The docomposition of N,O, can be described by the equation 2N,0, (soln) 4 NO,(soln) + O,(2) Consider thc data in the table for the reaction at 45 C in carbon ictrachloride solution. IN,010M) 2.011 205 1.768 426 1.538 765 1.243 Given the dala, calculate the avcrage ralc of reaction for cach sucCEssive time inlerval. What is the avcrage ralc of reaction for the Lime interval from 0x to 205 s? veragc ralc of reaction: What is the average rale ol reaction for the Lime inicrval from 205 s to 426 s? zvcrae raie of reaction: What is the average rate ol reaction for the time interval from 426is to 765 x avcrare rate of reaction:
The docomposition of N,O, can be described by the equation 2N,0, (soln) 4 NO,(soln) + O,(2) Consider thc data in the table for the reaction at 45 C in carbon ictrachloride solution. IN,010M) 2.011 205 1.768 426 1.538 765 1.243 Given the dala, calculate the avcrage ralc of reaction for cach sucCEssive time inlerval. What is the avcrage ralc of reaction for the Lime interval from 0x to 205 s? veragc ralc of reaction: What is the average rale ol reaction for the Lime inicrval from 205 s to 426 s? zvcrae raie of reaction: What is the average rate ol reaction for the time interval from 426is to 765 x avcrare rate of reaction:
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.22QAP
Related questions
Question
!
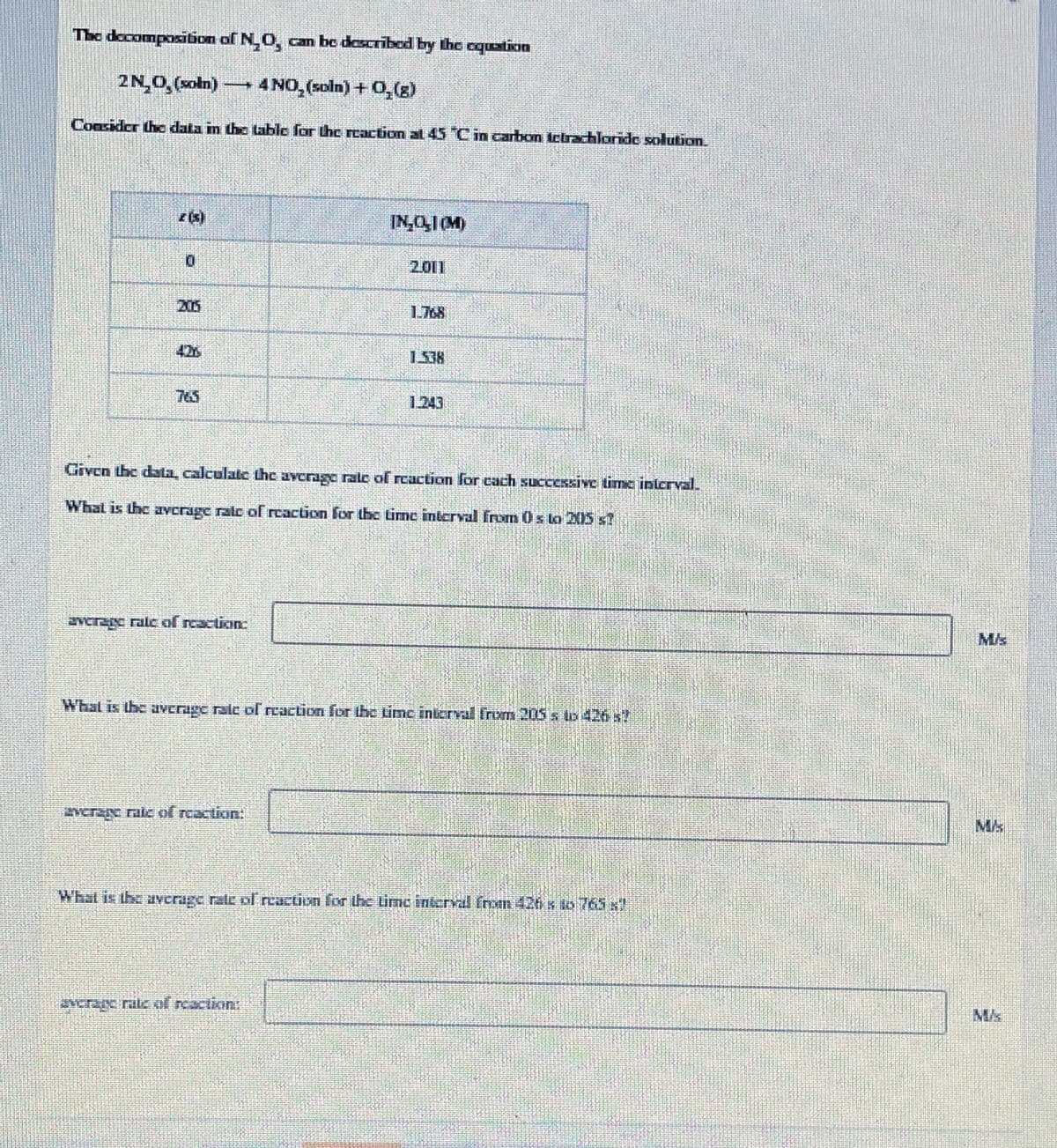
Transcribed Image Text:The docompositbion of N.O, can be describod by the oquation
2N0, (xoln)
→4NO, (xoln)+0,(g)
Consider thc data in the ttable for the caction at 45 Cin carbon ictrachloride solution.
IN,0,1(M)
2.011
205
1.768
426
1.538
765
1.243
Given the dla, calculate the average rale of reaction for cach successive time inlerval.
What is the avcrage ralc of rcaction for the Lime interval from 0 s to 2015 x7
verzgc ralc of rcaction
What is the average ralc of reaction for the time inarval Irom 2OS to 426 s
vcrare ratc of reaction:
What is the ayerage rate pl reaction for the time interval from 420 s to 765 x
vcrancrale of rcactikin
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

