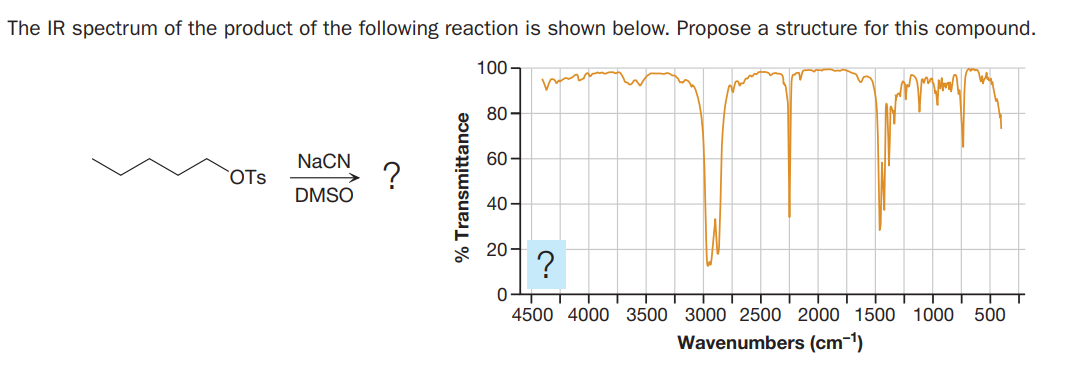
The IR spectrum of the product of the following reaction is shown below. Propose a structure for this compound. 100 - 80- NaCN 60- OTs DMSO 40- * 20- 4500 4000 3500 3000 2500 2000 1500 1000 500 Wavenumbers (cm-1)
Q: Identify the structure of the product in the following reaction. Provide a suitable mechanism with…
A: In this question, we will see the product and mechanism. You can see below.
Q: According to Table 23-1, the nitration of phenol results in a product mixture that is 50% ortho and…
A: The C-H bond weaker than the C-D bond due to this the elimination of D from the sigma complex is…
Q: (@) (€) Complete the following synthesis road map by (i) providing the structures of the major…
A: Here we have to write the reagent required and products formed in the sequence of reactions.
Q: 3. 9-Fluorinone (right) can be successfully reduced using sodium borohydride. When several drops of…
A: Reduction of ketone with sodium borohydride gives alcohol.
Q: [Grignard Synthesis of Benzoic Acid] Characterization 1. Explain melting point determination as a…
A: A question based on IR spectroscopy that is to be accomplished.
Q: Propose possible structures for compounds given the closed formula by analyzing the infrared…
A: Analysis of Ir spectrum
Q: The anisole derivative below undergoes a selective cross coupling reaction on only one of the two…
A:
Q: Describe stereoselective, regioselective, or chemoselective, the reactions below.
A: In case one we write the most stable structure in chair form, now their is two possibility to attack…
Q: Predict the major products when the following compounds are irradiated by light and treated with (1)…
A: INTRODUCTION: The structure of isopropyl benzene.
Q: Compare the following sets of small molecules and predict the order of their HOMO-LUMO gaps: a.…
A: The HOMO-LUMO gap decreases with increased conjugation within the system.
Q: Provide the structure of unknown compounds for the following reactions O3 CO2 H20 H2O HgSO/H2SO4 OH…
A: A scheme of incomplete organic reactions, which has to be completed with correct answer.
Q: of hex-3-ene will produce key absorbances on the product's IR spectrum of 1,718, 2,720, 2,820 and…
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: Choose the best efficient synthesis for transformation seen below * I- 1) dilute sulfuric acid; 2)…
A:
Q: Please propose possible mechanisms for the following reactions. H H H н н H H (i) ROOR H -CH3 -CD2H…
A: Interpretation- We have to propose the mechanism for the given following reactions which is given…
Q: Please provide the IR, 1H NMR and UV Spectra values of Benzoic Acid, 3-bromo-, methyl ester. And…
A: Structure of benzoic acid is: 1H-NMR spectrum of benzoic acid - 12.00-12.02 (s, 1H, A), 8.00-8.50…
Q: Heating the oxetane below to 560°C gives two products A and B (Scheme 2) A + CI Oxetane Scheme 2 The…
A:
Q: As we will learn in Chapter 17, reaction of (CH3)2CO with LIC≡CH followed by H2O affords compound D,…
A: In this reaction, Acetone reacts with lithium acetylide and tetrahedral lithium anion is formed,…
Q: Chemistry Treatment of this keto-aldehyde with KOH gives a compound C7H100 with the spectroscopic…
A:
Q: Predict the major products when the following compounds are irradiated by light and treated with (1)…
A: When alkylbenzenes are reacted with Br2 in the presence of light, the bromination occurs at the…
Q: Suzuki Cross Coupling Reaction Synthesis of an Unsymmetrical Biaryl via a Suzuki Cross Coupling…
A: 1H NMR: CHO resonates at 9-10 ppm IR: aromatic carbonyl group show IR absorption band ~1700 cm-1…
Q: You have reacted acetic anhydride with an alcohol in the presence of a small amount of concentrated…
A: Note : n-butanaol react with acetic anhydride and acid gives n-butyl acetate. 1735 cm-1 carbonyl…
Q: Outline mechanısm for the following reaction (1) PPh 3 CHO (11) NBULLI CH;OCH,Br (1) cyclohexanone…
A:
Q: What is the purpose for all of the following ? Carry out an electrophilic aromatic substitution…
A: Electrophilic aromatic substitution reaction are the subdtitution reaction in which an electrophilic…
Q: is this a one to one reaction? what does the chemical equation look like? Preparation of Acetanilide…
A: one to one reaction is that when from one reactant, one product is formed.
Q: S-ONa N=N (Figure Q11bi)
A:
Q: Discuss how you expect the FT-IR spectra of product 2 to change following treatment with LiAlH4 to…
A: The structure of (2) is given to be:
Q: Interpret the 1H NMR spectral provided for the Suzuki Cross-Coupling Reactions: Synthesis of an…
A: Suzuki coupling the coupling of the organohalides and organobornes in the presence of palladium…
Q: . The preparation of the sex pheromone of the boll worm moth, (E)-9,11-dodecadien-1-yl acetate from…
A: Organic compound synthesis:
Q: - For the following reactions, it was found that the diastereoselectivity decreased with the…
A: Diastereomers are a type of a stereoisomer. Diastereomers are defined as non-mirror image and…
Q: An organic lab student carried out the reaction of methylmagnesium iodide with acetone(CH3COCH3),…
A: The reaction is as follows CH3COCH3 + CH3MgI → (CH3)3COH Acetone methylmagnesium…
Q: what is the mechanisnm of these reactions? LiAIH, /THF 2) H30 HO CH3OH /AC HCL, Hz O pells HO…
A: The given reaction is reduction of ester and aldehyde by LiAlH4 followed by hydrolysis or workup.and…
Q: Suggest two possible structures of an unsaturated ketone that will be produced from the…
A: IR spectra deals results from the interaction of infrared radiation with matter by absorption. IR…
Q: OH NH, N2CI Anhydrous ZnCl2 NANO2 + HCI + NH, 300°C 0°-5°C Phenol aniline 1-chloro-2-phenyldiazene…
A: Please show the electron-flow mechanism of the general synthesis of Benzyl Chloride from Phenol.…
Q: Interpret the results of the UV-vis spectra and briefly explain why the 3-phenylpropionaldehyde…
A: UV-visible spectroscopy is an absorption spectroscopy. It uses light in visible regions and adjacent…
Q: The total synthesis of the natural product Laurenyne by Larry Overman et al (Ref.: J. Am. Chem. Soc.…
A: This is an example of Saegusa-Ito oxidation.
Q: Give mechanisms to explain the outcome of the following sequence of reactions, remembering to…
A:
Q: showing the flow of electrons. X A. 1. [(CH₂)₂CH)₂NLi 2. CH CH₂CH₂CH₂Br Provide a mechanism for each…
A:
Q: The IR spectrum of the product of the following reaction is shown below. Propose a structure for…
A: IR spectroscopy which is known as infrared spectroscopy helps to determine functional group and…
Q: Direct Cross-Dehydrogenative-Coupling (CDC) between Benzyl Ethers and Simple Ketones: DDQ O-RI 100°C…
A:
Q: -Butyne was treated with sodium amide followed by ethyl iodide. An IR spectrum of the product was…
A: Introduction : IR or Infrared Spectrum is the spectrum that deals with the infrared region of the…
Q: What are the major IR bands, 1H-nmr signals, And the m/z of the following compounds maleic…
A: IR, NMR, Mass spectral analysis of maleic anhydride is as follows.
Q: 13. Draw the structure of the compound described below Hydroboration-oxidation of an alkene, C,H12,…
A:
Q: A two-step synthesis of virstatin reactions are given. Give the yield and % yield of each synthesis.…
A: Since you have asked a question with multiple sub-part, we will solve the yield and percent yield…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Reaction of 2-methylpropanoic acid [(CH3)2CHCO2H] with SOCl2 followed by 2-methylpropan-1-ol forms X. X has a molecular ion at 144 and IR absorptions at 2965, 2940, and 1739 cm-1. Propose a structure for X.Is it possible to distinguish between the following compounds using the spectroscopic techniques covered in CHE331? If so, discuss how. If not, discuss why.Benzonitrile vs Benzaldehyde(a) UV spectroscopy(b) IR spectroscopy(c) Mass spectrometry(d) 13C NMR spectroscopy(e) 1H NMR spectroscopyWhat is the purpose for all of the following ? Carry out an electrophilic aromatic substitution reaction Prepare methyl m-nitrobenzoate by the nitration of methyl benzoate Verify the identity of a product through infrared spectroscopy
- Reaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G? We will learn about the details of this reaction in Chapter 22.Draw a structure consistent with the following set of data: a hydrocarbon with a molecular ion at m/z = 68 and IR absorptions at 3310, 3000−2850, and 2120cm−1.Reaction of pentanoyl chloride (CH3CH2CH2CH2COCl) with lithium dimethyl cuprate [LiCu(CH3)2] forms a compound J that has a molecular ion in its mass spectrum at 100, as well as fragments at m/z = 85, 57, and 43 (base). The IR spectrum of J has strong peaks at 2962 and 1718 cm-1. Propose a structure for J.
- Treatment of isobutene [(CH3)2C = CH2] with (CH3)3CLi forms a carbanion that reacts with CH2=O to form H after water is added to the reaction mixture. H has a molecular ion in its mass spectrum at m/z = 86, and shows fragments at 71 and 68. H exhibits absorptions in its IR spectrum at 3600–3200 and 1651 cm−1, and has the 1H NMR spectrum given below. Whatis the structure of H?Please provide the IR, 1H NMR and UV Spectra values of Benzoic Acid, 3-bromo-, methyl ester. And discuss how the results were obtain. For example the result for 3-(3-methoxyphenyl) propanoic acid would be written like this: 1 H NMR spectrumd (ppm in CDCI3 at 60 MHZ: 11.75 (Singlet, 1H, #7), 7.40-7.23 (Multiplet, 1H, #2), 6.85 (quartet, 3H, #1-3-6), 3.86 (Singlet, 3H, #5), 3.15-2.53 (Multiplet, 4H, #9-8). The discussion would be like this: the unknown molecule contained 12H. Revealing the presence of a peak of medium intensity at 11.75 ppm that we can associate with a carbocyclic acid. It also showed a high intensity peak at 3.86 ppm that we can associate with an ether, an alcohol or an amino group. Since the signal also shows that 3 protons are attached to it, it is therefore considered an ether (OCH3) in the unknown molecule. We also see two average peaks around 7.23 ppm and 6.85 ppm which show the presence of an aromatic.Reaction of C6H5CH2CH2OH with CH3COCl affords compound W, whichhas molecular formula C10H12O2. W shows prominent IR absorptions at3088–2897, 1740, and 1606 cm−1. W exhibits the following signals in its1H NMR spectrum: 2.02 (singlet), 2.91 (triplet), 4.25 (triplet), and 7.20–7.35(multiplet) ppm. What is the structure of W?
- An organic compound B with formula C6H14O has the following: IR Spectroscopy 2974 cm-1, 1080 cm-1 Mass Spectrometry 102 (M+), 87, 73 1H NMR Spectroscopy Eight signals at δ 1.10 (d, 3H), 1.13 (dd, 3H), 1.14 (dd, 3H), 1.59 (ddq, 1H), 1.60 (ddq, 1H), 3.19 (ddq, 1H), 3.51 (dq, 1H), 3.50 (dq, 1H). Compound B is obtained by the reaction of compound A with NaH followed by CH3CH2Br. The stereochemistry of A is "S" Using this information, deduce a plausible structure for Compound A with correct stereochemistry.Interpret the 1H NMR spectral provided for the Suzuki Cross-Coupling Reactions: Synthesis of an Unsymmetrical Biaryl via Suzuki Coupling Reaction.and label all major absorbances and what they indicate.c) FT-IR spectra of both nitro and nitrito-isomers were recorded immediately after preparation. Then small samples were exposed to heat (in oven) and their spectra were recorded again.

