I really dont understand this one I hope you can help me with this. Thank you so much. It has a 10 blanks.
I really dont understand this one I hope you can help me with this. Thank you so much. It has a 10 blanks.
Biochemistry
9th Edition
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Chapter1: Biochemistry: An Evolving Science
Section: Chapter Questions
Problem 1P
Related questions
Question
100%
I really dont understand this one I hope you can help me with this. Thank you so much. It has a 10 blanks.
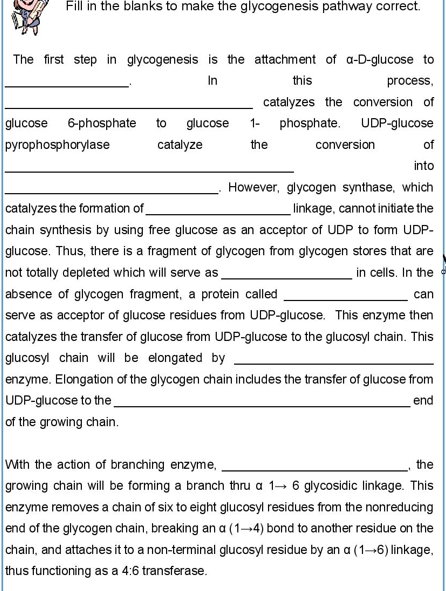
Transcribed Image Text:Fill in the blanks to make the glycogenesis pathway correct.
The first step in glycogenesis is the attachment of a-D-glucose to
In
this
process,
catalyzes the conversion of
UDP-glucose
glucose 6-phosphate to glucose
pyrophosphorylase
1- phosphate.
the
catalyze
conversion
of
into
However, glycogen synthase, which
linkage, cannot initiate the
catalyzes the formation of
chain synthesis by using free glucose as an acceptor of UDP to form UDP-
glucose. Thus, there is a fragment of glycogen from glycogen stores that are
not totally depleted which will serve as
in cells. In the
can
absence of glycogen fragment, a protein called
serve as acceptor of glucose residues from UDP-glucose. This enzyme then
catalyzes the transfer of glucose from UDP-glucose to the glucosyl chain. This
glucosyl chain will be elongated by
enzyme. Elongation of the glycogen chain includes the transfer of glucose from
UDP-glucose to the
end
of the growing chain.
With the action of branching enzyme,
the
growing chain will be forming a branch thru a 1-6 glycosidic linkage. This
enzyme removes a chain of six to eight glucosyl residues from the nonreducing
end of the glycogen chain, breaking an a (1-4) bond to another residue on the
chain, and attaches it to a non-terminal glucosyl residue by an a (1-6) linkage,
thus functioning as a 4:6 transferase.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781319114671
Author:
Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:
W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:
9781464126116
Author:
David L. Nelson, Michael M. Cox
Publisher:
W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul…
Biochemistry
ISBN:
9781118918401
Author:
Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:
WILEY

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Fundamentals of General, Organic, and Biological …
Biochemistry
ISBN:
9780134015187
Author:
John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:
PEARSON