The following reaction sequence was used in the synthesis of several derivatives of pros- taglandin C2. Analyze the structure using the chemistry you learned in this chapter and draw the structures of the synthetic intermediates A and B. 1. NANH, THF(solvent) NaCN A В 2. Br CI DMSO (racemic) (racemic) (racemic)
The following reaction sequence was used in the synthesis of several derivatives of pros- taglandin C2. Analyze the structure using the chemistry you learned in this chapter and draw the structures of the synthetic intermediates A and B. 1. NANH, THF(solvent) NaCN A В 2. Br CI DMSO (racemic) (racemic) (racemic)
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter9: Nucleophilic Substitution And Β-elimination
Section: Chapter Questions
Problem 9.63P
Related questions
Question
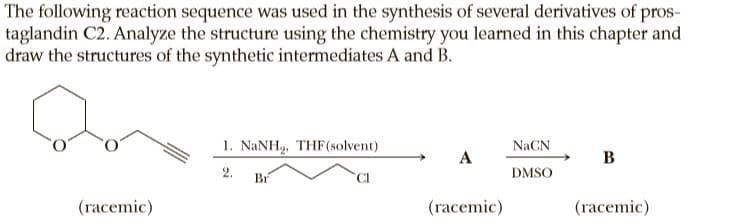
Transcribed Image Text:The following reaction sequence was used in the synthesis of several derivatives of pros-
taglandin C2. Analyze the structure using the chemistry you learned in this chapter and
draw the structures of the synthetic intermediates A and B.
1. NANH, THF(solvent)
NaCN
A
В
2.
Br
CI
DMSO
(racemic)
(racemic)
(racemic)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning