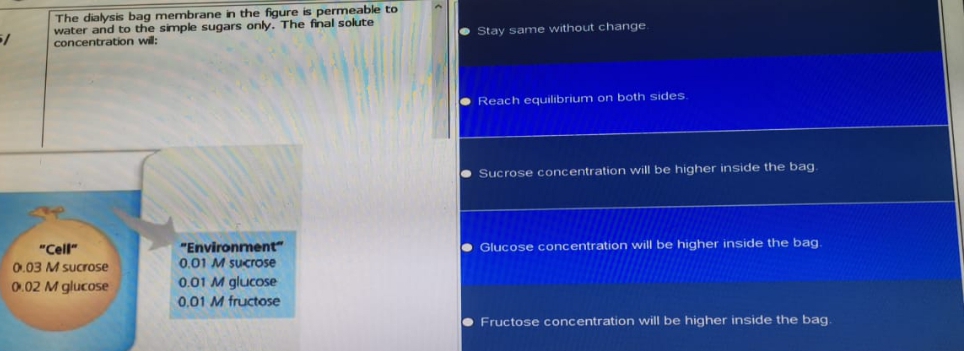
The dialysis bag membrane in the figure is permeable to water and to the simple sugars only. The final solute concentration wil: Stay same without change Reach equilibrium on both sides. Sucrose concentration will be higher inside the bag "Environment" 0.01 M SUcrose Glucose concentration will be higher inside the bag. "Cell" 0.03 M sucrose 0.02 M glucose 0.01 M glucose 0.01 M fructose Fructose concentration will be higher inside the bag
Q: Which of the following describes cotransport (secondary active transport)? Group of answer choices…
A: The plasma membrane of the cell is the regulatory component that is responsible for the regulation…
Q: Figure 1: Osmolarity of Potato 30.00 25.00 20.00 15.00 10.00 5.00 0.00 OM sucrose 0.1 M sucrose 0.2…
A: The concentration of osmotically active particles in solution, that may be quantitatively expressed…
Q: Water Water Starch Starch Glucose + Glucose Glucose The investigation is aimed at understanding how…
A: Hi! Since you have posted multiple questions , we will answer the first question as we are only…
Q: Construct a graph showing difusión time vs column height: don't forget to label axis. Consider how…
A: Diffusion is the movement of a substance from an area of high concentration to an area of low…
Q: The driving force for diffusion is Select one: the kinetic energy fro the molecules in motion. ATP…
A: The cell membrane is a structure which protects the cell and acts as a barrier and transports the…
Q: In the experimental conditions described below, how many molecules of dextrose do you have to add to…
A: Osmosis is defined as the movement of solvent particles from a region of higher concentration to a…
Q: Rank the following solutes from lowest (1) to highest (5) membrane permeability: Explain your…
A: The plasma membrane is the outer layer of the cell. It is also known as the cell membrane. It…
Q: Which of the following best describes a hypotonic solution? There is a higher concentration of…
A: Hypotonic means solute concentration is lower than solvent in a solution. When in a solution…
Q: When comparing the solute concentrations of two fluids, the fluid with the higher solute…
A: A solution has two parts Solute Solvent
Q: Water intoxication is caused by
A: Water intoxication results from drinking too much water that exceeds the kidney's ability to…
Q: Which of the following molecules can diffuse across the dialysis tubing? a. Iodine b.Starch…
A: Cell membrane is the semi permeable ( selectively permeable) membrane that surrounds a cell and…
Q: Which of the following would decrease the electrochemical gradient across a membrane? an ion channel…
A: The electrochemical gradient is the electrical and chemical gradient or concentration differences…
Q: Choose the best option about how cholesterol affects cell membrane fluidity? Select one: O a.…
A: Cholesterol is a lipid present in the cell membrane of animal cells. It is involved in the…
Q: If distilled water (no ions or salt) is added to red blood cells in the laboratory, the red blood…
A: If distilled water (no ions or salt) is added to red blood cells in the laboratory, the red blood…
Q: Assuming that you will need to include osmotic shock as one of the steps, calculate the maximum…
A: The difference in osmolarity across a membrane leads to the differential movement of water. If the…
Q: Which of the following statements is TRUE? A. Water and electrolytes move across the cell membranes…
A:
Q: In the Nernst equation [V = 62 log10 (Co/ Ci)], the term Co represents: the intracellular…
A: Nernst equation can be defined as one that establishes a relationship between the reduction…
Q: The "cell" has a sucrose concentration of 0.1M and a glucose concentration of 0.1M. The…
A: Osmosis is the diffusion of water across a semi-permeable membrane to maintain an equal…
Q: An amino acid mixture consisting of phenylalanine, glycine, and glutamic acid is to be separated by…
A: HPLC is often referred to as high-pressure liquid chromatography. It exploits the same basic…
Q: 3. Draw and label a diagram that demonstrates the difference between tonicity and osmolarity. Be…
A: The process of controlling bodily fluids and their compositions is known as osmoregulation. It keeps…
Q: Which of the following statements is correct? A. Short-chain fatty acids have higher transition…
A: Introduction The viscosity of the lipid bilayer of a cell membrane or a synthetic lipid membrane is…
Q: Select the correct lettered answer. ____ 1. In the dialysis bag experiment, sucrose and red dye…
A: A solution can be defined as the homologous mixture of solute and solvent. Most often the term…
Q: High High Fructose Water Low Lipids Low Simple diffusion Gigh Osmosis High Low Low Endocytosis Nat…
A: Thank you for the question Answer Simple diffusion = Lipids Facilitated diffusion = Fructose…
Q: What is the importance of acids and bases in our daily activities? State examples. What is the pH of…
A: Acids are the substances that donates H+ ions and have pH range between 1-6 Bases are those which…
Q: In an investigation of osmosis, apple cores were submerged in different molarity solutions to…
A:
Q: Which of the following statements, regarding the figure below, is correct? (Glucose]jow Glu Na…
A: Digestion occur in the small intestine where breakdown of food occur and thus absorption occur by…
Q: Two solutions are separated by a membrane that is impermeable to glucose. Solution A is 5 percent…
A: hypotonic solution is one which have a less solute concentration than surrounding while hypertonic…
Q: Rate of glucose transport The below graph plots the rate of glucose transport through a membrane…
A: GLUT transporters are found in various tissues and these transporters are responsible for the…
Q: List the following from which is MOST permeable to LEAST permeable to an intact cell membrane water…
A: The ease with which a molecule can flow through a cell membrane is referred to as "permeability".…
Q: Rate of glucose transport 0 The below graph plots the rate of glucose transport through a membrane…
A: The rate of glucose transport into a cell by a glucose transporter is given by the equation below.…
Q: A dialysis tube is constructed of a cellphone-like material. The molecular structure of the material…
A: Dialysis is a medical procedure that performs the removal of solutes, toxins and excess water from…
Q: atty acid increase membrane fluidity
A: The basic structure of the cell membrane (CM) is formed by the phospholipids and made this membrane…
Q: What type of transporter moves glucose into the cell against its concentration gradient? A.…
A: The plasma membrane of the organism is a selectively permeable one which only allows restricted…
Q: MULTIPLE CHOICE Which of the following has the lowest solute potential? A. a 0.5 molar solution of…
A: Solute potential :-
Q: The normal osmolarity and glucose level of plasma are 300mmol/L and 1g/L, respectively. Estimate the…
A: Mw of glucose= 180 Molar concentration (C) = Mass conc. (g/l) / Mw = 1/ 180 = 0.005 mol/l (Normal…
Q: The ability of the Goldenrod Gall Moth caterpillar to avoid freezing is due to
A: Cell membrane or plasma membrane consists of phospholipids and proteins. when you see the structure…
Q: If a skeletal muscle has depleted its stores of ATP how will the altered transport properties of the…
A: If a skeletal muscle has depleted with its stored ATP:-
Q: By calculating the osmolarity of each of the following solutions, which solutions can be used in…
A: M here signifies Molarity of solution . 1M solution of a nonionizing substance such as glucose is a…
Q: you have to add to the extracellular fluid in order to make it iso-osmotic relative to the…
A: Solutions that have the same particle concentration and thus have the same osmotic pressure are…
Q: An experiment was conducted with 100 volunteers and the following data was collected. Туре of acid…
A: Lactic acid is produced when there is anaerobic respiration is the muscles seen in activities which…
Q: A concentration gradient affects the direction that solutes diffuse. Describe how molecules move…
A: The molecules of the different elements present in the world are not static or fixed in one place.…
Q: ring dialysis, a semi-permeable membrane with a molecular weight cutoff of 10,000 is used. Select…
A: Urine is a fluid excretion derived from the filtration of blood in kidneys. Kidney is a bean-shaped…
Q: This is not a graded question
A: Lactic acid fermentation is a form of metabolism that occurs when there is a lack of oxygen or when…
Q: explain why the concentration of water should be nearly the same inside the cell and in the…
A: The movement of water is seen from higher concentration to a lower concentration through the…
Q: An example for Secondary active transport sodium potassium bumps Glucose entrance to red blood cells…
A: Secondary active transport is the transport of molecules across the cell membrane by utilising…
Q: Let’s say that the intracellular tonicity is 300 mosm. Checkmark the correct response to what would…
A: The ability of an extracellular solution to make water move into or out of a cell by osmosis is…
Q: extracellular concentration time extracellular concentration While studying the rate of transport of…
A: The phenomenon of natural passive transport (as contrast to active transport) of solutes along a…
Q: An animal cell with an internal osmolarity of 0.32 M (total concentration of all the solutes in…
A: Osmosis Osmosis is a physical process of movement of solvent molecules across a plasma membrane…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- You are developing a porous membrane for use in a dialysis system. The membrane must be able to retain both protein and glucose on the inlet side and allow other, smaller molecules to flow through. You have found that the membrane is 0.25 mm thick and contains long, rectangular pores with a width of 0.1 microns. 57% of the 50 cm^2 membrane surface area is covered with pores. A test fluid (viscosity = 1.5 cP, density = 1015 kg/m^3) is passed through the membrane. You can assume that the test fluid has a composition similar to that of blood plasma. An initial test is run at physiological conditions, and you observe that the flow rate of fluid through the membrane is 500 cm^3/min. _Given this data, what must the hydrodynamic pressure drop across the membrane in your test system be in pascals?On your bench, there are two tubes, Tube X and Tube Y. One tube contains 1.2 mL of 300 mosm Sucrose, and the other tube contains 1.2 mL of 300 mosm Urea. After adding 20 ul of blood to each tube, you observed the hemolysis in Tube X, but not in Tube Y. Which of the following two tubes contains Sucrose or Urea? Tube X = Tube Y = If the 2 solutions have the same osmolarity, explain how can they lead to different cell responses?A 139 lb male patient with severe lactate acidosis is given isotonic sodium lactate. The doctor orders isotonic sodium lactate 50. mL/ kg body mass to be administered intravenously. The rate of flow is 150 gtts/min, and the IV administration set delivers 20. gtts/mL, where the unit “gtts” stands for drops of liquid. Calculate what is the running time in minutes will be? Show all your calculations
- calculate the drop rate of an IV if 1000mL of a 5% glucose solution is to be administered over a period of a 8h and the drop factor is 20 gtt/mLA new 850 cm^2 membrane is being tested for use in a hemodialysis device. During testing, a model fluid mixture is used that contains solutes with average radii of 1 nm. In one test, the concentration of the filtrate is found to be 2.17 g/L, while the concentration on the feed-side surface is 8.52 g/L. Prior experimentation showed that the mass transfer coefficient in the device was approximately 9 × 10-4 cm/s. A) If the total filtration flow rate is measured to be 0.56 cm^3/s, what is the solute concentration in the feed solution? B) Explain how you could determine the average membrane pore radius for this membrane using this information.Composition of Cell Lysis Buffer Required Concentration of Reagent Molecular Weight Volume or Weight of Reagents Needed 40 mM Tris HCl 157.59 20 mM Acetic acid 60.05 1 mM EDTA 292.24 10 mM MgCl2 from 25mM stock 95.21 5% SDS 288.37 ddH2O 18.02
- G.What concentration of imidazole do you expect after you dialyze 1 mL of a sample containing 500 mM imidazole into 100 mL of dialysis buffer containing no imidazole? What if you repeat the process by moving the dialysis cassette to a fresh 100 mL of dialysis buffer?Can sucrose pass freely through the membrane of a dialysis tube? Explain using examples. (Biochemistry)Mannitol, a carbohydrate, is supplied as a 25% (w/v) solution. This hypertonic solution is given to patients who have sustained a head injury with associated brain swelling. (a) What volume should be given to provide a dose of 70. g? (b) How does the hypertonic mannitol benefi t brain swelling?
- If a dialysis tube that is permeable to water but not sucrose contains a 40% sucrose solution that is in a beaker of water, the solution in the beaker is said to be: cannot be determined isotonic hypertonic hypotonicuring dialysis, a semi-permeable membrane with a molecular weight cutoff of 10,000 is used. Select the true statements from the list below (more than one answer could apply) Group of answer choices A monoclonal antibody with a 150 kDa MW is retained and does not diffuse across the membrane A small enzyme with 20 kDa MW diffuses readily across the membrane Buffer salts with molecular weights ranging between 100 and 500 Da diffuse across the membrane until equilibrium (equal concentration) is reached on both sides of the membrane None of the above statements are correct Please answer asapA 500 mL infusion of glucose 5% is being given at a rate of 167 mL/hr. How long will the infusion run at the specified rate?

