The figure below represents the highest occupied molecular orbitals (HOMOS) and the lowest unoccupied molecular orbital (LUMO) for an organic compound with a ketone functional group. The and n orbitals represent the HOMOS and the x* represents the LUMO orbital. E n JI * Nonpolar solvent J Polar solvent H. H H H The left side HOMOS and LUMO represent the molecule in a nonpolar solvent, and the right side represent the HOMOS and LUMD in a polar solvent, such as water. You can see the change in the energy of the HOMOS and LUMO when the polar solvent interacts with the electrons in the aan n orbitals. Study the figure and choose which of the following statements are true or false about why there is a blue shift in the electronic transition. Select all that are True. the energy gap between the n MO and the MO is larger in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent the energy gap between the n MO and the MO is smaller in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent the polar solvent forms an H-bond with the non-polar bond electrons and lowers the energy of the MO much more than the * MO is lowered HOMO more than the a* LUMO MO much more than the x MO is lowered a manipolar solvent would have an extensive interaction with the banding electrons and lower the energy of the the polar solvent forms an H-bond with the polar nonbonding electrons in the n MO and lowers the energy of the it makes sense that a polar solvent would form a strong attraction for the electrons in a nonbonding orbital on the end of a very polar C-0 band; stronger IM forces mean lower energy for the n-electrons
The figure below represents the highest occupied molecular orbitals (HOMOS) and the lowest unoccupied molecular orbital (LUMO) for an organic compound with a ketone functional group. The and n orbitals represent the HOMOS and the x* represents the LUMO orbital. E n JI * Nonpolar solvent J Polar solvent H. H H H The left side HOMOS and LUMO represent the molecule in a nonpolar solvent, and the right side represent the HOMOS and LUMD in a polar solvent, such as water. You can see the change in the energy of the HOMOS and LUMO when the polar solvent interacts with the electrons in the aan n orbitals. Study the figure and choose which of the following statements are true or false about why there is a blue shift in the electronic transition. Select all that are True. the energy gap between the n MO and the MO is larger in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent the energy gap between the n MO and the MO is smaller in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent the polar solvent forms an H-bond with the non-polar bond electrons and lowers the energy of the MO much more than the * MO is lowered HOMO more than the a* LUMO MO much more than the x MO is lowered a manipolar solvent would have an extensive interaction with the banding electrons and lower the energy of the the polar solvent forms an H-bond with the polar nonbonding electrons in the n MO and lowers the energy of the it makes sense that a polar solvent would form a strong attraction for the electrons in a nonbonding orbital on the end of a very polar C-0 band; stronger IM forces mean lower energy for the n-electrons
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter17: Conjugation And Molecular Orbital (mo) Theory
Section: Chapter Questions
Problem 15E
Related questions
Question
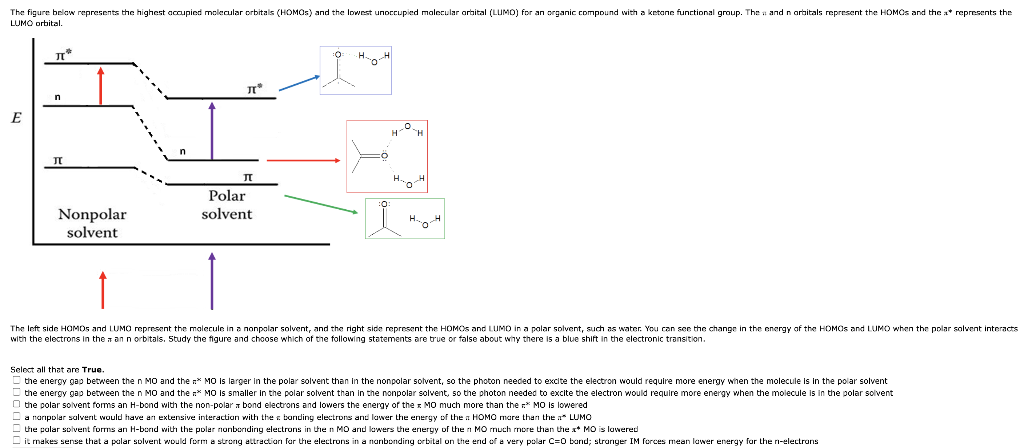
Transcribed Image Text:The figure below represents the highest accupied molecular orbitals (HOMOS) and the lowest unoccupied molecular orbital (LUMO) for an organic compound with a ketone functional group. The and n orbitals represent the HOMOS and the a* represents the
LUMO orbital.
л"
E
I
Nonpolar
solvent
I*
Л
Polar
solvent
To
The left side HOMOS and LUMO represent the molecule in a nonpolar solvent, and the right side represent the HOMOS and LUMD in a polar solvent, such as water. You can see the change in the energy of the HOMOS and LUMO when the polar solvent interacts
with the electrons in the ann orbitals. Study the figure and choose which of the following statements are true or false about why there is a blue shift in the electronic transition.
Select all that are True.
the energy gap between the n MO and the n* MO is larger in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent
the energy gap between the n MO and the * MO is smaller in the polar solvent than in the nonpolar solvent, so the photon needed to excite the electron would require more energy when the molecule is in the polar solvent
the polar solvent forms an H-bond with the non-polar bond electrons and lowers the energy of the x MO much more than the * MO is lowered
a nanpolar solvent would have an extensive interaction with the bonding electrons and lower the energy of the a HOMO more than the a* LUMO
the polar solvent forms an H-bond with the polar nonbonding electrons in the n MO and lowers the energy of the n MO much more than the x* MO is lowered
it makes sense that a polar solvent would form a strong attraction for the electrons in a nonbonding orbital on the end of a very polar C=0 band; stronger I
forces mean lower energy for the n-electrons
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,