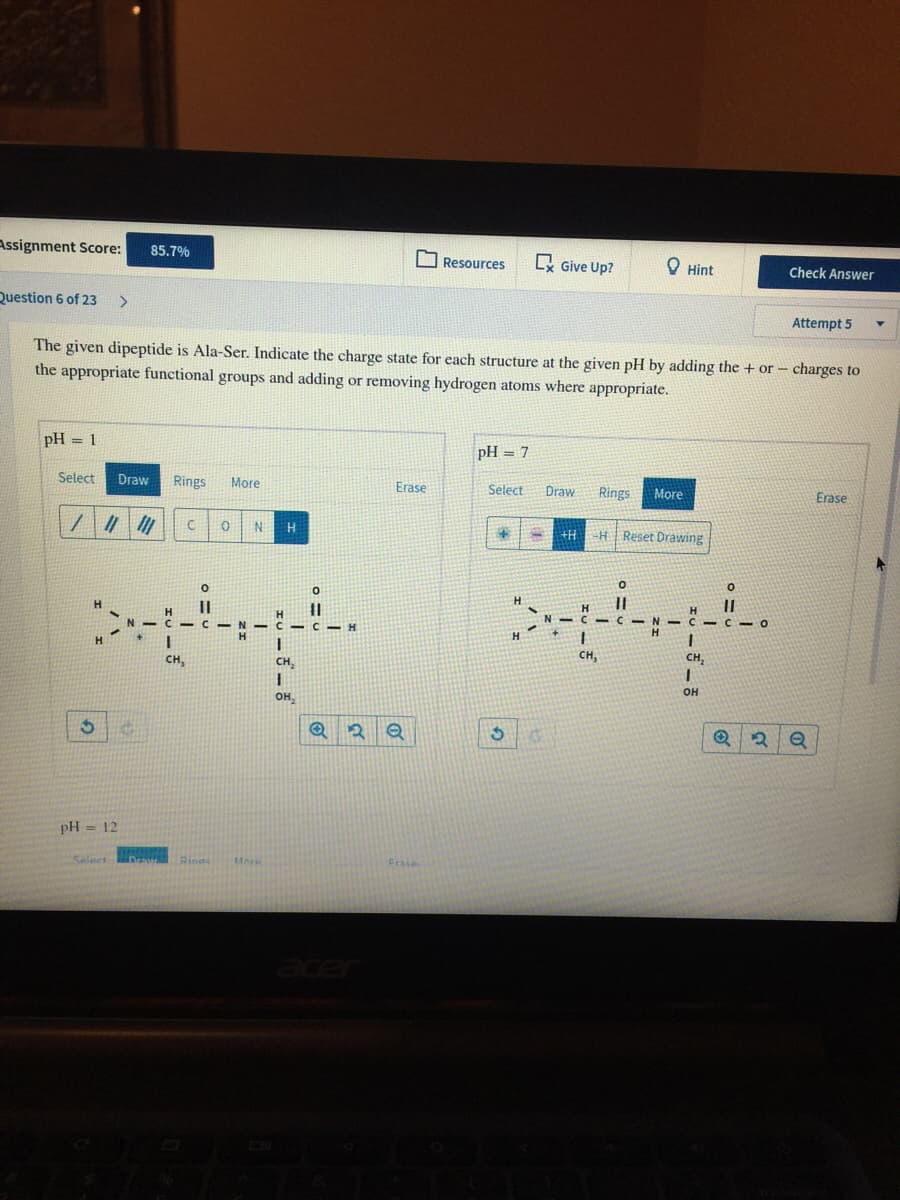
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or - charges to the appropriate functional groups and adding or removing hydrogen atoms where appropriate. pH = 1 pH = 7 %3D Select Draw Rings More Erase Select Draw Rings More Erase H HReset Drawing +H H. -CI CIO H H. CH, CH, CH, CH, он OH, pH = 12 of
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or - charges to the appropriate functional groups and adding or removing hydrogen atoms where appropriate. pH = 1 pH = 7 %3D Select Draw Rings More Erase Select Draw Rings More Erase H HReset Drawing +H H. -CI CIO H H. CH, CH, CH, CH, он OH, pH = 12 of
Chapter13: Dimensional Analysis/units Conversion
Section: Chapter Questions
Problem 1.3P
Related questions
Question
Transcribed Image Text:Assignment Score:
85,7%
Resources
x Give Up?
O Hint
Check Answer
Question 6 of 23
>
Attempt 5
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or - charges to
the appropriate functional groups and adding or removing hydrogen atoms where appropriate.
pH = 1
pH = 7
Select
Draw
Rings
More
Erase
Select
Draw
Rings
More
Erase
H.
+H
H Reset Drawing
H.
||
V C C-O
H
C - C
H.
CH,
CH,
CH,
CH,
он
он,
pH = 12
Selact
More
Frase
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

