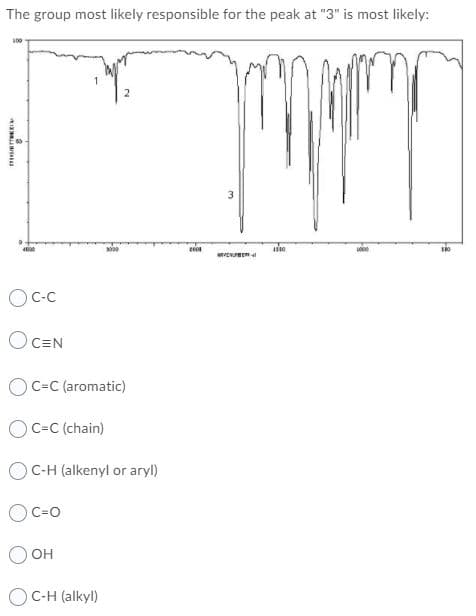
The group most likely responsible for the peak at "3" is most likely: OCEN OC=C (aromatic) OC=C (chain) OC-H (alkenyl or aryl) OC=0 O OH OC-H (alkyl)
Q: A compound was isolated as a minor constituent in an extract from garden cress. Its spectra are…
A:
Q: Of these groups, which would have the highest priority assignment in the Cahn-Ingold Prelog (R/S &…
A: Given groups are : Of these groups, which would have the highest priority assignment in the…
Q: Identify the bond signified by the IR peak in the box. 4000 3000 2000 O sp³ C-H O carboxylic O-H O…
A: IR spectroscopy is an important tool for the determination of the functional group in the compound.…
Q: What is the most likely m/z value for the base peak in the mass spectrum of 3-methylpentane?
A: The tallest peak in the mass spectra is known as the base peak. The structure of 3-Methylpentane is…
Q: Using the Molecular Ion to Identify a Compound Pent-1-ene and pent-1-yne are low-boiling…
A: In the mass spectrum, the heaviest ion (the one with the greatest m/z value) is likely to be the…
Q: The mass spectrum of compound A shows the molecular ion at m/z 85, an M + 1 peak at m/z 86 of…
A: The corresponding molecular formula C5H11N contains either one pi bond or one ring.
Q: The group most likely responsible for the peak at "3" is most likely: C-H (alkenyl or aryl) OCc-o…
A: In the given IR spectra, many peaks are observed. Peaks in IR indicates the presence of specific…
Q: Which compound correlates to the following mass spectrum? 100 20 50 75 100 125 150 m/z HO Br Br…
A: A mass spectrum is an important tool for the determination of the mass of a compound. A mass…
Q: How many carbon atoms are present in a compound that gives an (M+) peak of 100% and a (M+1) peak of…
A: Number of carbon atoms in a molecule = (Peak height of the M+1 peak/Peak height of M+) x 100
Q: m-Nitromethyl benzoate: Put in the molecular formula of the ions responsible for peaks at 135 and…
A: The inspecting of an unknown compound by its molecular formula is done by calculating the index of…
Q: How could you distinguish the mass spectrum of 2,2-dimethylpropane from those of pentane and…
A: Given distinguish the mass spectrum of 2,2-dimethylpropane Vs pentane and 2-methylbutane
Q: 7 Predict the masses and the structures of the most abundant fragments observed in the mass spectra…
A: Mass spectrometry- Mass spectrometry is an analytical technique which measures mass to charge (m/z)…
Q: Isoamyl acetate: Match the peaks to the appropriate number on the structure. A letter may correspond…
A:
Q: 100 43 80 60 27 29 28 40 41 42 39 15 58 10 20 30 40 50 60 Mass/charge (mle) Relative abundance (%)…
A: Given: Mass spectrum To find: Which compound will show this mass spectrum . Solution: Mass…
Q: The group most likely responsible for the peak at "1" is most likely: O C=N OC=C (aromatic) O OH O…
A: IR spectroscopy is also called as infrared spectroscopy. This is the called so because of the…
Q: 2) Indicate the type of bond responsible for the key absorbance peaks for 2-phenylethan-1-ol. (Be…
A: Solution -
Q: Question attached
A: IR spectroscopy is a technique which is used to determine the actual structure of a compound. It…
Q: Which of the following statements is correct regarding the mass spectrum of cyclohexanol (MW = 100)?…
A: A question based on mass spectrum that is to be accomplished.
Q: Which amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3…
A: In mass spectrometry, neutral and negatively charged particles are not detected.
Q: 8. What is the molecular formula of a saturated acyclic hydrocarbon that has an M peak at m/z = 100…
A: Mass spectrometry is an important tool for the determination of the molecular mass of the compound…
Q: Question attached
A: Infra-red (IR) spectroscopy is an analytical tool that is employed to determine the structure of the…
Q: Explain the peaks in the following mass spectra: a. 2-pentanone: m/e 86, 71, 57, 43 (100 %) b.…
A: The mass spectrum is plotted between the mass to charge ratio and the relative abundance of the…
Q: The mass spectrum of an alcohol will usually contain the following peak: O M+18 O M-16 O M-18 O M-15
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 2-Methylpentanal and 4-methyl-2-pentanone are constitutional isomers with the molecular formula…
A: Draw the structure of give isomer of 2-Maethyl pentanal and 4- methyl-2-pentanone molecule. Above…
Q: What distinguishes the mass spectrum of 2,2-dimethylpropane from the mass spectra of pentane and…
A: Mass spectrum is a plot of mass to charge ratio of the compound versus intensity. The mass to charge…
Q: How can the major product be identified in the infrared spectrum? A medium strong peak at 1674 cm1 O…
A:
Q: The mass spectrum of 2,3-dimethylpentane shows peaks at m/z = 57 and 43. Propose possible structures…
A: In the given question we have to draw the fragments observed in the mass spectrum. here, we have…
Q: 10. The two spectra below are of phenol and benzaldehyde. Assign them. 1.00 5.15 100 10
A: In proton NMR spectrum by seeing signal splitting and chemical shift we can identify the type of…
Q: What is the most likely m/z value for the base peak for 3-methylpentane? A. 15 В. 29 С. 43 D. 57 Е.…
A: Base peak: The peak due to a fragmented ion in the mass spectrum with highest relative abundance is…
Q: Consider the following mass spectrum, which exhibits a parent peak of 79 m/z, a "P+2" peak of 81…
A: Isomers have same molar mass, but different structures.
Q: Methyl benzoate: Match the peaks to the appropriate number on the structure. A letter may correspond…
A: Introduction: The NMR stands Nuclear Magnetic Resonance. NMR spectroscopy is used to determine the…
Q: this is mass spectrum. a. Methylcyclohexane b. 2-Methyl-pentene c. 2-methtl-2-hexanol d. ethyl…
A: Answer is below--
Q: A mass spectrum shows significant peaks at m/z = 87, 115, 140, and 143. Which of the following…
A: The IUPAC name of the given compound is
Q: Give the major peak that would be present in the functional group r following compounds: 2800 Y OH…
A: 1) C-H of alkane have =3000 - 2850 cm-1 (streching) 2) O-H of carboxylic group have frequency values…
Q: HC-CH-OH The mass spectrum below most likely belongs to which structure? Explain your answer. 45 100…
A: In mass spectrometry ,the molecule is breaked into fragments and m÷ z value is measured.
Q: The mass spectrum of 4,5,6-trimethylheptan-3-one (drawn below) has 3 major peaks at 113, 86 & 57…
A: A question based on mass spectrum that is to be accomplished.
Q: The molecular ion in the mass spectrum of 3-methyl-2-pentene appears at m/z 84. Propose a structural…
A: The m/z ratio peak is obtained from a cationic charged species generated during electron ionization…
Q: CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH3 100 - F 80 60 E 40 A 20 C 10 20 30 40 50 60 70 80 90 100 110 120…
A: N - octane molecular ion peak is at = 114. n - octane produces m,z at 15, 29, 43, 57, 71 ,85. (see…
Q: 1. Which one of the given compounds is consistent with the mass spectrum below? 100 BO 60 20 20 25…
A: Ans: 2-butanol. Molecular formula : C4H10O. Molecular ion = 74. Loss of methyl appears at m/z 59.…
Q: The spectrum shown belongs to one of the structural isomers below. Which one? 200 180 160 140 120…
A:
Q: The group most likely responsible for the peak at "1" is most likely: OC=C (chain) Occ OC-H (alkyl)…
A: Peaks in IR indicates the presence of specific functional groups, type of bond in it. In the given…
Q: which peaks can be expected when 5-methylhexane-2,4-dione is analyzed by mass spectrometry?
A: The structure of 5-Methylhexane-2,4-dione is as follows:
Q: After submitting a sample for mass spectral analysis you are given the following spectrum: 100 80 40…
A: Beta homolysis of 3-pentanol results m/z = 59. m/z 59 cleary indicates the compound is 3-pentanol.
Q: 12. The mass spectrum is usually depicted as a chart with increasing m/z from left to right. The…
A: (12) In the mass spectroscopy, the peak in most left is known as molecular ionic peak, it gives the…
Q: How many carbon atoms are expected to be in a hydrocarbon sample if it shows M and M+1 peak…
A: In case of carbon, M and M+1 peaks are expected as M peak for 12C & M+1 peak for 13C
Q: TReferences The molecular ion in the mass spectrum of 3-methyl-2-pentene appears at m/z 84. Propose…
A: Mass fragments of 3methyl 2-pentene.
Q: 9. The mass spectrum of which compound has an M+2 peak that has intensity that is about 25% of its…
A: Isotopic Aboundance of Chlorine is 75:25 for 35Cl and 37Cl repectively and M+2 peak is related to…
Q: 3. What structures correspond to theses peaks? Show your reason.
A: The structure of ketones can be derived using mass spectral analysis. In case of ketone, the…
Q: Label 3 peaks and what functional groups and their wavelength they could possibly correlate with in
A: A question based on IR spectroscopy that is to be accomplished.
6
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- For each structure below, use numbers to indicate chemically equivalent and distinct hydrogens,and make a table showing the predicted integration and multiplicity of each peak cluster.Complete the table below, and provide a structure of a compound that is consistent with the molecular formula C7H12O2 and the following 13C DEPT data. Please show your work. Also, place emphasis on the meaning of the "no peak no peak" dataFor norbornene and 3-bromobenzaldehyde: 1. determine the unique protons present 2. predict the multiplicities and draw its corresponding tree diagrams
- Make a sketch of the partial mass spectrum of 1-bromoheptane, showing the position of the peaks for: [Masses: H=1; C=12; Br=79 and Br=81 (isotopes)] i) The Molecular ion(s) ii) The fragments resulting from a split between carbons 3 and 4. iii) The fragments resulting from a split between carbons 5 and 6.Explain how the peaks in this spectrum occur. Especially the one at 77, which involves two fragmentations. The structure this corresponds to is 1-phenyl-1-butanoneIdentify the Campesterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.
- Choose the most possible compound for this FTIR spectrum. a. 2-pentanone b. 1-pentanol c. 2-methyl pentane d. 1-bromopentaneExplain all splitting patterns observed for absorptions between 0 and 8 ppm for the alcohol shown below. Which proton or protons will give rise to a doublet at 1.4 , which ones will give rise to a singlet at 2.7, which ones will give rise to a quartet at 4.7 and which ones will give rise to multiplets at 7.4?Assign the IR spectra given in Questions 1 - 3 to their corresponding structures from the ones below. The number in parentheses next to each possible answer corresponds to the structure's Chemical Abstracts Services (CAS) number. 1-bromobutane (109-65-9) 1-cyclopropylethan-1-one (765-43-5) 1-ethyl-3-methylbenzene (620-14-4) 1,2-dimethoxynenzene (91-16-7) 2,3-dimethylbutan-2-ol (594-60-5) 3-methylbutan-1-ol (123-51-3) 3,3-dimethylbutan-2-one (75-97-8) 4-benzylpiperidine (31252-42-3) 4-methoxybenzaldehyde (123-11-5) acetic anhydride (108-24-7) acetonitrile (75-05-8) acetyl chloride (75-36-5) anisole (100-66-3) benzaldehyde (100-52-7) benzoic acid (65-85-0) benzophenone (119-61-9) benzyl acetate (140-11-4) bromobenzene (108-86-1) cyclohex-2-en-1-one (930-68-7) cyclohexanone (108-94-1) cyclopent-2-en-1-one (930-30-3) diethylamine (109-89-7) dimethyl malonate (108-59-8) ethyl 2-cyanoacetate (105-56-6) ethynylbenzene (536-74-3) heptanoic acid…
- A prominent (M+ . -18) peak suggests that the compound might be what? A. Alkane B. Alcohol C. Ether D. KetonePent-1-ene and pent-1-yne are low-boiling hydrocarbons that have differentmolecular ions in their mass spectra. Match each hydrocarbon to its massspectrum.Which compound gives rise to a major [M − 18] peak in the mass spectrum? Group of answer choices A. 1-bromoheptane B. 4-methylheptane C. 2-heptanol D. 3-ethylheptane

