The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: Ry E=- In this equation R, stands for the Rydberg energy, and n stands for the principal quantum number of the orbital that holds the electron. (You can find the valu of the Rydberg energy using the Data button on the ALEKS toolbar.) Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n= 4 to an orbita with n = 10. Round your answer to 3 significant digits. um
The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: Ry E=- In this equation R, stands for the Rydberg energy, and n stands for the principal quantum number of the orbital that holds the electron. (You can find the valu of the Rydberg energy using the Data button on the ALEKS toolbar.) Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n= 4 to an orbita with n = 10. Round your answer to 3 significant digits. um
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter5: Electron Configurations And The Periodic Table
Section: Chapter Questions
Problem 31QRT
Related questions
Question
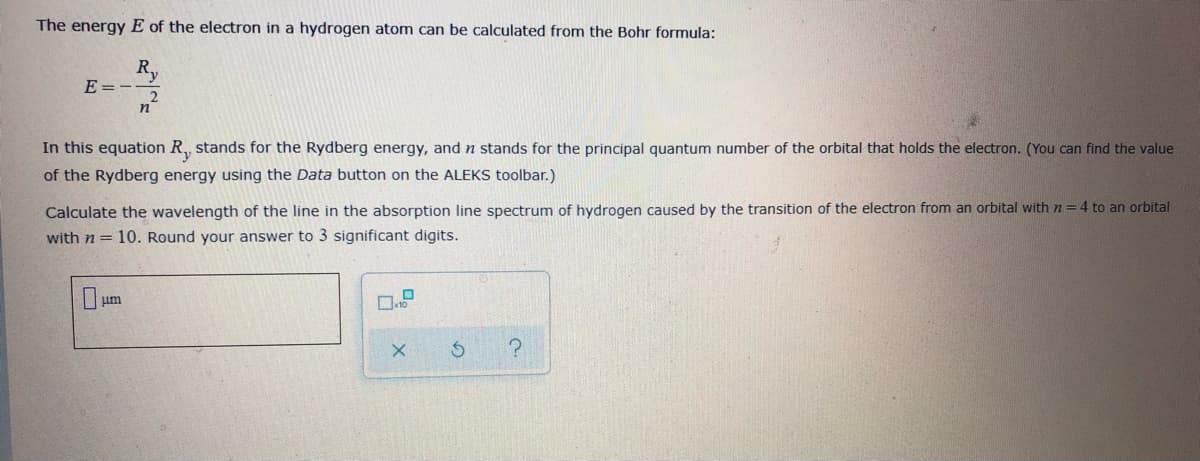
Transcribed Image Text:The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula:
R,
E = -
n°
In this equation R,, stands for the Rydberg energy, and n stands for the principal quantum number of the orbital that holds the electron. (You can find the value
of the Rydberg energy using the Data button on the ALEKS toolbar.)
Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n=4 to an orbital
with n = 10. Round your answer to 3 significant digits.
Lum
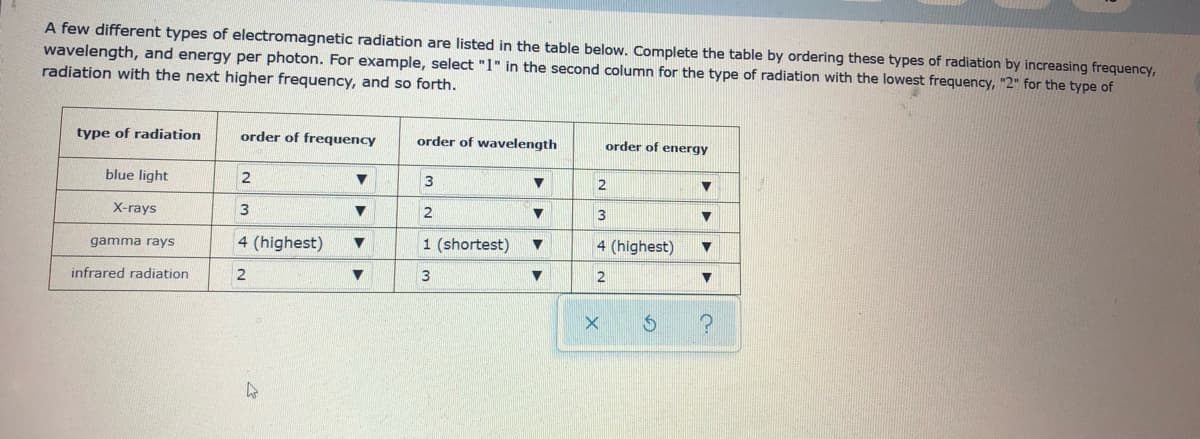
Transcribed Image Text:A few different types of electromagnetic radiation are listed in the table below. Complete the table by ordering these types of radiation by increasing frequency,
wavelength, and energy per photon. For example, select "1" in the second column for the type of radiation with the lowest frequency, "2" for the type of
radiation with the next higher frequency, and so forth.
type of radiation
order of frequency
order of wavelength
order of energy
blue light
2
3
2
X-rays
3
3
4 (highest)
1 (shortest)
4 (highest)
gamma rays
2
infrared radiation
2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning
