ________ from the_______pathway is activated by _______
5 which molecule functions as both an amino group donor and acceptor in this pathway ?
6 during the degradation of histidine, the amino acid group will give rise to _______ while the carbon skeleton will give rise to _______ does not need energy.
7 to which chemical messenger will the decarboxylation of histidine give rise?
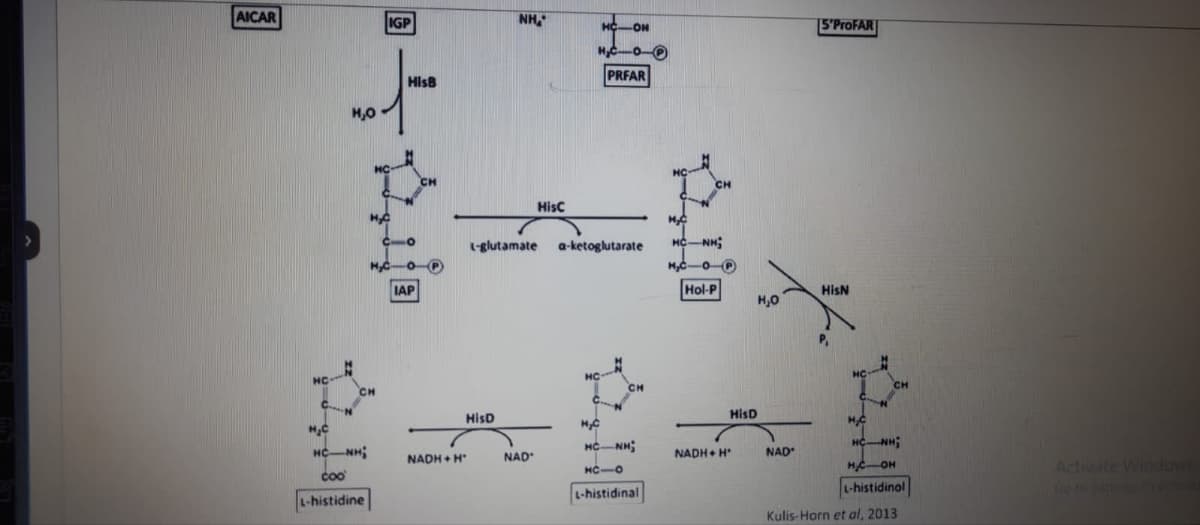
Transcribed Image Text:AICAR
HC
H₂O
CH
H₂C
HỆ—NH;
coo
L-histidine
IGP
HisB
IAP
NADH+H
NH₂
HisD
HisC
L-glutamate a-ketoglutarate
NAD
HC ON
H₂C-09
PRFAR
HC
CH
H₂C
HỘ NHỎ
HC-O
L-histidinal
CH
H₂C
MỘ NHẬ
M,C109
Hol-P
H₂O
HisD
NADH+H*
S'ProFAR
NAD
HisN
P₁
HC
CH
H₂C
HỒ—NH;
H₂C-OH
L-histidinol
Kulis-Horn et al, 2013
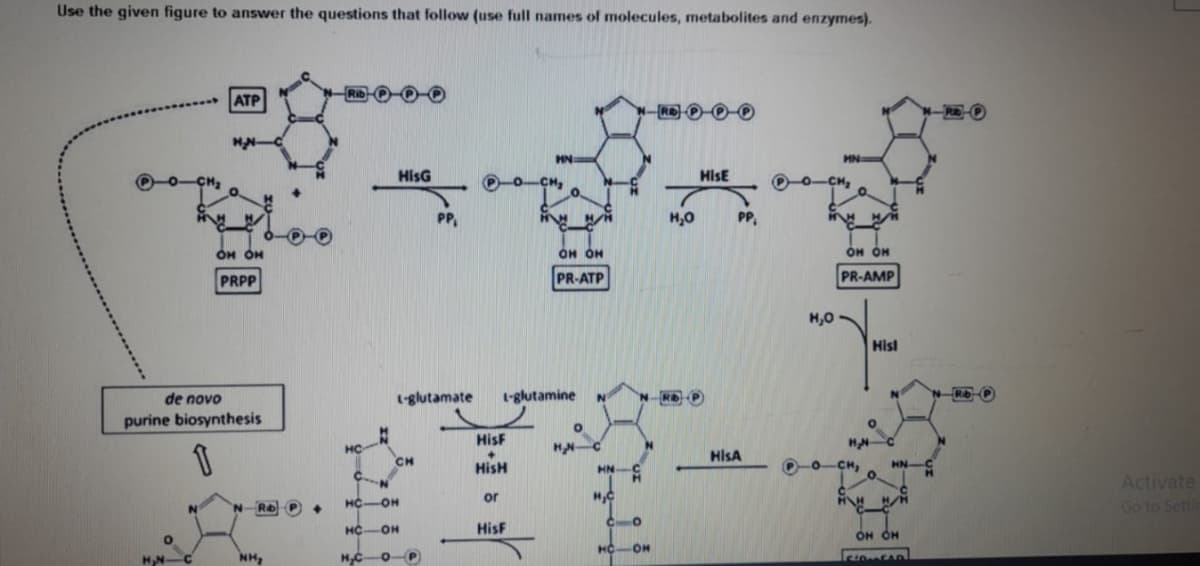
Transcribed Image Text:Use the given figure to answer the questions that follow (use full names of molecules, metabolites and enzymes).
-CH₂
H₂N-
ATP
H₂N-
OH OH
PRPP
de novo
purine biosynthesis
1
N
Rib
NH₂
+
Rib
HC
#
HisG
L-glutamate
CH
PP₁
HC- OH
HC OH
H₂C OP
HisF
HisH
or
HN:
HisF
-CH₂
HHHH
L-glutamine N
OH OH
PR-ATP
O
H₂N-
HN
H₂C
NRD℗ ℗ ℗
N-RD-P
CO
N
H₂O
HC -OH
HisE
PP₁
HISA
℗0
HN
-CH₂
H₂O
OH OH
PR-AMP
CH,
Hist
H₂N-
H
O
HN-
OH OH
CONCAD
RD-P
Activate
Go to Settin
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

